Abstract
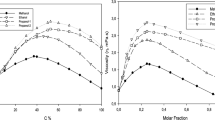
Shear viscosity deviations Δη have been investigated by using density (ρ) and kinematic viscosity (ν) measurements for isobutyric acid + water (IBA + W) mixtures over the entire range of mole fractions at atmospheric pressure and at two temperatures (301.15 and 315.15 K). This study extends the temperature range from the five other temperatures investigated in a previous work, 1.055 K≤(T−T c )≤14.055 K, both far from and close to the critical temperature. This system exhibits very large positive values of Δη due to increased hydrogen bonding interactions and the correlation length between unlike molecules in the critical region, and to very large differences between the molar volumes of the pure components at low temperatures. The results were also fitted with the Redlich–Kister polynomial equations and the recently proposed Herráez correlation equation. Comparisons between the two models at different temperatures and number of parameters are discussed. We note that, in this system where the shear viscosity η as a function of mole fraction (x 1) of IBA presents a maximum, experimental data are in agreement with the two correlation models when more than three parameters are employed, especially for temperatures far from the critical temperature.
Similar content being viewed by others
References
Toumi, A., Bouanz, M.: Critical behavior of the binary-fluid isobutyric acid-water with added ion (K+, Cl−). Eur. Phys. J. E 2, 211–216 (2000). doi:10.1007/PL00013667
Toumi, A., Bouanz, M., Gharbi, A.: Coexistence curves of the binary mixture isobutyric acid-water with added ions (K+, Cl−). Chem. Phys. Lett. 362, 567–573 (2002). doi:10.1016/S0009-2614(02)01107-7
Toumi, A., Bouanz, M.: Effect of the (K+, Cl−) ions on the order parameters and on the Lorenz-Lorentz relation in the isobutyric acid-water critical mixture. J. Mol. Liq. 122, 74–83 (2005). doi:10.1016/j.molliq.2005.05.001
Ouerfelli, N., Bouanz, M.: Excess molar volume and viscosity of isobutyric acid + water binary mixtures near and far away from the critical temperature. J. Solut. Chem. 35(1), 121–137 (2006). doi:10.1007/s10953-006-8944-1
Greer, S.C.: Coexistence curves at liquid-liquid critical points: Ising exponents and extended scaling. Phys. Rev. A 14, 1770–1780 (1976). doi:10.1103/PhysRevA.14.1770
Venkataraman, T.S., Narducci, L.M.: Critical properties of the binary fluid system—isobutyric acid–water. J. Phys. C, Solid State Phys. 10, 2849–2861 (1977). doi:10.1088/0022-3719/10/15/019
Wong, N.C., Knobler, C.M.: Light scattering studies of phase separation in isobutyric acid + water mixtures. J. Chem. Phys. 69(2), 725–735 (1978). doi:10.1063/1.436582
Chou, Y.C., Goldburg, W.I.: Phase separation and coalescence in critically quenched isobutyric acid + water and 2,6-lutidine + water mixtures. Phys. Rev. A 20, 2105–2113 (1979). doi:10.1103/PhysRevA.20.2105
Chou, Y.C., Goldburg, W.I.: Angular distribution of light scattered from critically quenched liquid mixtures. Phys. Rev. A 23, 858–964 (1981). doi:10.1103/PhysRevA.23.858
Beysens, D., Bourgou, A., Calmettes, P.: Experimental determinations of universal amplitude combinations for binary fluids. I. Statics. Phys. Rev. A 26, 3589–3609 (1982). doi:10.1103/PhysRevA.26.3589
Maher, J.V., Goldburg, W.I., Phol, D.W., Lanz, M.: Critical behavior in gels saturated with binary liquid mixtures. Phys. Rev. Lett. 53, 60–63 (1984). doi:10.1103/PhysRevLett.53.60
Andrew, W.V., Khoo, T.B.K., Jacobs, D.T.: Testing the Lorentz–Lorenz relation in the near-critical binary fluid mixture isobutyric acid and water. J. Chem. Phys. 85(7), 3985–3991 (1986). doi:10.1063/1.450920
Xia, K.Q., Maher, J.V.: Dynamic light scattering from binary-liquid gels. Phys. Rev. A 37, 3626–3629 (1988)
Berg, R.F., Moldover, M.R.: Critical exponent for the viscosity of four binary liquids. J. Chem. Phys. 89(6), 3694–3704 (1988)
Jacobs, D.T., Greer, S.C.: Amplitude of the anomaly in the mass density near a liquid-liquid critical point. Phys. Rev. E 54, 5358–5363 (1996)
Venkatesu, P.: Polymer modifies the critical region of the coexisting liquid phases. J. Phys. Chem. B 110, 17339–17346 (2006)
Herráez, J.V., Belda, R., Diez, O., Herráez, M.: An equation for the correlation of viscosities of binary mixtures. J. Sol. Chem. 37(2), 233–248 (2008)
Redlich, O., Kister, A.T.: Algebraic representation of thermodynamic properties and the classification of solutions. Ind. Eng. Chem. 40, 345–348 (1948)
Beysens, D., Bourgou, A.: Accurate determination of alpha and Delta exponents in critical binary fluids by refractive-index measurements. Phys. Rev. A 19, 2407–2412 (1979)
Physikalisch-Technische Bundesanstalt, 2nd edn., licence, Nr. 13/1985, p. 6, Chempro GmbH, D6450 Hanau (1985)
Ouerfelli, N., Bouanz, M.: A shear viscosity study of cerium(III) nitrate in concentrated aqueous solutions at different temperatures. J. Phys. C, Solid State Phys. 8, 2763–2774 (1996)
Van Bib, B.: International Critical Tables, vol. 3. McGraw-Hill, New York (1928), p. 27
Levitte, B.P.: Findlay’s Practical Physical Chemistry, 9th edn. Longman, New York (1973)
Weast, R.C., Astle, M.J.: In: CRC Handbook of Chemistry and Physics, 60th edn. CRC Press, Boca Raton (1980)
Woermann, D., Sarholz, W.: Die Viskosität kritischer Mischungen Experimentelle Untersuchungen am System Isobuttersäure-Wasser. Ber. Bunsenges. Z. Phys. Chem. 69, 319–326 (1965)
Beysens, D., Bourgou, A., Paladin, G.: Experimental determinations of universal amplitude combination for binary fluids. II. Dynamics. Phys. Rev. A 30, 2686–2703 (1984)
Fort, R.J., Moore, W.R.: Viscosities of binary liquid mixtures. Trans. Faraday Soc. 62, 1112–1119 (1966)
Colter, A.K., Grunwald, E.: Application of differential refractometry to the measurement of association constants for molecular complex formation. J. Phys. Chem. 74, 3637–3642 (1970)
Erdey Gruz, T.: Transport Phenomena in Aqueous Solutions. Hilger, London (1958)
Abramowitz, M., Stegun, I.A. (eds.): Handbook of Mathematical Functions with Formulas, Graphs, and Mathematical Tables, 9th edn. Dover, New York (1972)
Tomiska, J.: Calculation of the thermodynamics of ternary systems based upon experimental data of e.m.f. measurements. CALPHAD 5, 81–92 (1981)
Tomiska, J.: Zur Konversion der Anpassungen thermodynamischer Funktionen mittels einer Reihe Legendre’scher Polynome und der Potenzreihe. CALPHAD 5, 93–102 (1981)
Tomiska, J.: Mathematical conversions of the thermodynamic excess functions represented by the Redlich–Kister expansion and by the Chebyshev polynomial series to power series representations and vice-versa. CALPHAD 8, 283–294 (1984)
Peralta, R.D., Infante, R., Cortez, G., Ramírez, R.R., Wisniak, J.: Densities and excess volumes of binary mixtures of 1,4-dioxane with either ethyl acrylate, or butyl acrylate, or methyl acrylate, or styrene at T=298.15 K. J. Chem. Thermodyn. 35, 239–250 (2003)
Peralta, R.D., Infante, R., Cortez, G., Elizalde, L.E., Wisniak, J.: Density, excess volumes and partial volumes of the systems of p-xylene + ethyl acrylate, butyl acrylate, methyl methacrylate, and styrene at 298.15 K. Thermochim. Acta 421, 59–68 (2004)
Wisniak, J., Peralta, R.D., Infante, R., Cortez, G., Lopez, R.G.: Densities, isobaric thermal compressibilities and derived thermodynamic properties of the binary systems of cyclohexane with allyl methacrylate, butyl methacrylate, methacrylic acid, and vinyl acetate at T=(298.15 and 308.15) K. Thermochim. Acta 437, 1–6 (2005)
Wisniak, J., Villarreal, I., Peralta, R.D., Infante, R., Cortez, G., Soto, H.: Densities and volumes of mixing of the ternary system toluene + butyl acrylate + methyl methacrylate and its binaries at 298.15 K. J. Chem. Thermodyn. 39, 88–95 (2007)
Arfken, G.: Mathematical Methods for Physicists, 3rd edn. Academic Press, Orlando (1985)
Dattoli, G., Srivastava, H., Zhukovsky, K.: Orthogonality properties of the Hermite and related polynomials. J. Comput. Appl. Math. 182(1), 165–172 (2005)
Jones, G., Dole, M.: The viscosity of aqueous solutions of strong electrolytes with special reference to barium chloride. J. Am. Chem. Soc. 51, 2950–2964 (1929)
Nakagawa, T.: Is viscosity B coefficient characteristic for solute-solvent interaction? J. Mol. Liq. 63, 303–316 (1995)
Ruiz Holgado, M.E., de Schaefer, C.R., de Araneibia, E.L.: Viscosity study of 1-propanol with polyethylene glycol 350 monomethyl ether systems at different temperatures. J. Mol. Liq. 79, 257–267 (1999)
Longuet-Higgins, H.C.: The statistical thermodynamics of multicomponent systems. Proc. R. Soc. (Lond.) 205A, 247–269 (1951)
Matsubayashi, N., Nakahara, M.: Dynamics in regular solution: concentration and viscosity dependence of orientational correlation of a benzene molecule. J. Chem. Phys. 94, 653–661 (1991)
Van Ness, H.C., Abbott, M.M.: Classical Thermodynamics of Nonelectrolyte Solutions. McGraw-Hill, New York (1982)
Grunberg, L., Nissan, A.H.: Mixture law for viscosity. Nature 164, 799–800 (1949)
Hind, R.H., McLaughlin, E., Ubbelohde, A.R.: Structure and viscosity of liquids-camphor and pyrene mixtures. Trans. Faraday Soc. 56, 328–334 (1960)
Katti, P.K., Chaudhri, M.M.: Viscosities of binary mixtures of benzyl acetate with dioxane, aniline and m-cresol. J. Chem. Eng. Data 9, 442–443 (1964)
McAllister, R.A.: The viscosities of liquid mixtures. Am. Inst. Chem. Eng. 6, 427–431 (1960)
Heric, E.L., Brewer, J.C.: Viscosity of some binary liquid nonelectrolyte mixtures. J. Chem. Eng. Data 12, 574–583 (1967)
Auslander, G.: Prediction of the McAllister model parameters from pure component properties for liquid binary n-alkane systems. Br. Chem. Eng. 9, 610–618 (1964)
Toumi, A., Bouanz, M.: Volumetric and refractive index properties of isobutyric acid–water binary mixtures at temperatures ranging from 300.15 to 313.15 K. J. Mol. Liq. 139, 55–60 (2008)
Falkenhagen, H.: Theorie der Elektrolyte. Hirzel, Leipzig (1971)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ouerfelli, N., Kouissi, T., Zrelli, N. et al. Competition of Viscosity Correlation Equations in Isobutyric Acid + Water Binary Mixtures Near and Far Away from the Critical Temperature. J Solution Chem 38, 983–1004 (2009). https://doi.org/10.1007/s10953-009-9423-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-009-9423-2