Abstract
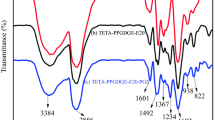
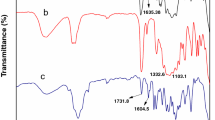
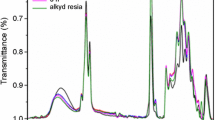
Due to the complex heterogeneous film forming process of two-component waterborne polyurethane (2K-WPU), the crosslinking reaction rate of 2K-WPU cannot meet the demand of efficient application in coatings. In order to improve the crosslinking reaction rate of 2K-WPU, a waterborne polyol containing tertiary amine groups was synthesized from rosin based epoxy resin and secondary amine compound, and then autocatalytic 2K-WPU was prepared by crosslinking the rosin based waterborne amino polyol with polyisocyanate. The structure of the polyol from rosin based epoxy resin was characterized with Fourier infrared (FT-IR) and nuclear magnetic resonance (NMR). The crosslinking kinetics and the crosslinked product of the rosin based waterborne amino polyol were also compared with a commercial acrylic polyol. It was shown from the results that the crosslinking reaction rate of the rosin based waterborne amino polyol was faster than that of the commercial acrylic polyol, which indicated the tertiary amine groups chemically bonded in the rosin based polyols could autocatalyze the crosslinking reaction of 2K-WPUs with catalysts free. The film of the rosin based waterborne amino polyol had excellent impact strength, adhesion, flexibility, hardness, gloss, fullness and solvent resistance, showing a good application prospect in the field of waterborne coatings.











Similar content being viewed by others
References
Chattopadhyay DK, Raju KVSN (2007) Structural engineering of polyurethane coatings for high performance applications. Prog Polym Sci 32:352–418
Montano V, Vogel W, Smits A, Zwaag S, García SJ (2021) From scratch closure to electrolyte barrier restoration in self- healing polyurethane coatings. ACS Appl Polym Mater 3:2802–2812
Huybrechts J, Bruylants P, Vaes A, Marre AD (2000) Surfactant-free emulsions for waterborne, two-component polyurethane coatings. Prog Org Coat 38:67–77
Melchiors M, Sonntag M, Kobusch C, Jurgents E (2000) Recent developments in aqueous two-component polyurethane (2K-PUR) coatings. Prog Org Coat 40:99–109
Wicks Z, Wicks D, Rosthauser J (2002) Two package waterborne urethane systems. Prog Org Coat 44:161–183
Rink HP, Mayer B (1998) Water-based coatings for automotive refinishing. Prog Org Coat 34:175–180
Dvorchak MJ (1997) Using “High performance two-component waterborne polyurethane” wood coatings. J Coat Technol 69:47–52
Barletta M, Vesco S, Tagliaferri V, Trovalusci F (2014) Experimental evaluation of plowing and scratch hardness of aqueous two-component polyurethane (2K-PUR) coatings on glass and polycarbonate. Prog Org Coat 77:636–645
Otts DB, Pereira KJ, Jarret WL, Urban MW (2005) Dynamic colloidal processes in waterborne two-component polyurethanes and their effects on solution and film morphology. Polymer 46:4776–4788
Rosthauser JW, Nefzger H, Cline RL, Erhart, GC (2000) Delayed action catalysts for carpet backing and air frothed foam: US, 6140381 [P].2000-10-31
Blank WJ, He ZA, Hessell ET (1999) Catalysis of the isocyanate-hydroxyl reaction by non-tin catalysts. Prog Org Coat 35:19–29
Arenivar JD (1989) Bismuth carboxylates for polyurethane catalysis. In: Proceedings of the SPI 32 ND annual technical/marketing conference, pp 623–627
Sardon H, Irusta L, Fernandez-Berridi MJ (2009) Synthesis of isophorone diisocyanate (IPDI) based waterborne polyurethanes: comparison between zirconium and tin catalysts in the polymerization process. Prog Org Coat 66:291–295
Bachmann M, Ktelhn A, Winter B, Meys R, Muller LJ, Bardow A (2021) Renewable carbon feedstock for polymers—environmental benefits from synergistic use of biomass and CO2. Faraday Discuss 230:227–246
Malani RS, Malshe VC, Thorat BN (2021) Polyols and polyurethanes from renewable sources: past, present and future-part 1: vegetable oils and lignocellulosic biomass. J Coat Technol Res. https://doi.org/10.1007/s11998-021-00490-0
Rosu L, Varganici CD, Mustata F, Rusu T, Rosu D, Rosca I, Tudorachi N, Teaca CA (2018) Enhancing the thermal and fungal resistance of wood treated with natural and synthetic derived epoxy resins. ACS Sustain Chem Eng 6:5470–5478
Kugler S, Ossowicz P, Malarczyk-Matusiak K, Wierzbicka E (2019) Advances in rosin-based chemicals: the latest recipes, applications and future trends. Molecules 24:1651
Wilbon PA, Chu FX, Tang CB (2013) Progress in renewable polymers from natural terpenes, terpenoids, and rosin. Macromol Rapid Commun 34:8–37
Li R, Zhang P, Liu T, Muhunthan B, Xin JN, Zhang JW (2018) Use of hempseed-oil-derived polyacid and rosin-derived anhydride acid as co-curing agents for epoxy materials. ACS Sustain Chem Eng 6:4016–4025
Ng F, Couture G, Philippe C, Boutevin B, Caillol S (2017) Bio-based aromatic epoxy monomers for thermoset materials. Molecules 22:149
Liu XQ, Zhang JW (2010) High-performance biobased epoxy derived from rosin. Polym Int 59:607–609
Mustata F, Rosu D, Varganici CD, Rosu L, Rosca I, Tudorachi N (2022) Assessing the thermal and fungal behavior of eco-friendly epoxy thermosets derived from vegetable oils for wood protective coatings. Prog Org Coat 163:106612
Penczek P, Matynia T (1974) Glycidyl esters of maleopimaric acid as cycloaliphatic epoxy resins. Polimery 19:609–612
Wang HH, Liu B, Liu XQ, Zhang JW, Xian M (2008) Synthesis of biobased epoxy and curing agents using rosin and the study of cure reactions. Green Chem 10:1190–1196
Li C, Liu XQ, Zhu J, Zhang CZ, Guo JS (2013) Synthesis, characterization of a rosin-based epoxy monomer and its comparison with a petroleum-based counterpart. J Macromol Sci A 50:321–329
Mantzaridis C, Brocas AL, Llevot A, Cendejas G, Auvergne R, Caillol S, Carlotti S, Cramail H (2013) Rosin acid oligomers as precursors of DGEBA-free epoxy resins. Green Chem 15:3091–3098
He L, Cui SQ, Shang SB, Wang D, Song J (2013) Properties of rosin-based waterborne polyurethanes/cellulose nanocrystals composites. Carbohyd Polym 96:510–515
Yu CL, Yan CF, Shao JT, Zhang FA (2021) Preparation and properties of rosin-based cationic waterborne polyurethane dispersion. Colloid Polym Sci 299:1489–1498
Liu XQ, Huang W, Jiang YH, Zhu J, Zhang CZ (2012) Preparation of a bio-based epoxy with comparable properties to those of petroleum-based counterparts. eXPRESS Polym Lett 6:293–298
Khairiah Binti HB, Wong CS, Maisara Shahrom BRS, Liow CH, Norhafiza YB, Nor Rabb’iatul AN (2010) FT-IR spectroscopy analysis of the prepolymerization of palm-based polyurethane. ECS J Solid State SC 18:1–8
El-Ghazawy RA, El-Saeed AM, Al-Shafey HI, Abdul-Raheim ARM, El-Sockary MA (2015) Rosin based epoxy coating: synthesis, identification and characterization. Eur Polym J 69:403–415
Wu GM, Bian JN, Liu GF, Chen J, Huo SP, Jin C, Kong ZW (2020) Self-catalytic two-component waterborne polyurethanes with amino polyols from biomass based epoxy resin. J Polym Environ 28:713–724
Vyazovkin S (2017) Isoconversional kinetics of polymers: the decade past. Macromol Rapid Commun 38:1600615
Blaine RL, Kissinger HE (2012) Homer Kissinger and the Kissinger equation. Thermochim Acta 540:1–6
Mishra RK, Mohanty K (2017) Pyrolysis kinetics and thermal behavior of waste sawdust biomass using thermogravimetric analysis. Bioresour Technol 251:63–74
Zhou D, Grant DJW (2004) Model dependence of the activation energy derived from nonisothermal kinetic data. J Phys Chem A 108:4239–4246
Silva AL, Bordado JC (2004) Recent developments in polyurethane catalysis: catalytic mechanisms review. Catal Rev 46:31–51
Lucio B, Luis D (2017) Structural and thermal degradation properties of novel metallocene-polyurethanes. Polym Degrad Stabil 136:39–47
Kiepek E, Zhou YH, Hoz S, Rozental E, Kazmaier PM, Buncel E (2005) Dissociation of the phenanthroimidazole dimer-A highly delocalized radical-Gomberg revisited. Can J Chem 83:1448–1459
Li SF, Zou T, Liu XL, Tao M (2014) Synthesis and characterization of benzoxazine monomers from rosin and their thermal polymerization. Des Monom Polym 17:40–46
Wang H, Liu HC, Cao ZX, Li WH, Huang X, Zhu Y, Ling FW, Xu H, Wu Q, Peng Y, Yang B, Zhang R, Kessler O, Huang GS, Wu JR (2020) Room-temperature autonomous self-healing glassy polymers with hyperbranched structure. Proc Natl Acad Sci USA 117:202000001
Qi Z, Ba L, Tan X, Tu M, Cheng J, Zhang J (2016) Tunable shape memory properties of rigid–flexible epoxy networks. J Mater Sci 51:10596–10607
Garcia-Bernabe A, Navarro-Gorris A (2012) Broadband dielectric spectrocopy studies of PAMAM-OH dendrimer. In: 6th International conference on times of polymers (top) and composites, vol 1459, pp 202–204
Mannari VM, Massingill L (2006) Two-component high-solid polyurethane coating systems based on soy polyols. Jct Res 3:151–157
Acknowledgements
The authors gratefully acknowledge the financial support from the Natural Science Foundation of Jiangsu Province (BK20191134).
Funding
Funding was provided by the Natural Science Foundation of Jiangsu Province (Grant No.: BK20191134).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, G., Chen, J., Yang, Z. et al. Preparation and Properties of Autocatalytic Biobased Waterborne Polyol from Rosin Based Epoxy Resin. J Polym Environ 30, 3340–3350 (2022). https://doi.org/10.1007/s10924-022-02433-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-022-02433-1