Abstract
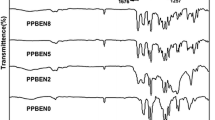
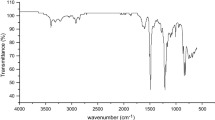
Two monomers, 4,4′-bis(4-phenoxybenzoyl)diphenyl (BPOBDP) and 2,6-diphenoxybenzonitrile (DPOBN), were conveniently prepared via simple synthetic procedures from commercially readily available materials. A series of novel poly(aryl ether ketone)s containing biphenylene moieties and pendant cyano groups were synthesized by the Friedel-Crafts acylation copolycondensation of isophthaloyl chloride (IPC) with a mixture of BPOBDP and DPOBN, over a wide range of BPOBDP/DPOBN molar ratios, in the presence of anhydrous AlCl3 and N-methylpyrrolidone (NMP) in 1,2-dichloroethane (DCE). The copolymers with 10–50 mol% DPOBN are semicrystalline and had higher T gs than those of the conventional PEEK and PEKK due to the incorporation of biphenylene moieties and pendant cyano groups. The copolymer VI with 50 mol% DPOBN had not only high T g of 168 °C, but also moderate T m of 333 °C, having good potential for melt processing. The copolymer VI had tensile strength of 103.4 MPa, Young’s modulus of 2.62 GPa, and elongation at break of 20.7 % and exhibited high thermal stability and excellent resistance to organic solvents.






Similar content being viewed by others
References
Staniland PA (1989) In: Allen G, Bevington JC (eds) Comprehensive polymer science, vol 5. Pergamon, Oxford, pp 483–497
Harris JE, Winslow PA, Botkin JH, Maresca LM, Clendinning RA, Cotter RJ, Matzner M, Kwiatkowski GT (1993) Macromolecules 26:2366–2371
(1992) Handbook of polymer synthesis, part A: Kricheldorf HR (ed). Marcel Dekker, New York, p 545
Nguyen HX, Ishida H (1987) Polym Compos 8:57–73
Ohno M, Takata T, Endo T (1995) J Polym Sci Polym Chem Ed 33:2647–2655
Khan S, Pratte JF, Chang IY, Krueger WH (1990) Int SAMPE Symp Exhib 35:1579–1593
Rao VL, Sabeena PU, Saxena A, Gopalakrishnan C, Krishnan K, Ravindran PV, Ninan KN (2004) Eur Polym J 40:2645–2651
Rose JB (1986) In: Seymour RB, Kirshenbaum GS (eds) High performance polymers: their origin and development. Elsevier, New York, pp 187–193
Cotter RJ (1995) Engineering plastics: a handbook of polyarylethers. Gordon and Breach Science Publishers, Basel
Yonezawa N, Okamoto A (2009) Polym J 41:899–928
Singh R, Hay AS (1991) Macromolecules 24:2637–2639
Chan KP, Wang Y, Hay AS (1995) Macromolecules 28:653–655
Ben-Haida A, Colquhoun HM, Hodge P, Williams DJ (2006) Macromolecules 39:6467–6472
Liu B, Robertson GP, Kim DS, Guiver MD, Hu W, Jiang Z (2007) Macromolecules 40:1934–1944
Wang J, Gao Y, Hlil AR, Hay AS (2008) Macromolecules 41:298–300
Colquhoun HM, Hodge P, Paoloni FPV, Terry McGrail P, Cross P (2009) Macromolecules 42:1955–1963
Sakaguchi Y, Tokai M, Kato Y (1993) Polymer 34:1512–1515
Zolotukhin MG, Dosieret M, Fougnies C, Villers D, Gileva NG, Fatykhov AA (1995) Polymer 36:3575–3583
Krishnaswamy RK, Kalika DS (1996) Polymer 37:1915–1923
Zolotukhin MG, Rueda DR, Balta Calleja FJ, Cagiao ME, Sedova EA, Gileva NG (1997) Polymer 38:1471–1476
Rueda DR, Zolotukhin MG, Cagiao ME, Balta Calleja FJ, Villers D, Dosiere M (1996) Macromolecules 29:7016–7021
Zolotukhin MG, Colquhoun HM, Sestiaa LG, Rueda DR, Flot D (2003) Macromolecules 36:4766–4771
Zhang W, Ji X, Yuan J, Na H, Wang J, Wu Z (1993) Chem J Chin Univ 14:738–740
Ritter H, Thorwirth R, Muller G (1993) Makromol Chem 194:1469–1481
Ohno M, Takata T, Endo T (1994) Macromolecules 27:3447–3448
Zhang Y, Sun X, Xu R, Niu Y, Wang G, Jiang Z (2006) Mater Chem Phys 99:465–469
Liu B, Wang G, Hu W, Jin Y, Chen C, Jiang Z, Zhang W, Wu Z, Wei Y (2002) J Polym Sci Polym Chem Ed 40:3392–3398
Wu Z, Zheng Y, Yan H, Nakamura T, Nozawa T, Yosomiya R (1989) Angew Makromol Chem 173:163–181
Yu X, Zheng Y, Wu Z, Tang X (1990) J Appl Polym Sci 41:2649–2654
Cao J, Su W, Chang C, Zheng Y, Wu Z (1994) Acta Polym Sin 4:477–481
Cao J, Su W, Wu Z, Kitayama T, Hatada K (1994) Polymer 35:3549–3556
Konnecke K (1992) Angew Makromol Chem 198:15–29
Kruger KN, Zachmann HG (1993) Macromolecules 26:5202–5208
Cai M, Xiao F, Ding N, Song C (2009) Polym Adv Technol 20:981–986
Cai M, Zhu M, Wang P, Song C (2010) Polymer 51:1293–1300
Jiang J, Ding N, Cai M (2011) Polym Int 60:240–246
Huang B, Zhu M, Cai M (2011) J Appl Polym Sci 119:647–653
Cai M, Chen M, Yu Y, Song C (2013) Polym Adv Technol 24:466–472
Wang Y, Wang P, Xu Q, Yan T, Cai M (2014) J Polym Res 21:533–540
Kricheldorf HR, Meier J, Schwarz G (1987) Makromol Chem Rapid Commun 3:529–532
Mercer FW, McKenzie MT, Bruma M, Schulz B (1994) Polym Int 33:399–407
Mercer FW, Easteal A, Bruma M (1997) Polymer 38:707–714
Janson V, Gors HC (1984) Preparation of aromatic polymer. PCT Int WO 8:403,891
Bonner WH (1962) Aromatic polyketones and preparation. US Patent 3065205
Olah GA (ed) (1964) Friedel-Crafts and related reactions, Vol. 3. Interscience, New York, pp 1003–1014
Tan LK, Brownstein S (1982) J Org Chem 47:4737–4739
Gileva NG, Zolotukhin MG, Salazkin SN, Sultanova VS, Hörhold H-H, Raabe D (1988) Acta Polym 39:452–455
Botkin JH, Cotter RJ, Matzner M, Kwiatkowaki GT (1993) Macromolecules 26:2372–2376
Acknowledgments
The authors thank the National Natural Science Foundation of China (Project 21264010) and the Natural Science Foundation of Jiangxi Province of China (Project 20132BAB203015) for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, X., Yan, T., Xu, Q. et al. Synthesis and characterization of novel poly(aryl ether ketone)s containing biphenylene moieties and pendant cyano groups. J Polym Res 23, 24 (2016). https://doi.org/10.1007/s10965-015-0904-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-015-0904-2