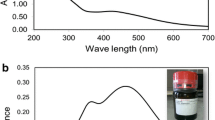
Abstract
Persuaded by the necessity of reduction of toxicity, silver nanoparticles (Ag-NPs) were synthesized from the reaction between AgNO3 solution and Mizuna (Brassica rapa var. japonica) leaf extract. This study aimed to investigate the role of synthesis temperature on particle properties. Thus, four synthesis-temperatures, 25 °C (room temperature), 60 °C, 80 °C, and 100 °C were applied. Ag-NPs were characterized using ultraviolet–visible (UV–Vis) spectrophotometer, energy dispersive X-ray spectrometer, X-ray diffractometer, transmission electron microscope, atomic absorption spectrometry and dynamic light scattering techniques. The high-temperature (80 and 100 °C) synthesis showed higher stability, demostrated lowest release of silver ion (0.02 µg/mL, 0.002 µg/mL) compared to low-temperature synthesis. In addition, high-temperature synthesis showed higher C-content (> 50%) with lower Ag-content (< 30%) than low-temperature (25 and 60 °C) synthesis. Moreover, a complete surface encapsulation is found on the particles synthesised at 100 °C by TEM analysis. The synthesized Ag-NPs were exposed to Caco-2 cell lines for cytotoxicity assessment. The high-temperature synthesis Ag-NPs exerted significantly lower cytotoxicity in terms of Caco-2 cell viability and lactate dehydrogenase (LDH) activity assay. Upon exposure to 1, 5 and 10 µg/mL Ag-NPs for 48 h the cell viability (96.33%, 90.66% and 89.66%) was not changed for high-temperature synthesis particles as well as the LDH activity was unchanged. However, low-temperature synthesis showed significant cytotoxicity (81%, 66% and 44.33%) under similar experimental conditions. Thus, characterization and cytotoxicity techniques clearly demonstrate the successful synthesis of Ag-NPs with the encapsulation of bio-molecules that enhances the particle stability and reduce cytotoxicity.








Similar content being viewed by others
References
S.J. Yu, Y.G. Yin, J.F. Liu, Environ. Sci. Process. Impacts 15, 78–92 (2013)
M. Akter, M.T. Sikder, M.M. Rahman, A.K.M.A. Ullah, K.F.B. Hossain, S. Banik, T. Hosokawa, T. Saito, M. Kurasaki, J. Adv. Res. 9, 1–16 (2018)
K. Kneipp, H. Kneipp, J. Kneipp, Acc. Chem. Res. 39, 443–450 (2006)
M. Bruchez, M. Moronne, P. Gin, S. Weiss, A.P. Alivisatos, Science 281, 2013–2016 (1988)
X. Hangxun, W.Z. Brad, K.S. Suslick, Chem. Synth. Rev. 42, 2555–2567 (2013)
S.N. Aboutorabi, M. Nasiriboroumand, P. Mohammad, H. Sheibani, H. Barani, J. Inorg. Organomet. Polym. 28(6), 2525–2532 (2018)
M.J. Piao, K.A. Kang, I.K. Lee, H.S. Kim, S. Kim, J.Y. Choi et al., Toxicol. Lett. 201, 92–100 (2011)
H. Kang, J.T. Buchman, R.S. Rodriguez, H.L. Ring, J. He, K.C. Bantz, C.L. Haynes, Chem. Rev. 119, 664–699 (2019)
T.C. Prathna, N. Chandrasekaran, A.M. Raichur, A. Mukherjee, Colloids Surf. A 377, 212–216 (2011)
T. Yonezawa, D. Čempel, M.T. Nguyen, Bull. Chem. Soc. Jpn. 91, 1781–1798 (2018)
P. Verma, Y. Kuwahara, K. Mori, H. Yamashita, Bull. Chem. Soc. Jpn. 92, 19–29 (2019)
J. Zhou, J. An, B. Tang, S. Xu, Y. Cao, B. Zhao et al., Langmuir 24, 10407–10413 (2008)
B.J. Wiley, Y. Xiong, Z.L. Yuan, Y. Yin, Y. Xia, Nano Lett. 6, 765–768 (2006)
Y. Sun, Y. Xia, Science 298, 2176–2179 (2002)
M. Maillard, P. Huang, L. Brus, Nano Lett. 3, 1611–1615 (2003)
Y. Gao, P. Jiang, L. Song, J. Wang, L. Liu, D. Liu et al., J Cryst. Growth 289, 376–380 (2006)
J. An, B. Tang, X. Ning, J. Zhou, S. Xu, B. Zhao et al., J. Phys. Chem. 111, 18055–18059 (2007)
R. Jin, Y. Cao, C.A. Mirkin, G.C. Schatz, J. Zheng, Science 294, 1901–1903 (2001)
A. Tao, P. Sinsermsuksakul, P. Yang, Chemie 45, 4597–4601 (2006)
B. Pietrobon, M. McEachran, V. Kitaev, ACS Nano 39, 21–26 (2008)
K. Caswell, C.M. Bender, C.J. Murphy, Nano Lett. 3, 667–669 (2003)
C. Xue, G.S. Metraux, J.E. Millstone, C.A. Mirkin, J. Am. Chem. Soc. 130, 8337–8344 (2008)
S.W. Lee, S.H. Chang, Y.S. Lai, C.C. Lin, C.M. Tsai, Y.C. Lee et al., Materials 7, 7781–7798 (2014)
M. Akter, M.M. Rahman, A.K.M.A. Ullah, M.T. Sikder, T. Hosokawa, T. Saito, M. Kurasaki, J. Inorg. Organomet. Polym. 28, 1483–1493 (2018)
M.M. Rahman, R.A. Uson-Lopez, M.T. Sikder, G. Tan, T. Hosokawa, T. Saito, M. Kurasaki, Chemosphere 196, 453–466 (2018)
A.K.M.A. Ullah, M.F. Kabir, M. Akter, A.N. Tamanna, A. Hossain, A.R.M. Tareq, M.N.I. Khan, A.K.M.F. Kibria, M. Kurasaki, M.M. Rahman, RSC Adv. 8, 37176–37183 (2018)
J.M. Ashraf, M.A. Ansari, H.M. Khan, A.M.A. Alzohairy, I. Choi, Sci. Rep. 6, 20414 (2016)
D. Sarvamangala, K. Kondala, N. Sivakumar, M.S. Babu, S. Manga, Int. Res. J. Pharm. 4, 240–243 (2013)
R. Parameshwaran, S. Kalaiselvam, R. Jayavel, Mater. Chem. Phys. 140, 135–147 (2013)
M. Behravan, A.H. Panahi, A. Naghizadeh, M. Ziaee, R. Mahdavi, A. Mirzapour, Int. J. Biol. Macromol. 124, 148–154 (2019). https://doi.org/10.1016/j.ijbiomac.2018.11.101
M.M.I. Haytham, J. Radiat. Res. Appl. Sci. 8, 265–275 (2015)
J. Park, J. Joo, S.G. Kwon, Y. Jang, T. Hyeon, Angew. Chem. Int. Ed. 46, 4630–4660 (2007)
A.K.M.A. Ullah, A.K.M.F. Kibria, M. Akter, M.N.I. Khan, M.A. Maksud, R.A. Jahan, S.H. Firoz, J. Saudi Chem. Soc. 21, 830–836 (2017)
A.K.M.A. Ullah, A.K.M.F. Kibria, M. Akter, M.N.I. Khan, A.R.M. Tareq, S.H. Firoz, Water Conserv. Sci. Eng. 1, 249–256 (2017)
R.M. Gangan, K. Anand, A. Phulukdaree, A. Chuturgoon, Colloids Surf. B 105, 87–91 (2013)
X.C. Jiang, W.M. Chen, C.Y. Chen, S.X. Xiong, A.B. Yu, Nanoscale Res. Lett. 6(1), 32 (2011)
K. Ali, B. Ahmed, S. Dwivedi, Q. Saquib, A.A. Al-Khedhairy, J. Musarrat, PLoS ONE (2015). https://doi.org/10.1371/journal.pone.0131178
J. Mytych, J. Zebrowski, A. Lewinska, M. Wnuk, Mol. Neurobiol. 54, 1–16 (2016)
G. Kapildeva, M. Manickavasagama, N. Thajuddinb, K. Premkumarc, A. Ganapathi, Colloid Surf. B 106, 86–92 (2013)
P. Velmurugan, S. Hong, A. Aravinthan, S.H. Jang, P.I. Yi, Y.C. Song, E.S. Jung, J.S. Park, S. Sivakumar, Arab. J. Sci. Eng. (2016). https://doi.org/10.1007/s13369-016-2254-8
K. Kalishwaralal, E. Banumathi, S.R.K. Pandian, V. Deepak, J. Muniyandi, S.H. Eom et al., Colloid Surf. B 73, 51–57 (2009)
J.Y. Wang, M.F. Rahman, H.M. Duhart, G.D. Newport, T.A. Patterson, R.C. Murdock et al., Expression changes of dopaminergic system-related genes in PC12 cells induced by manganese, silver, or copper nanoparticles. Neurotoxicology 30, 926–933 (2009)
M.V.D.Z. Park, A.M. Neigh, J.P. Vermeulen, L.J.J. de la Fonteyne, H.W. Verharen, J.J. Briedé et al., Biomaterials 32, 9810–9817 (2011)
C. Carlson, S.M. Hussain, A.M. Schrand, L.K. Braydich-Stolle, K.L. Hess, R.L. Jones et al., J. Phys. Chem. B 112, 13608–13619 (2008)
A.K.M.A. Ullah, A.N. Tamanna, A. Hossain, M. Akter, M.F. Kabir, A.R.M. Tareq, A.K.M. Fazle Kibria, M. Kurasaki, M.M. Rahman, M.N.I. Khanh, RSC Adv. 9, 13254–13262 (2019)
X. Yang, A.P. Gondikas, S.M. Marinakos, M. Auffan, J. Liu, H. Hsu-Kim, J.N. Meyer, Environ. Sci. Technol. 46, 1119–1127 (2012)
Acknowledgements
We are humbly showing our gratitude to Ms. Miyako Komori, Prof. Kamiya and Dr. Parvin Begum for giving instrumental support during experiments. We are also showing our appreciation to the ‘Hokkaido University Innovation Research Organization Nanotechnology Integrated Research’ for giving us experimental support with their facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akter, M., Ullah, A.K.M.A., Rahaman, M.S. et al. Stability Enhancement of Silver Nanoparticles Through Surface Encapsulation via a Facile Green Synthesis Approach and Toxicity Reduction. J Inorg Organomet Polym 30, 1956–1965 (2020). https://doi.org/10.1007/s10904-019-01373-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-019-01373-z