Abstract
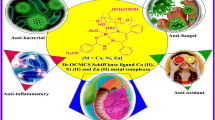
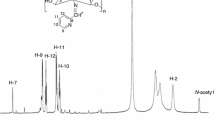
Six new N,N,O tridentate water soluble hydrazide based O-carboxymethyl chitosan Schiff base derivatives have been prepared through in-situ synthesis. The degree of substitution of schiff base derivatives was calculated theoretically and the derivatives were prepared with 1:1 proportion. Biopolymer Schiff base derivatives with their various bonding sites and different proton-carbon environment were characterised by FT-IR and 1H, 13C NMR. The crystallinity of synthesized Schiff bases was studied by X-ray diffraction. Thermal decomposition of all synthesized O-carboxymethyl chitosan Schiff base ligands was investigated by thermo gravimetric analysis which revealed the presence of Schiff base functionality to elongate the temperature to > 300 ºC. Scanning electron microscopic studies revealed that O-carboxymethyl chitosan Schiff base derivatives were affecting the crystallinity of chitosan. Additionally, the synthesized novel Schiff base derivatives were characterized for anti-inflammatory, total antioxidant and antidiabetic studies and they showed better biological and pharmaceutical results with standard drug.
Graphic Abstract











Similar content being viewed by others
References
K. Mohan, S. Ravichandran, T. Muralisankar, V. Uthayakumar, R. Chandirasekar, C. Rajeevgandhi, P. Seedevi, Int. J. Biol. Macromol. (2018). https://doi.org/10.1016/j.ijbiomac.2018.12.241
K. Zhu, S. Shi, Y. Cao, A. Lu, J. Hu, L. Zhang, Carbohydr. Polym. (2019). https://doi.org/10.1016/j.carbpol.2019.02.054
Y. Li, C. Cao, Y. Pei, X. Liu, K. Tang, Int. J. Biol. Macromol. 130, 715–719 (2019)
L. Panariello, M.-B. Coltelli, M. Buchignani, A. Lazzeri, Eur. Polym. J. (2019). https://doi.org/10.1016/j.eurpolymj.2019.02.004
S. Sahraee, J.M. Milani, B. Ghanbarzadeh, H. Hamishehkar, Int. J. Biol. Macromol. 97, 373–381 (2017)
Q. Yao, C. Wu, P. Luo, X. Xiang, J. Liu, L. Mou, J. Bao, Process Biochem. 45(9), 1477–1485 (2010)
A. Singh, P.K. Dutta, H. Kumar, A.K. Kureel, A.K. Rai, Carbohydr. Polym. 193, 99–107 (2018)
A.M. Abdel-Mohsen, J. Jancar, D. Massoud, Z. Fohlerova, H. Elhadidy, Z. Spotz, A. Hebeish, Int. J. Pharm. 510(1), 86–99 (2016)
U. Nwankwo, R. Bucher, A.B.C. Ekwealor, M. Maaza, F.I. Ezema, Vacuum (2018). https://doi.org/10.1016/j.vacuum.2018.12.011
H. Mittal, S. Sinha Ray, B. Singh Kaith, J. Kaur Bhatia, Sukriti, J. Sharma, S.M. Alhassan, Eur. Polym. J. (2018). https://doi.org/10.1016/j.eurpolymj.2018.10.013
J.E. Dos Santos, E.R. Dockal, É.T.G. Cavalheiro, Carbohydr. Polym. 60(3), 277–282 (2005)
K. Muraleedharan, C.H. Viswalekshmi, K. Sarada, Polym. Bull. 74(1), 39–54 (2016)
F.A. Al-Sagheer, E.I. Ibrahim, K.D. Khalil, Eur. Polym. J. 58, 164–172 (2014)
L. Higueras, G. Lopez-Carballo, R. Gavara, P. Hernández-Munoz, Food Bioprocess Technol. 8(3), 526–538 (2014)
A.J. Al-Manhel, A.R.S. Al-Hilphy, A.K. Niamah, J. Saudi Soc. Agric. Sci. 17(2), 186–190 (2018)
N. Mati-Baouche, P.-H. Elchinger, H. de Baynast, G. Pierre, C. Delattre, P. Michaud, Eur. Polym. J. 60, 198–212 (2014)
I. Aranaz, N. Acosta, C. Civera, B. Elorza, J. Mingo, C. Castro, A. Heras Caballero, Polymers 10(2), 213 (2018)
M. Lucic Skoric, I. Terzic, N. Milosavljevic, M. Radetic, Z. Saponjic, M. Radoicic, M. Kalagasidis Krusic, Eur. Polym. J. 82, 57–70 (2016)
T.D.A. Senra, S.P. Campana-Filho, J. Desbrieres, Eur. Polym. J. 104, 128–135 (2018)
J. Zhao, Z. Zou, R. Ren, X. Sui, Z. Mao, H. Xu, B. Wang, Eur. Polym. J. 108, 212–218 (2018)
H. Bidgoli, A. Zamani, M.J. Taherzadeh, Carbohyd. Res. 345(18), 2683–2689 (2010)
A.L. Bukzem, R. Signini, D.M. dos Santos, L.M. Lião, D.P.R. Ascheri, Int. J. Biol. Macromol. 85, 615–624 (2016)
Y. Li, Y. Tan, K. Xu, C. Lu, X. Liang, P. Wang, RSC Adv. 5(38), 30303–30309 (2015)
B. Fonseca-Santos, M. Chorilli, Mater. Sci. Eng. C 77, 1349–1362 (2017)
L. Upadhyaya, J. Singh, V. Agarwal, R.P. Tewari, J. Control. Release 186, 54–87 (2014)
R. Jayakumar, M. Prabaharan, S.V. Nair, S. Tokura, H. Tamura, N. Selvamurugan, Prog. Mater Sci. 55(7), 675–709 (2010)
B.J. Gangani, P.H. Parsania, Spectrosc. Lett. 40(1), 97–112 (2007)
F. Bar, H. Hopf, M. Knorr, O. Schroder, J. Krahl, Fuel 180, 278–283 (2016)
P. Singh, D.P. Singh, K. Tiwari, M. Mishra, A.K. Singh, V.P. Singh, RSC Adv. 5(56), 45217–45230 (2015)
V. Arumugam, W. Kaminsky, N.S.P. Bhuvanesh, D. Nallasamy, RSC Adv. 5(73), 59428–59436 (2015)
F. Rahim, H. Ullah, M. Taha, A. Wadood, M.T. Javed, W. Rehman, K.M. Khan, Bioorg. Chem. 68, 30–40 (2016)
A.M. Ajlouni, Q. Abu-Salem, Z.A. Taha, A.K. Hijazi, W. Al Momani, J. Rare Earths 34(10), 986–993 (2016)
K. Tiwari, M. Mishra, V.P. Singh, RSC Adv. 3(30), 12124 (2013)
H. Zhu, J. Fan, B. Wang, X. Peng, Chem. Soc. Rev. 44(13), 4337–4366 (2015)
T. Anand, A.S.K. Kumar, S.K. Sahoo, Photochem. Photobiol. Sci. 17(4), 414–422 (2018)
S. Yadav, I. Yousuf, M. Usman, M. Ahmad, F. Arjmand, S. Tabassum, RSC Adv. 5(63), 50673–50690 (2015)
A. Jamadar, A.-K. Duhme-Klair, K. Vemuri, M. Sritharan, P. Dandawate, S. Padhye, Dalton Trans. 41(30), 9192 (2012)
B. Doshi, E. Repo, J.P. Heiskanen, J.A. Sirviö, M. Sillanpää, Carbohydr. Polym. 167, 326–336 (2017)
Z. Yu, C. Xiao, Y. Huang, M. Chen, W. Wei, X. Yang, X. Fan, RSC Adv. 8(32), 17860–17877 (2018)
D. Bhattacharya, M. Das, D. Mishra, I. Banerjee, S.K. Sahu, T.K. Maiti, P. Pramanik, Nanoscale 3(4), 1653 (2011)
H. Wang, C.A. Thorling, X. Liang, K.R. Bridle, J.E. Grice, Y. Zhu et al., J. Mater. Chem. B 3(6), 939–958 (2015)
I.M. El-Sherbiny, Eur. Polym. J. 45(1), 199–210 (2009)
A. Naz, S. Arun, S.S. Narvi, M.S. Alam, A. Singh, P. Bhartiya, P.K. Dutta, Int. J. Biol. Macromol. 110, 215–226 (2018)
G. He, C. Wang, J. Cao, L. Fan, S. Zhao, Y. Chai, J. Environ. Chem. Eng. (2019). https://doi.org/10.1016/j.jece.2019.102953
H. Zeng, L. Wang, D. Zhang, P. Yan, J. Nie, V.K. Sharma, C. Wang, Chem. Eng. J. (2018). https://doi.org/10.1016/j.cej.2018.10.001
S. Rahmani, S. Hakimi, A. Esmaeily, F.Y. Samadi, E. Mortazavian, M. Nazari, M.R. Tehrani, Int. J. Pharm. (2019). https://doi.org/10.1016/j.ijpharm.2019.02.016
T. Agarwal, R. Narayan, S. Maji, S. Behera, S. Kulanthaivel, T.K. Maiti, S. Giri, Int. J. Biol. Macromol. 93, 1499–1506 (2016)
X. Zhao, L. Zhou, Q. Li, Q. Zou, C. Du, Carbohydr. Polym. 195, 225–234 (2018)
P.R. Sivashankari, M. Prabaharan, Chitosan based biomaterials (chap. 5), vol. 1 (Wiley, New York, 2017), pp. 117–133
M. Manimohan, S. Pugal Mani, M. Aboobucker Sithique, New J. Chem. 43, 9540–9554 (2019)
W. Liu, Y. Qin, S. Liu, R. Xing, H. Yu, X. Chen, P. Li, Int. J. Biol. Macromol. 114, 942–949 (2018)
H. Srinivasan, V. Kanayairam, R. Ravichandran, Int. J. Biol. Macromol. 107, 662–667 (2018)
Z. Jiang, Y. Song, J. Qiao, Y. Yang, W. Zhang, W. Liu, B. Han, Int. J. Biol. Macromol. (2019). https://doi.org/10.1016/j.ijbiomac.2019.02.080
T. Baran, A. Mentes, H. Arslan, Int. J. Biol. Macromol. 72, 94–103 (2015)
M. Manimohan, S. Pugalmani, M. Aboobucker Sithique, Int. J. Biol. Macromol. 136, 738–754 (2019)
T. Baran, A. Mentes, J. Mol. Struct. 1115, 220–227 (2016)
S. Aslkhademi, N. Noshiranzadeh, M.S. Sadjadi, K. Mehrani, N. Farhadyar, Polyhedron (2018). https://doi.org/10.1016/j.poly.2018.12.023
W. Liu, Y. Qin, S. Liu, R. Xing, H. Yu, X. Chen, P. Li, Carbohydr. Polym. 160, 97–105 (2017)
T. Baran, E. Aciksoz, A. Mentes, Carbohydr. Polym. 142, 189–198 (2016)
A.A. Alhwaige, H. Ishida, S. Qutubuddin, Carbohydr. Polym. (2019). https://doi.org/10.1016/j.carbpol.2019.01.016
R. Antony, S. Theodore David, K. Saravanan, K. Karuppasamy, S. Balakumar, Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 103, 423–430 (2013)
T. Baran, A. Mentes, Int. J. Biol. Macromol. 79, 542–554 (2015)
T. Baran, T. Inanan, A. Mentes, Carbohydr. Polym. 145, 20–29 (2016)
T. Baran, A. Mentes, J. Mol. Struct. 1134, 591–598 (2017)
R. Sribalan, M. Kirubavathi, G. Banuppriya, V. Padmini, Bioorg. Med. Chem. Lett. 25(19), 4282–4286 (2015)
P. Kasangana, P. Haddad, T. Stevanovic, Antioxidants 4(2), 410–426 (2015)
M.I. Kazeem, J.O. Adamson, I.A. Ogunwande, Biomed. Res. Int. 2013, 1–6 (2013)
Acknowledgements
The authors thankful to the PG and Research Department of Chemistry, Islamiah College (Autonomous), Vaniyambadi, Tamil Nadu, India for availing needed laboratory and the Instrument facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Manimohan, M., Pugalmani, S. & Sithique, M.A. Biologically Active Water Soluble Novel Biopolymer/Hydrazide Based O-Carboxymethyl Chitosan Schiff Bases: Synthesis and Characterisation. J Inorg Organomet Polym 30, 3658–3676 (2020). https://doi.org/10.1007/s10904-020-01487-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-020-01487-9