Abstract
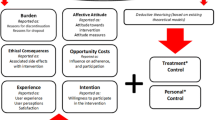
The purpose of this study was to develop a brief instrument, the Feelings About genomiC Testing Results (FACToR), to measure the psychosocial impact of returning genomic findings to patients in research and clinical practice. To create the FACToR, we modified and augmented the Multidimensional Impact of Cancer Risk Assessment (MICRA) questionnaire based on findings from a literature review, two focus groups (N = 12), and cognitive interviews (N = 6). We evaluated data from 122 participants referred for evaluation for inherited colorectal cancer or polyposis from the New EXome Technology in (NEXT) Medicine Study, an RCT of exome sequencing versus usual care. We assessed floor and ceiling effects of each item, conducted principal component analysis to identify subscales, and evaluated each subscale’s internal consistency, test-retest reliability, and construct validity. After excluding items that were ambiguous or demonstrated floor or ceiling effects, 12 items forming four distinct subscales were retained for further analysis: negative emotions, positive feelings, uncertainty, and privacy concerns. All four showed good internal consistency (0.66–0.78) and test-retest reliability (0.65–0.91). The positive feelings and the uncertainty subscales demonstrated known-group validity. The 12-item FACToR with four subscales shows promising psychometric properties on preliminary evaluation in a limited sample and needs to be evaluated in other populations.
Similar content being viewed by others
References
Apse, K. A., Biesecker, B. B., Giardiello, F. M., Fuller, B. P., & Bernhardt, B. A. (2004). Perceptions of genetic discrimination among at-risk relatives of colorectal cancer patients. Genetics in Medicine, 6(6), 510–516. https://doi.org/10.1097/01.GIM.0000144013.96456.6C.
Berwick, D., Murphy, J., & Goldman, P. (1991). Performance of a five-item mental health screening test. Medical Care, 29(2), 169–176.
Biesecker, B. B., Woolford, S. W., Klein, W. M. P., Brothers, K. B., Umstead, K. L., Lewis, K. L. et al (2017). PUGS: A novel scale to assess perceptions of uncertainties in genome sequencing. Clinical Genetics, 1–8. https://doi.org/10.1111/cge.12949.
Bombard, Y., Veenstra, G., Friedman, J. M., Creighton, S., Currie, L., Paulsen, J. S., et al. (2009). Perceptions of genetic discrimination among people at risk for Huntington’s disease: A cross sectional survey. BMJ, 338. Retrieved from http://www.bmj.com/content/338/bmj.b2175.abstract
Broadstock, M., Michie, S., & Marteau, T. (2000). Psychological consequences of predictive genetic testing: A systematic review. European Journal of Human Genetics : EJHG, 8, 731–738. https://doi.org/10.1038/sj.ejhg.5200532.
Cella, D., Hughes, C., Peterman, A., Chang, C.-H., Peshkin, B. N., Schwartz, M. D., et al. (2002). A brief assessment of concerns associated with genetic testing for cancer: The multidimensional impact of cancer risk assessment (MICRA) questionnaire. Health Psychology : Official Journal of the Division of Health Psychology, American Psychological Association, 21(6), 564–572. https://doi.org/10.1037/0278-6133.21.6.564.
Cho, A. H., Killeya-Jones, L. A., O’Daniel, J. M., Kawamoto, K., Gallagher, P., Haga, S., et al. (2012). Effect of genetic testing for risk of type 2 diabetes mellitus on health behaviors and outcomes: Study rationale, development and design. BMC Health Services Research, 12(1), 16. https://doi.org/10.1186/1472-6963-12-16.
Chung, W. W., Chen, C. A., Cupples, L. A., Roberts, J. S., Hiraki, S. C., Nair, A. K., et al. (2009). A new scale measuring psychologic impact of genetic susceptibility testing for Alzheimer disease. Alzheimer Dis Assoc Disord, 23(1), 50–56 Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/19266699.
Crozier, S., Robertson, N., & Dale, M. (2014). The psychological impact of predictive genetic testing for Huntington’s disease: A systematic review of the literature. Journal of Genetic Counseling, 24(1), 29–39. https://doi.org/10.1007/s10897-014-9755-y.
GeneTests. (2017). Retrieved July 10, 2017, from https://www.genetests.org/
Graves, K. D., Gatammah, R., Peshkin, B. N., Krieger, A., Gell, C., Valdimarsdottir, H. B., et al. (2011). BRCA1/2 genetic testing uptake and psychosocial outcomes in men. Familial Cancer, 10(2), 213–223. https://doi.org/10.1007/s10689-011-9425-2.
Graves, K. D., Vegella, P., Poggi, E. A., Peshkin, B. N., Tong, A., Isaacs, C., et al. (2012). Long-term psychosocial outcomes of BRCA1/BRCA2 testing: Differences across affected status and risk-reducing surgery choice. Cancer Epidemiology Biomarkers and Prevention, 21(3), 445–455. https://doi.org/10.1158/1055-9965.EPI-11-0991.
Graves, K. D., Leventhal, K. G., Nusbaum, R., Salehizadeh, Y., Hooker, G. W., Peshkin, B. N., et al. (2013). Behavioral and psychosocial responses to genomic testing for colorectal cancer risk. Genomics, 102(2), 123–130. https://doi.org/10.1016/j.ygeno.2013.04.002.
Gray, S. W., Martins, Y., Feuerman, L. Z., Bernhardt, B. A, Biesecker, B. B., Christensen, K. D., et al. (2014). Social and behavioral research in genomic sequencing: Approaches from the clinical sequencing exploratory research consortium outcomes and measures working group. Genetics in Medicine: Official Journal of the American College of Medical Genetics, 16(10), 727–735. https://doi.org/10.1038/gim.2014.26.
Gritz, E. R., Vernon, S. W., Peterson, S. K., Baile, W. F., Marani, S. K., Amos, C. I., et al. (1999). Distress in the cancer patient and its association with genetic testing and counseling for hereditary non-polyposis colon cancer. Cancer Research Therapy and Control, 8(1–2), 35–49. Retrieved from https://www.scopus.com/inward/record.uri?eid=2-s2.0-0032903642&partnerID=40&md5=edb57bec967cd68aec6ab0264ea87900
Gritz, E. R., Peterson, S. K., Vernon, S. W., Marani, S. K., Baile, W. F., Watts, B. G., et al. (2005). Psychological impact of genetic testing for hereditary nonpolyposis colorectal cancer. Journal of Clinical Oncology, 23(9), 1902–1910. https://doi.org/10.1200/JCO.2005.07.102.
Gryfe, R. (2009). Inherited colorectal cancer. Clinics in Colon and Rectal Surgery, 1(212), 198–208. https://doi.org/10.1055/s-0029-1242459.
Halbert, C. H., Stopfer, J. E., McDonald, J., Weathers, B., Collier, A., Troxel, A. B., et al. (2011). Long-term reactions to genetic testing for BRCA1 and BRCA2 mutations: Does time heal women’s concerns? Journal of Clinical Oncology, 29(32), 4302–4306. https://doi.org/10.1200/JCO.2010.33.1561.
Hamilton, J. G., Lobel, M., & Moyer, A. (2009). Emotional distress following genetic testing for hereditary breast and ovarian cancer: A meta-analytic review. Health Psychology, 28(4), 510–518. https://doi.org/10.1037/a0014778.Emotional.
Kazis, L. E., Selim, A., Rogers, W., Ren, X. S., Lee, A., Miller, D. R., et al. (1990). Veterans RAND 12 item health survey (VR-12): A white paper summary, (June 2016), 12–16.
Kazis, L. E., Miller, D. R., Clark, J. A., Skinner, K. M., Lee, A., Rogers, W. H., et al. (2004). Improving the response choices on the veterans SF-36 health survey role functioning scales. Journal of Ambulatory Care Management, 27(3), 263–280.
Kelly, M. J., Dunstan, F. D., Lloyd, K., & Fone, D. L. (2008). Evaluating cutpoints for the MHI-5 and MCS using the GHQ-12: A comparison of five different methods. BMC Psychiatry, 8(1), 10. https://doi.org/10.1186/1471-244X-8-10.
Kroenke, K., Spitzer, R. L., & Williams, J. B. W. (2001). The PHQ-9: Validity of a brief depression severity measure. Journal of General Internal Medicine, 16(9), 606–613. https://doi.org/10.1046/j.1525-1497.2001.016009606.x.
Lewis, K. L., Hooker, G. W., Connors, P. D., Hyams, T. C., Wright, M. F., Caldwell, S., et al. (2016). Participant use and communication of findings from exome sequencing: A mixed-methods study. Genetics in Medicine, 18(6), 577–583. https://doi.org/10.1038/gim.2015.133.
Manchanda, R., Loggenberg, K., Sanderson, S., Burnell, M., Wardle, J., Gessler, S., et al. (2015). Population testing for cancer predisposing BRCA1/BRCA2 mutations in the ashkenazi-jewish community: A randomized controlled trial. Journal of the National Cancer Institute, 107(1), 1–11. https://doi.org/10.1093/jnci/dju379.
Martin, A., Rief, W., Klaiberg, A., & Braehler, E. (2006). Validity of the brief patient health questionnaire mood scale (PHQ-9) in the general population. General Hospital Psychiatry, 28(1), 71–77. https://doi.org/10.1016/j.genhosppsych.2005.07.003.
Myers, R. E., Manne, S. L., Wilfond, B., Sifri, R., Ziring, B., Wolf, T. A., et al. (2011). A randomized trial of genetic and environmental risk assessment (GERA) for colorectal cancer risk in primary care: Trial design and baseline findings. Contemporary Clinical Trials, 32(1), 25–31. https://doi.org/10.1016/j.cct.2010.08.013.
NIH Research Portfolio Online Reporting Tools (RePORT). (2013). Genetic testing: how it is used for healthcare.
O’Neill, S. C., Rini, C., Goldsmith, R. E., Valdimarsdottir, H., Cohen, L. H., & Schwartz, M. D. (2009). Distress among women receiving uninformative BRCA1/2 results: 12-month outcomes. Psycho-Oncology, 18(10), 1088–1096. https://doi.org/10.1002/pon.1467.
Patrick, D. L., Burke, L. B., Gwaltney, C. J., Leidy, N. K., Martin, M. L., Molsen, E., et al. (2011). Content validity—Establishing and reporting the evidence in newly developed patient-reported outcomes (PRO) instruments for medical product evaluation: ISPOR PRO good research practices task force report: Part 2—Assessing respondent understanding. Value in Health, 14(8), 978–988. https://doi.org/10.1016/j.jval.2011.06.013.
Penziner, E., Williams, J. K., Erwin, C., Bombard, Y., Wallis, A., Beglinger, L. J., et al. (2008). Perceptions of discrimination among persons who have undergone predictive testing for Huntington’s disease. American Journal of Medical Genetics. Part B, Neuropsychiatric Genetics : The Official Publication of the International Society of Psychiatric Genetics, 147(3), 320–325. https://doi.org/10.1002/ajmg.b.30600.
Read, C. Y., Perry, D. J., & Duffy, M. E. (2005). Design and psychometric evaluation of the psychological adaptation to genetic information scale. Journal of Nursing Scholarship, 37(3), 203–208. https://doi.org/10.1111/j.1547-5069.2005.00036.x.
Rini, C., O’Neill, S. C., Valdimarsdottir, H., Goldsmith, R. E., Jandorf, L., Brown, K., et al. (2009). Cognitive and emotional factors predicting decisional conflict among high-risk breast cancer survivors who receive uninformative BRCA1/2 results. Health Psychology, 28(5), 569–578. https://doi.org/10.1037/a0015205.
Rumpf, H. J., Meyer, C., Hapke, U., & John, U. (2001). Screening for mental health: Validity of the MHI-5 using DSM-IV Axis I psychiatric disorders as gold standard. Psychiatry Research, 105(3), 243–253. https://doi.org/10.1016/S0165-1781(01)00329-8.
Selim, A. J., Rogers, W., Fleishman, J. A., Qian, S. X., Fincke, B. G., Rothendler, J. A., et al. (2009). Updated U.S. population standard for the veterans RAND 12-item health survey (VR-12). Quality of Life Research, 18(1), 43–52. https://doi.org/10.1007/s11136-008-9418-2.
Snaith, R. P. (2003). The hospital anxiety and depression scale. Health and Quality of Life Outcomes, 1, 29. https://doi.org/10.1186/1477-7525-1-29.
Spielberger, C. D. (1989). State-Trait Anxiety Inventory: Bibliography (2nd Editio ed.). Palo Alto: Consulting Psychologists Press.
Spitzer, R. L., Kroenke, K., Williams, J. B. W., & Lo, B. (2006). A brief measure for assessing generalized anxiety disorder. Archives of Internal Medicine, 166, 1092–1097. https://doi.org/10.1001/archinte.166.10.1092.
Vernon, S. W., Gritz, E. R., Peterson, S. K., Amos, C. I., Perz, C. A., Baile, W. F., et al. (1997). Correlates of psychologic distress in colorectal cancer patients undergoing genetic testing for hereditary colon cancer. Health Psychology, 16(1), 73–86. https://doi.org/10.1037/0278-6133.16.1.73.
Voils, C. I., Coffman, C. J., Edelman, D., Maciejewski, M. L., Grubber, J. M., Sadeghpour, A., et al. (2012). Examining the impact of genetic testing for type 2 diabetes on health behaviors: Study protocol for a randomized controlled trial. Trials, 13(1), 121. https://doi.org/10.1186/1745-6215-13-121.
Wakefield, C. E., Hanlon, L. V, Tucker, K. M., Patenaude, A. F., Signorelli, C., McLoone, J. K., et al. (2016). The psychological impact of genetic information on children: A systematic review. Genetics in Medicine: Official Journal of the American College of Medical Genetics, (August 2015), 1–8. https://doi.org/10.1038/gim.2015.181.
Ware, J. E., & Gandek, B. (1998). Overview of the SF-36 health survey and the international quality of life assessment (IQOLA) project. Journal of Clinical Epidemiology, 51(11), 903–912. https://doi.org/10.1016/S0895-4356(98)00081-X.
Watts, K. J., Meiser, B., Mitchell, G., Kirk, J., Saunders, C., Peate, M., et al. (2012). How should we discuss genetic testing with women newly diagnosed with breast cancer? Design and implementation of a randomized controlled trial of two models of delivering education about treatment-focused genetic testing to younger women newly diagnosed. BMC Cancer, 12(1), 320. https://doi.org/10.1186/1471-2407-12-320.
Weinberg, D. S., Myers, R. E., Keenan, E., Ruth, K., Sifri, R., Ziring, B., et al. (2014). Genetic and environmental risk assessment and colorectal cancer screening in an average-risk population: A randomized trial. Annals of Internal Medicine, 161(8), 537–545. https://doi.org/10.7326/M14-0765.
Weiss, D. S., & Marmar, C. R. (1996). The impact of event scale-revised. In J. Wilson & T. M. Keane (Eds.), Assessing psychological trauma and PTSD (pp. 399–411). New York: Gilford.
Westin, S. N., Sun, C. C., Lu, K. H., Schmeler, K. M., Soliman, P. T., Lacour, R. A., et al. (2011). Satisfaction with ovarian carcinoma risk-reduction strategies among women at high risk for breast and ovarian carcinoma. Cancer, 117(12), 2659–2667. https://doi.org/10.1002/cncr.25820.
Author Contributions Section
Meng Li, Caroline S. Bennette, David L. Veenstra and Donald L. Patrick contributed to the design of the work, analysis and interpretation of data, and drafting and revising the manuscript, approved the final version to be published, and agreed to be accountable for all aspects of the work.
Laura M. Amendola, M. Ragan Hart, Patrick Heagerty, Bryan Comstock, Peter Tarczy-Hornoch, Stephanie M. Fullerton, Dean A Regier, Wylie Burke, Susan B. Trinidad and Gail P. Jarvik contributed to the acquisition of data and critical revisions of the manuscript, approved the final version to be published, and agreed to be accountable for all aspects of the work.
Funding
This research received funding from the National Human Genome Research Institute and National Cancer Institute (grant numbers: U01HG0006507 and U01HG007307).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Authors Meng Li, Caroline S. Bennette, Laura M. Amendola, M. Ragan Hart, Patrick Heagerty, Bryan Comstock, Peter Tarczy-Hornoch, Stephanie M. Fullerton, Dean A Regier, Wylie Burke, Susan B. Trinidad, Gail P. Jarvik, David L. Veenstra, and Donald L. Patrick declare that they have no conflict of interest.
Informed Consent
This study obtained informed consent from all study participants.
Human Studies and Informed Consent
This study was approved by the University of Washington Institutional Review Board. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Animal Studies
No animal studies were carried out by the authors for this article.
Electronic supplementary material
ESM 1
(DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Li, M., Bennette, C.S., Amendola, L.M. et al. The Feelings About genomiC Testing Results (FACToR) Questionnaire: Development and Preliminary Validation. J Genet Counsel (2018). https://doi.org/10.1007/s10897-018-0286-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10897-018-0286-9