Abstract
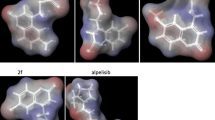
New series of Pt(II)–azaindazole complexes, were synthesized and also characterized by, analytical, spectral and computational tools. All synthesized Pt(II)-complexes, appeared as mononuclear with bi-dentate mode of bonding. Octahedral arrangement as well as square-planer, were proposed geometries around platinum atoms in all complexes. The best atomic-skeletons, were demonstrated by using Gaussian09 program. Applying MOE module (V. 2015), extensive molecular docking process was executed upon all new synthesizes. This docking study was interested in kinase protein (3ce3) and health cell–DNA (5ahr), to predict the degree of cancer-inhibition and also the mode of interaction. A significant inhibition activity, was clearly predicted with Pt(II)–4b and Pt(II)–4c complexes, against 3ce3 protein-receptors. While the absence of any significant effect towards 5ahr, which is favorable trend of therapeutic agent. Various backbone receptors (amino acids) were attacked through H-bonding, from most tested inhibitors, especially of 3ce3 protein. DNA-degradation study, which executed in vitro, displayed a complete degradation for DNA with most screened compounds except 4d and 4e derivatives. This feature points to the promising antitumor activity of most synthesizes, especially the Pt(II) complexes, as a predicted role of its complexes.








Similar content being viewed by others
References
B. Rosenberg, L. Vancamp, J.E. Trosko, V.H. Mansour, Nature 222, 385–386 (1969)
M.D. Živković, J. Kljun, T. Ilic-Tomic, A. Pavic, A. Veselinović, D.D. Manojlović, J. Nikodinovic-Runic, I. Turel, Inorg. Chem. Front. 5, 39–53 (2018)
N. Muhammad, Z. Guo, Metal-based anticancer chemotherapeutic agents. Curr. Opin. Chem. Biol. 19, 144–153 (2014)
Z.H. Siddik, Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene 22, 7265–7279 (2003)
V. Brabec, Front. Multifunct. Nanosyst. 57, 229–250 (2002)
Y. Jung, S.J. Lippard, Direct cellular responses to platinum-induced DNA damage. Chem. Rev. 107(5), 1387–1407 (2007)
L. Kelland, The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 7, 573–584 (2007)
X. Wang, Fresh platinum complexes with promising antitumor activity. Anti-Cancer Agents Med. Chem. 10, 396–411 (2010)
J. Zhang, L. Wang, Z. Xing, D. Liu, J. Sun, X. Li, Y. Zhang, Anti-Cancer Agents Med. Chem. 10, 272–282 (2010)
N.J. Wheate, S. Walker, G.E. Craig, R. Oun, Dalton Trans. 39, 8113–8127 (2010)
U.K. Bandarage, M.P. Clark, E. Perola, H. Gao, M.D. Jacobs, A. Tsai, J. Gillespie, J.M. Kennedy, F. Maltais, M.W. Ledeboer, I. Davies, W. Gu, R.A. Byrn, K.N. Addae, H. Bennett, J.R. Leeman, S.M. Jones, C. O’Brien, C. Memmott, Y. Bennani, P.S. Charifson, A.C.S. Med, Chem. Lett. 8(2), 261–265 (2017)
W.H. Mahmoud, F.N. Sayed, G.G. Mohamed, Appl. Organomet. Chem. 32(6), e4347 (2018)
J. Sun, J. Jin, R.D. Beger, C.E. Cerniglia, H. Chen, J. Ind. Microbiol. Biotechnol. 44(10), 1471–1481 (2017)
K. Nejati, Z. Rezvani, B. Massoumi, Dyes Pigm. 75, 653–657 (2007)
H.Y. Lee, X. Song, H. Park, M.-H. Baik, D. Lee, J. Am. Chem. Soc. 132(34), 12133–12144 (2010)
M. Badea, R. Olar, E. Cristurean, D. Marinescu, A. Emandi, P. Budrugeac, E. Segal, J. Therm. Anal. Calor. 77(3), 815–824 (2004)
S.M. Abdallah, Arab. J. Chem. 5(2), 251–256 (2012)
U.K. Bandarage, M.P. Clark, E. Perola, H. Gao, M.D. Jacobs, A. Tsai, J. Gillespie, J.M. Kennedy, F. Maltais, M.W. Ledeboer, I. Davies, W. Gu, R.A. Byrn, K.N. Addae, H. Bennett, J.R. Leeman, S.M. Jones, C. O’Brien, C. Memmott, Y. Bennani, P.S. Charifson, A.C.S. Med, Chem. Lett. 8(2), 261–265 (2017)
A.M.K. El-Dean, A.A. Geies, T.A. Mohamed, Indian J. Chem. B 30, 878–882 (1991)
M. Lacan, K. Tabakovic, Croat. Chem. Acta 47, 127–133 (1975)
G. Kaupp, M.A. Metwally, F.A. Amer, E. Abdel-Latif, Eur. J. Org. Chem. 2003(8), 1545–1551 (2003)
J. Marmur, A procedure for the isolation of deoxyribonucleic acid from micro-organisms. J. Mol. Biol. 3, 208–218 (1961)
A. Wolfe, G.H. Shimer, T. Meehan, Biochemistry 26(20), 6392–6396 (1987)
G.A. Al-Hazmi, N.M. El-Metwally, O.A. El-Gammal, A.A. El-Asmy, Spectrochim. Acta A 69, 56–61 (2008)
A.I. Vogel, Text Book of Quantitative Inorganic Analysis (Longman, London, 1986)
M.J. Frisch et al., Gaussian 09, Revision D (Gaussian Inc., Wallingford, 2010)
R. Dennington, T. Keith, J. Millam, Gauss View, Version 4.1.2 (Semichem Inc., Shawnee Mission, 2007)
U. El-Ayaan, N.M. El-Metwally, M.M. Youssef, S.A.A. El Bialy, Spectrochim. Acta A 68, 1278–1286 (2007)
R.K. Ray, G.R. Kauffman, Inorg. Chem. Acta 173, 207–214 (1990)
K.H. Mashat, B.A. Babgi, M.A. Hussien, Synthesis, structures, DNA-binding and anticancer activities of some copper (I)-phosphine complexes. Polyhedron 158, 164–172 (2019)
F.A. Saad, J.H. Al-Fahemi, H. El-Ghamry, A.M. Khedr, M.G. Elghalban, N.M. El-Metwaly, J. Therm. Anal. Calorim. 131(2), 1249–1267 (2018)
F.A. Saad, M.G. Elghalban, N. El-Metwaly, H. El-Ghamry, A.M. Khedr, Appl. Organomet. Chem. 31, ec3721 (2017)
M.S. Refat, I.M. El-Deen, Z.M. Anwer, S. El-Ghol, J. Mol. Struct. 920, 149–162 (2009)
A.A. Abou-Hussen, N.M. El-Metwaly, E.M. Saad, A.A. El-Asmy, J. Coord. Chem. 58, 1735–1749 (2005)
W.R. Mason, H.B. Gray, J. Am. Chem. Soc. 90(21), 5721–5729 (1968)
R. Srinivasan, I. Sougandi, R. Venkatesan, P.S. Rao, J. Chem. Sci. 115, 91–102 (2003)
G.A.A. Al-Hazmia, A.A. El-Zahhar, K.S. Abou-Melha, F.A. Saad, M.H. Abdel-Rhman, A.M. Khedr, N.M. El-Metwaly, J. Coord. Chem. 68(6), 993–1009 (2015)
G.A. Al-Hazmi, K.S. Abou-Melha, N.M. El-Metwaly, K.A. Saleh, Bioorg. Chem. Appl. 2018, 22 (2018)
M.S. Refat, N.M. El-Metwaly, Spectrochim. Acta A 81, 215 (2011)
H.A. Katouah, J.H. Al-Fahemi, M.G. Elghalban, F.A. Saad, I.A. Althagafi, N.M. El-Metwaly, A.M. Khedr, Mater. Sci. Eng. C 96, 740–756 (2019)
B.D. Cullity, Elements of X-ray Diffraction, 2nd edn. (Addison-Wesley Inc., Boston, 1993)
S. Velumani, X. Mathew, P.J. Sebastian, S.K. Narayandass, D. Mangalaraj, Sol. Energy Mater. Sol. Cell 76(3), 347–358 (2003)
M. Badea, A. Emandi, D. Marinescu, E. Cristurean, R. Olar, A. Braileanu, P. Budrugeac, E. Segal, J. Therm. Anal. Calorim. 72, 525–531 (2003)
M. Gaber, A.M. Khedr, M. Elsharkawy, Appl. Organomet. Chem. 31(12), e3885 (2017)
A.Z. El-Sonbati, A.F. Shoair, A.A. El-Bindary, M.A. Diab, A.S. Mohamed, J. Mol. Liq. 209, 635–647 (2015)
L.H. Abdel-Rahman, A.M. Abu-Dief, N.M. Ismail, M. Ismael, J. Inorg. Nano-Metal Chem. 47, 467–480 (2017)
R.C. Chikate, S.B. Padhye, Polyhedron 24, 1689–1700 (2005)
S. Sagdinc, B. Köksoy, K.F. Andemirli, S.H. Bayari, J. Mol. Struct. 917, 63–70 (2009)
I. Fleming, Frontier Orbital’s and Organic Chemical Reactions (Wiley, London, 1976)
S.K. Tripathi, R. Muttineni, S.K. Singh, J. Theor. Biol. 334, 87–100 (2013)
S. Gross, R. Rahal, N. Stransky, C. Lengauer, K.P. Hoeflich, J. Clin. Invest. 125(5), 1780–1789 (2015)
A.A. Fadda, E. Abdel-Latif, A. Fekri, A.R. Mostafa, J. Heterocycl. Chem. (2019). https://doi.org/10.1002/jhet.3452(2019)
S.S.A. El-Karima, M.M. Anwara, Y.M. Syama, M.A. Naelb, H.F. Alia, M.A. Motaleb, Bioorg. Chem. 81, 481–493 (2018)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
El-Metwaly, N., Al-Fahemi, J.H., Althagafi, I. et al. Docking Approach to Predict Inhibition Activity of New Pt(II) Complexes Against Kinase Protein and Human DNA: Full Characterization, HF-FC Modeling and Genotoxicity. J Inorg Organomet Polym 30, 907–922 (2020). https://doi.org/10.1007/s10904-019-01233-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-019-01233-w