Abstract
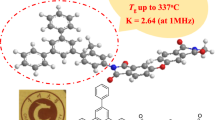
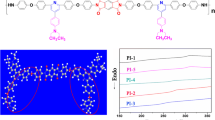
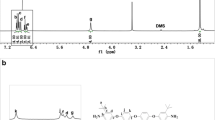
A series of novel organo-soluble and optical transparent polyimides were synthesized via conventional one-step polycondensation from a novel diamine, 3,3′-diterbutyl-4,4′-diaminodiphenyl-4″-morpholinophenylmethane (TAMPM) and various commercial aromatic dianhydrides. The structures of monomer and polymers were confirmed by FT-IR and NMR. The solubility, thermal stability, optical transparency, dielectric and hydrophobic properties of polyimides were investigated. The results showed polymers presented excellent solubility in common organic solvents, such as N,N-dimethylformamide (DMF), N,N-dimethyl acetamide (DMAc), dimethyl sulfoxide (DMSO), N-methyl-2-pyrrolidone (NMP), chloroform (CHCl3), tetrahydrofuran (THF) and dichloromethane (CH2Cl2). The polymers exhibited prominent thermal stability and high optical transparency with the glass transition temperature (T g) in the range of 311–355 °C, and the cutoff wavelengths in the range of 287–344 nm. Furthermore, all of the polymers also presented low dielectric constants in the range of 2.85–3.16 at 1 MHz, and outstanding hydrophobic properties with the contact angle in the rang of 85.1–94.2 o.










Similar content being viewed by others
Abbreviations
- TAMPM:
-
3,3′-diterbutyl-4,4′-diaminodiphenyl-4″-morpholinophenylmethane
- PMDA:
-
Pyromellitic dianhydride
- BPDA:
-
3,3′,4,4′-biphenyltetracarboxylic dianhydride
- ODPA:
-
4,4′-oxydiphthalic anhydride
- BTDA:
-
3,3,4,4′-benzophenone tetracarboxylic dianhydride
- 6FDA:
-
(4,4-hexafluoroisopropylidene)-diphthalic anhydride
References
Ding MX (2007) Prog Polym Sci 32:623–668
Jeon YW, Lee DH (2015) Environ Eng Sci 32:71–85
Liaw DJ, Wang KL, Huang YC, Lee KR, Lai JY, Ha CS (2012) Prog Polym Sci 37:907–974
Kurosawa T, Higashihara T, Ueda M (2013) Polym Chem 4:16–30
Ghosh A, Sen SK, Banerjee S, Voit B (2012) RSC Adv 2:5900–5926
Huang XH, Huang W, Zhou YF, Yan DY (2011) Chin J Polym Sci 29:506–512
Zhao JJ, Gong CL, Zhang SJ, Shao Y, Li YF (2010) Chin Chem Lett 21:277
Mehdipour-Ataei S, Sarrafi Y, Hatami M (2004) Eur Polym J 40:2009–2015
Yi L, Li CY, Huang W, Yan DY (2014) J Polym Res 21:572
Huang XH, Huang W, Liu JY, Meng LL, Yan DY (2012) Polym Int 61:1503–1509
Zhang SJ, Bu QQ, Li YF, Gong CL, Xu XY, Li H (2011) Mater Chem Phys 128:392–399
Zhang SJ, Li YF, Ma T, Zhao JJ, Xu XY, Yang FC, Xiang XY (2010) Polym Chem 1:485–493
Xia SL, Yi LF, Sun Z, Wang YH (2013) J Polym Res 20:219
Ghaemy M, Khajeh S (2011) Chin J Polym Sci 29:465–474
Li YQ, Chu YY, Fang RC, Ding SJ, Wang YL, Shen YZ, Zheng AM (2012) Polymer 53:229–240
Gong SM, Liu M, Xia SL, Wang YH (2014) J Polym Res 21:542
Liu CJ, Pei XL, Huang XH, Wei C, Sun XY (2015) Chin J Chem 33:277–284
Thiruvasagam P (2012) J Polym Res 19:9965
Huang YC, Wang KL, Lee WY, Liao YA, Liaw DJ, Lee KR, Lai JY (2015) J Polym Sci A Polym Chem 53:405–412
Liaw DJ, Wang KL, Chang FC, Lee KR, Lai JY (2007) J Polym Sci A Polym Chem 45:2367–2374
Barzic AI, Stoica I, Fifere N, Vlad CD, Hulubei C (2013) J Polym Res 20:130
Chen GF, Pei XL, Liu JT, Fang XZJ (2013) J Polym Res 20:159
Guo YZ, Shen DX, Ni HJ, Liu JG, Yang SY (2013) Prog Org Coat 76:768–777
Li H, Zhang SJ, Gong CL, Liang Y, Qi ZG, Li YF (2015) Polym Int 64:352–360
Tapaswi PK, Choi MC, Nagappan S, Ha CS (2015) J Polym Sci A Polym Chem 53:479–488
Gao YF, Zhou YM, He M, Wang HY, Cui YP, Zhang T (2014) Des Monomers Polym 17:590–600
Wang XM, Liu F, Lai JC, Fu ZQ, You XZ (2014) J Fluor Chem 164:27–37
Guan Y, Wang DM, Song GL, Dang GD, Chen CH, Zhou HW, Zhao XG (2014) Polymer 55:3634–3641
Wang JY, Liu C, Su GX, Jian XG (2012) High Perform Polym 24:356–365
Ishii J, Yokotsuka H, Saito T, Hasegawa M (2011) J Photopolym Sci Technol 24:287–291
Pakhuruddin MZ, Ibrahim K, Aziz AA (2013) J Optoelectron Adv Mat 7:377–380
Zhao XJ, Liu JG, Rui JM, Fan L, Yang SY (2007) J Appl Polym Sci 103:1442–1449
Qiu ZM, Wang JH, Zhang QY, Zhang SB, Ding MX, Gao LX (2006) Polymer 47:8444–8452
Matsumoto T, Kurosaki T (1997) Macromolecules 30:993–1000
Dine-Hart RA, Wright WW (1971) Makromol Chem 143:189–192
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 51163003 and 21264005), the fund of Guangxi Natural Science Foundation (Nos. 2014GXNSFAA118040 and 2013GXNSFDA019008), Guangxi Ministry-Province Jointly-Constructed Cultivation Base for State Key Laboratory of Processing for Non-ferrous Metal and Featured Materials (13KF-3), and Guangxi Funds for Specially-appointed Expert.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, X., Mei, M., Liu, C. et al. Synthesis and characterization of novel highly soluble and optical transparent polyimides containing tert-butyl and morpholinyl moieties. J Polym Res 22, 169 (2015). https://doi.org/10.1007/s10965-015-0820-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-015-0820-5