Abstract
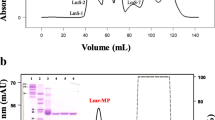
A procoagulant metalloproteinase called CCSV-MPase was purified from C. cerastes venom by successive chromatographic methods starting with gel-filtration through Sephadex G-75; ion-exchange DEAE-Cellulose A-50; affinity chromatography on Benzamidine Sepharose 6B and RP-HPLC on a C8 column. CCSV-MPase has been isolated to an extent of about tenfolds and its molecular mass was evaluated at 70 kDa by SDS–PAGE. CCSV-MPase hydrolyzes casein and fibrinogene as natural substrates. Its proteolytic activity was inhibited by EDTA and 1.10-phenanthroline, a chelators of bivalent cation metals and Zn2+ respectively. CCSV-MPase is therefore a Zn2+-metalloproteinase with fibrinogenolytic but not hemorrhagic activity. It greatly decreased levels of plasmatic fibrinogen when administered to rats for 24 h. This fibrinogenase hydrolyzes the Bβ chain of human fibrinogen in vitro releasing fibrinopeptide B only. LC MS/MS analysis of tryptic fragments of CCSV-MPase demonstrated that it showed some sequence similarities with four other venom metalloproteinases. CCSV-MPase could be considered as a potential therapeutic agent as it is a non-hemorrhagic enzyme and could be useful in thrombotic diseases because of its defibrinogenation of blood.






Similar content being viewed by others
Abbreviations
- ACN:
-
Acetonitrile
- BAEE:
-
Benzoyl-l-arginine ethyl ester hydrochloride
- CCSV-MPase:
-
Cerastes cerastes snake venom metalloproteinase
- CID:
-
Collision ion dissociation
- DEAE:
-
Di ethyl amino ethylene
- EDTA:
-
Ethylene diamine-tetra-acetic acid
- HPLC:
-
High performance liquid chromatography
- PMSF:
-
Phenyl methyl sulfonyl fluoride
- IAM:
-
2-Iodoacetamide
- MHD:
-
Minimal hemorrhage diameter
- TCEP:
-
Tris (2-carboxyethyl)phosphine hydrochloride
- TFA:
-
Tri fluorophosphate acid
References
Abib H, Laraba-Djebari F (2003) Can J Physiol Pharmacol 28:1141–1147
Bello CA, Hermogenes AL, Magalhaes A, Veiga SS, Gremski LH, Richardson M, Sanchez EF (2006) Biochimie 88:189–200
Bjarnason JB, Fox JW (1995) In Barett AJ (éd) Methods in enzymology, vol 248. San Diego, pp 345–368
Bieber AL (1979) In: Lee CY (ed) Snake venoms. Springer, Berlin, pp 295–304
Braud S, Bon C, Wisner C (2000) Biochimie 82:851–859
Castro HC, Zingali RB, Albuquerque MG, Pujol-Luz M, Rodrigues CR (2004) Cell Mol Life Sci 61:843–856
Castro HC, Rodriguez C (2006) Toxin Rev 25:291–381
Chérifi F, Laraba-Djebari F (2007) Toxines émergentes : nouveaux risques, Librairie Lavoisier 372–373
Chérifi F, Laraba-Djebari F (2008) Toxines et fonctions cholinergique neuronales et non neuronales, Edition la SFET, pp 153–154
El-Asmar MF, Shaban E, Hagag M, Tu A (1986) Toxicon 24:1037–1044
Estevao-Costa MI, Martins MS, Sanchez EF, Diniz CR, Chavez-Olortegui C (2000) Toxicon 38:139–144
Freitas MA, Geno PW, Summer LW, Cooke ME, Hudenberg SA, Ownby CL, Kaiser II, Odell GU (1992) Toxicon 30:461–464
Hahn BS, Chang IM, Kim YS (1995) Toxicon 33:929–994
Hutton RA, Warrel DA (1993) Blood Rev 7:840–855
Kamiguti AS (2005) Toxicon 45:1041–1049
Kamiguti AS, Hay CRM, Zuzul M (1996) Biochem J 320:635–641
Kondo H, Kondo S, Ikeezawa H, Morato R, Ohsaka A (1960) Jpn J Med Sci Biol 25:202–205
Kornalik F (1990) In: Shier WT, Mebs D (éd) Handbook of toxicology. New York, pp 697–709
Laemmli UK (1970) Nature 227:680–685
Laraba-Djebari F, Martin-Eauclaire MF (1990) Toxicon 28:637–646
Laraba-Djebari F, Martin-Eauclaire MF, Marchot PA (1992) Toxicon 30:1399–1410
Laraba-Djebari F, Martin-Eauclaire MF, Mauco G, Matchot P (1995) Eur J Biochem 233:756–765
Leonardi A, Fox JW, Trampus-Bakija A, Krizaj I (2007) Toxicon 49:833–842
Liu S, Sun MZ, Greenway FT (2006) Biochem Biophys Res Commun 384:1279–1287
Marrakchi N, Barbouche R, Guermazi S, Karoui H, Bon C, El-Ayeb M (1997) Eur J Biochem 247:121–128
Marrakchi N, Barbouche R, Guermazi S, Bon C, El-Ayeb M (1997) Toxicon 35:261–272
Mion G, Olive F, Hermandez E, Martin YN, Viellefosse S, Goyffon M (2002) Bull Soc Path Exot 95:132–138
Mukherjee AK, Ghossal SK, Maity CR (2000) Toxicon 38:163–175
Nishida S, Fujita T, Kohno N, Atoda H, Morita T, Takeya H, Kido I, Paine MJ, Kawabata S, Iwanaga S (1995) Biochemistry 34:1771–1778
Nishikata MJ (1984) Biochem 95:53–61
Oussedik-Oumehdi H, Laraba-Djebari F (2008) Immunopharmacol Immunotoxicol 30:37–52
Retzios AD, Markland FS (1994) Thromb Res 74:355–367
Rivière G, Bon C (1999) Ann Inst Pasteur de Paris 10:173–182
Rodrigues VM, Soares AM, Guerra-Sa R, Rodrigues V, Fontes MR, Giglio JR (2000) Arch Biochem Biophys 381:213–224
Rodrigues VM, Soares AM, Andriao-Escarso SH, Franceschi AM, Rucavado A, Gutierrez JM, Giglio JR (2001) Biochimie 83:471–479
Siigur E, Samel M, Tonismagi K, Subbi J, Siigur J, Tu AT (1998) Thromb Res 90:39–49
Siigur E, Aaspollu A, Trummal K, Tonismagi K, Tammiste I, Kalkkinen N, Siigur J (2004) Biochim Biophys Acta 1702:41–51
Swenson S, Markland FS (2005) Toxicon 45:1021–1039
Takeya H, Nishida S, Miyata T, Kawada S, Saisaka Y, Morita T, Iwanaga S (1992) J Biol Chem 267:14109–14177
Tu TA, Baker B, Wongvibulsin S, Willis T (1996) Toxicon 34:1295–1300
Van Der Walt SJ, Joubert FJ (1971) Toxicon 9:153–161
Zouari-Kessentini R, Luis-José L, Karry A, Kallech-Ziri O, Srairi-Abid N, Bazaa A, Loret E, Bezzine S, El-Ayeb M, Marrakchi N (2009) Toxicon 53:444–453
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chérifi, F., Rousselle, JC., Namane, A. et al. CCSV-MPase, a Novel Procoagulant Metalloproteinase from Cerastes cerastes Venom: Purification, Biochemical Characterization and Protein Identification. Protein J 29, 466–474 (2010). https://doi.org/10.1007/s10930-010-9273-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-010-9273-1