Abstract
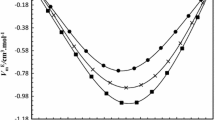
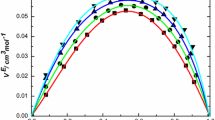
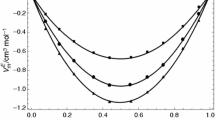
Densities of the binary systems of toluene with butyl methacrylate, allyl methacrylate, methacrylic acid, and vinyl acetate have been measured as a function of composition at 20, 30 and 40 °C at atmospheric pressure, using an Anton Paar DMA 5000 oscillating U-tube densimeter. The excess molar volumes are negative for the system toluene + butyl methacrylate and positive for the three other binaries, and become more so as the temperature increases from 20 to 40 °C. The system toluene + allyl methacrylate presents near ideal behavior. The apparent volumes were used to calculate values of the partial excess molar volumes at infinite dilution. The excess coefficient of thermal expansion is positive for the four binary systems. The calculated excess molar volumes were correlated with the Redlich–Kister equation and with a series of Legendre polynomials. An explanation of the results is given based by the FT-IR (ATR) and 13C NMR spectra of equimolar mixtures of the different systems.
Similar content being viewed by others
References
Peralta, R.D., Infante, R., Cortez, G., Villarreal, L., Wisniak, J.: Volumetric properties of cyclohexane with ethyl acrylate, butyl acrylate, methyl methacrylate, and styrene at 298.15 K. Thermochim. Acta 390, 47–53 (2002)
Peralta, R.D., Infante, R., Cortez, G., Torres-Lubián, J.R., Wisniak, J.: Volumetric properties of 1,2-dimethylbenzene + ethyl acrylate, butyl acrylate, methyl methacrylate, and styrene at 298.15 K. Thermochim. Acta 402, 247–252 (2003)
Peralta, R.D., Infante, R., Cortez, G., Cisneros, A., Wisniak, J.: Densities and excess volumes of benzene with ethyl acrylate, butyl acrylate, methyl methacrylate, and styrene at 298.15 K. Thermochim. Acta 398, 39–46 (2003)
Peralta, R.D., Infante, R., Cortez, G., Rodríguez, O., Wisniak, J.: Volumetric properties of toluene with ethyl acrylate, butyl acrylate, methyl methacrylate, and styrene at 25 °C. J. Solution Chem. 31, 175–186 (2002)
Peralta, R.D., Infante, R., Cortez, G., López, R.G., Wisniak, J.: Volumetric properties of 1,1-dimethylethyl methyl ether with ethyl acrylate, butyl acrylate, methyl methacrylate, and styrene at 298.15 K. Int. J. Thermophys. 24, 173–183 (2003)
Peralta, R.D., Infante, R., Cortez, G., Ramírez, R.R., Wisniak, J.: Densities and excess volumes of binary mixtures of 1,4-dioxane with either ethyl acrylate, or styrene at T=298.15 K. J. Chem. Thermodyn. 35, 239–250 (2003)
Peralta, R.D., Infante, R., Cortez, G., Ramos, L.F., Wisniak, J.: Excess molar volumes of tetrahydrofuran with ethyl acrylate, butyl acrylate, methyl methacrylate, and styrene at 298.15 K. Phys. Chem. Liq. 41, 361–369 (2003)
MacKnight, W.J., McKenna, L.W., Read, B.E., Stein, R.S.: Properties of ethylene–methacrylic acid copolymers. J. Phys. Chem. 72, 1122–1126 (1968)
Resa, J.M., Iglesias, M., González, C., Lanz, J., Mtz. de Ilarduya, J.A.: Excess volumes of binary mixtures of vinyl acetate and aromatic hydrocarbons. J. Chem. Thermodyn. 33, 723–732 (2001)
Peralta, R.D., Infante, R., Cortez, G., Cadenas, G., Wisniak, J.: Densities, excess volumes, and partial molar volumes of m-xylene + ethyl acrylate, + butyl acrylate, + methyl methacrylate, and + styrene at 298.15 K. Int. J. Thermophys. 24, 1061–1071 (2003)
Peralta, R.D., Infante, R., Cortez, G., Angulo, J.L., Wisniak, J.: Volumetric properties of ethylbenzene with ethyl acrylate, butyl acrylate, methyl methacrylate, and styrene at 298.15 K. Phys. Chem. Liq. 40, 649–660 (2003)
Gong, H., Chen, W., Chou, Y., Chen, M., Zheng, G.: Excess volumes of the mixing of benzene and toluene with some polar solvents at 293.15 K. Wuli Huaxue Xuebao 1, 293–298 (1985)
George, J., Sastry, N.V., Prasad, D.H.L.: Excess molar enthalpies and excess molar volumes of methyl methacrylate + benzene, + toluene, + p-xylene, + cyclohexane and + aliphatic diethers (diethyl, diisopropyl and dibutyl). Fluid Phase Equilib. 214, 39–51 (2003)
Wisniak, J., Sandoval, L.E., Peralta, R.D., Infante, R., Cortes, G., Elizalde, L.E., Soto, H.: Density and volumes of mixing of the ternary system ethylbenzene + styrene + ethyl acrylate and its binaries at 298.15 K. J. Solution Chem. 36, 135–152 (2007)
Van Ness, H.C., Abbott, M.M.: Classical Thermodynamics of Nonelectrolyte Solutions. McGraw-Hill, New York (1982)
Glasstone, S.: Textbook of Physical Chemistry. Van Nostrand, New York (1946)
Redlich, O., Kister, A.T.: Thermodynamics of nonelectrolytic solutions. Algebraic representation of thermodynamic properties and the classification of solutions. Ind. Eng. Chem. 40, 345–348 (1948)
Tomiska, J.: Zur Konversion der Anpassungen Thermodynamischer Funktionen Mittels einer Reihe Legendre’scher Polynome und der Potenzreihe. CALPHAD 5, 93–102 (1981)
Tomiska, J.: Mathematical conversions of the thermodynamic excess functions represented by the Redlich–Kister expansion, and by the Chebyshev polynomial series to power series representations and vice-versa. CALPHAD 8, 283–294 (1984)
Wisniak, J., Polishuk, A.: Analysis of residues—A useful tool for phase equilibrium data analysis. Fluid Phase Equilib. 164, 61–82 (1999)
Shacham, M., Wisniak, J., Brauer, N.: Error analysis of linearization methods in regression of data for the van Laar and Margules equations. Ind. Eng. Chem. Res. 32, 2820–2825 (1993)
TRC Thermodynamic Tables—Hydrocarbons. Thermodynamics Research Center, The Texas A&M University System, College Station, Texas, extant 2004; Table db-3220-0 (October 31, 2000)
DIPPR 801 Database, Properties for Industrial Process Design. Design Institute for Physical Properties (DIPPR), American Institute of Chemical Engineers, New York, extant 2006
Yaws, C.: Chemical Properties Handbook. McGraw-Hill, New York (1999)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wisniak, J., Cortez, G., Peralta, R.D. et al. Some Thermodynamic Properties of the Binary Systems of Toluene with Butyl Methacrylate, Allyl Methacrylate, Methacrylic Acid and Vinyl Acetate at 20, 30 and 40 °C. J Solution Chem 36, 997–1022 (2007). https://doi.org/10.1007/s10953-007-9165-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-007-9165-y