Abstract
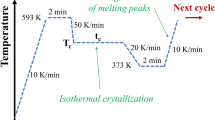
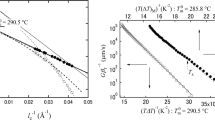
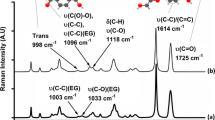
The crystallization and melting behaviors of poly(trimethylene terephthalate) (PTT) have been studied by differential scanning calorimetry (DSC), wide-angle X-ray diffraction (WAXD), and solid-state NMR. At certain crystallization temperatures (Tc) for a given time, the isothermally crystallized PTT exhibits two melting endotherms, which is similar to that of PET and PBT. At higher crystallization temperature (Tc = 210 °C), the low-temperature endotherm is related to the melting of the original crystals, while the high-temperature endotherm is associated with the melting of crystals recrystallized during the heating. The peak temperatures of these double-melting endotherms depend on crystallization temperature, crystallization time, and cooling rate from the melt as well as the subsequent heating rate. At a low cooling rate (0.2 °C/min) or a high heating rate (40 °C/min), these two endotherms tend to coalesce into a single endotherm, which is considered as complete melting without reorganization. WAXD results confirm that only one crystal structure exists in the PTT sample regardless of the crystallization conditions even with the appearance of double melting endotherms. The results of NMR reveal that the annealing treatment increases proton spin lattice relaxation time in the rotation frame, T1ρ H, of the PTT. This phenomenon suggests that the mobility of the PTT molecules decreases after the annealing process. The equilibrium melting temperature (T om ) determined by the Hoffman-Weeks plot is 248 °C.
Similar content being viewed by others
References
T. H. Ng and H. L. Williams, Makromol. Chem., 182, 3323 (1981).
C. C. Gonzalez, J. M. Perena and A. Bello, J. Polym. Sci., Polym. Phys. Ed., 26, 1397 (1988).
T. Imamura, T. Sato and T. Matsumoto, Jpn. Pat. 08,232,117 (1996).
K. Dangayach, H. Chuah, W. Gergen, P. Dalton and F. Smith, Plastics-Saving Planet Earth, 55th ANTEC Proc., 2097 (1997).
K. Tsumashima, and M. Suzuki, Jpn. Pat. 08,104,763 (1996).
Z. Gao, A. Molnar, F. G. Morin and A. Eisenberg, Macromolecules, 25, 6460 (1992).
V. T. McBrierty, D. C. Douglass and T. K. Kwei, Magn. Reson. in Chem., 32, 853 (1994).
D. L. Vanderhar and J. D. Barnes, Macromolecules, 27, 2826 (1994).
P. P. Chu, J. M. Huang, H. D. Wu, C. R. Chiang and F. C. Chang, J. Polym. Sci., Polym. Phys. Ed., 37, 1155 (1999).
J. M. Huang, P. P. Chu and F. C. Chang, Polymer, 41, 1741 (2000).
R. J. Samuels, J. Polym. Sci., Polym. Phys. Ed., 13, 1417 (1975).
W. M. Prest and D. J. Luca, J. Appl. Phys., 46, 4136 (1975).
R. C. Roberts, Polymer, 10, 17 (1969).
G. E. Sweet and J. P. Bell, J. Polvm. Sci., Polym. Phys. Ed., 10, 273 (1972).
M. Todoki and T. Kawaguchi, J. Polym. Sci., Polym. Phys. Ed., 15, 1067 (1977).
J. Boon, G. Challa and D. W. Krevelen, J. Polym. Sci., Polym. Phys. Ed., 6, 1791 (1968).
Y. Lee and R. S. Porter, Macromolecules, 20, 1336 (1987).
Y. Lee, R. S. Porter and J. S. Lin, Macromolecules, 22, 1756 (1989).
M. Pyda, A. Boller, J. Grebowicz, H. Chuah, V. Lebedev and B. Wunderlich, J. Polym. Phys. Ed., 36, 2499 (1998).
S. Peres, P. D. Suzie, J. F. Revol and F. Brisse, Polymer, 20, 419 (1979).
R. E. Robertson and M. E. Michols, J. Polym. Sci., Polym. Phys. Ed., 30, 755 (1992).
J. D. Hoffman and J. J. Weeks, J. Chem. Phys., 42, 4301 (1965).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, JM., Ju, MY., Chu, P.P. et al. Crystallization and melting behaviors of poly(trimethylene terephthalate). J Polym Res 6, 259–266 (1999). https://doi.org/10.1007/s10965-006-0096-x
Issue Date:
DOI: https://doi.org/10.1007/s10965-006-0096-x