Abstract
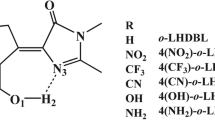
This article presents a comprehensive therotical investigation of excited state intramolecular proton transfer (ESIPT) for some newly-designed diphenylethylene derivatives containing 2-(2-hydroxy-phenyl)-benzotriazole moiety with various substituted groups. The calculation shows the structural parameters and Mulliken charges of phototautomers enol (E) and keto (K) of these compounds exhibit no or tiny changes from S0 to S1. The calculated results suggest that HOMO and LUMO + 1 of the compounds displays excellent overlapping nature, and thus the absorption and emission could be from the electron transition of HOMO→LUMO + 1. The electron density distribution in the frontier orbital of E and K are influenced remarkably by various substituted groups in S0 and S1 states. Electron density distribution deficiency in 2-(2-hydroxy-phenyl)-benzotriazole part is observed in L + 1 for these derivatives. The calculation also suggests the potential energy curves of ESIPT are shown to be a strong relationship with electron donor-acceptor groups. The absorption spectra, normal emission spectra and ESIPT spectra of the derivatives were also calculated.




Similar content being viewed by others
References
Ogawa AK, Abou-Zied OK, Tsui V, Jimenez R, Case DA, Romesberg FE (2000) A phototautomerizable model DNA base pair. J Am Chem Soc 122:9917–9920. doi:10.1021/ja001778n
Abou-Zied OK, Jimenez R, Romesberg FE (2001) Tautomerization dynamics of a model base pair in DNA. J Am Chem Soc 123(19):4613–4614. doi:10.1021/ja003647s
Rodembusch FS, Campo LF, Stefani V, Rigacci A (2005) The first silica areogels fluorescent by excited state intramolecular proton transfer mechanism(ESIPT). J Mater Chem 15:1537–1541. doi:10.1039/b416958a
Lim SJ, Seo J, Soo YP (2006) Photochromic switching of excited-State intramolecular proton-transfer (ESIPT) fluorescence: a unique route to high-contrast memory switching and nondestructive readout. J Am Chem Soc 128(45):14542–14547. doi:10.1021/ja0637604
Cardoso MB, Samios D, Silveira NP, Rodembusch FS, Stefani V (2007) ESIPT-exhibiting protein probes: a sensitive method for rice proteins detection during starch extraction. Photochem Photobiol Sci 6(1):99–102. doi:10.1039/b610999c
Mutai T, Tomoda H, Ohkawa T, Yabe Y, Araki K (2008) Switching of polymorph-dependent ESIPT luminescence of an imidazo[1, 2-a]pyridine derivative. Angew Chem Int Ed 47(49):9522–9524. doi:10.1002/anie.200803975
Chou PT, Huang CH, Pu SC, Cheng YM, Liu YH, Wang Y, Chen CT (2004) Tuning excited-state charge/proton transfer coupled reaction via the dipolar functionality. J Phys Chem A 108(31):6452–6454. doi:10.1021/jp0476390
Yu WS, Cheng CC, Cheng YM, Wu PC, Song YH, Chi Y, Chou PT (2003) Excited-state intramolecular proton transfer in five-membered hydrogen-bonding systems: 2-pyridyl pyrazoles. J Am Chem Soc 125:10800–10801. doi:10.1021/ja035382y
Chen CL, Lin CW, Hsieh CC, Lai CH, Lee GH, Wang CC, Chou PT (2009) Dual excited-state intramolecular proton transfer reaction in 3-hydroxy-2-(pyridine-2-yl)-4H-chromen-4-one. J Phys Chem A 113:205–214. doi:10.1021/jp809072a
Chen KY, Hsieh CC, Cheng YM, Lai CH, Chou PT (2006) Extensive spectral tuning of the proton transfer emission from 550 to 675 nm via a rational derivatization of 10-hydroxybenzo[h]quinoline. Chem Commun 4395–4397. doi:10.1039/b610274c
Chen KY, Cheng YM, Lai CH, Hsu CC, Ho ML, Lee GH, Chou PT (2007) Ortho green fluorescence protein synthetic chromophore; excited-state intramolecular proton transfer via seven-membered-ring hydrogen-bonding system. J Am Chem Soc 129:4534–4535. doi:10.1021/ja070880i
Seo J, Kim S, Park SY (2004) Strong solvatochromic fluorescence from the intramolecular charge-transfer state created by excited-state intramolecular proton transfer. J Am Chem Soc 126(36):11154–11155. doi:10.1021/ja047815i
Sakai K, Ichikawaa M, Taniguchi Y (2006) Photoluminescent mechanism of a proton-transfer laser dye in highly doped polymer films. Chem Phys Lett 420:405–409. doi:10.1016/j.cplett.2005.12.074
Kim TI, Kang HJ, Han G, Chung SJ, Kim Y (2009) A highly selective fluorescent ESIPT probe for the dual specificity phosphatase MKP-6. Chem Commun 5895–5897. doi:10.1039/B911145J
Zhang H, Han LF, Zachariasse KA, Jiang YB (2005) 8-hydroxyquinoline benzoates as highly sensitive fluorescent chemosensors for transition metal ions. Org Lett 7(19):4217–4220. doi:10.1021/ol051614h
Sun WH, Li SY, Hu R, Qian Y, Wang SQ, Yang GQ (2009) Understanding solvent effects on luminescent properties of a triple fluorescent ESIPT compound and application for white light emission. J Phys Chem A 113(20):5888–5895. doi:10.1021/jp900688h
Park S, Kwon OH, Kim S, Park S, Choi MG, Cha M, Park SY, Jang DJ (2005) Imidazole-based excited-state intramolecular proton-transfer materials: synthesis and amplified spontaneous emission from a large single crystal. J Am Chem Soc 127(28):10070–10074. doi:10.1021/ja0508727
Paterson MJ, Robb MA, Blancafort L, DeBellis AD (2004) Theoretical study of benzotriazole UV photostability: ultrafast deactivation through coupled proton and electron transfer triggered by a charge-transfer state. J Am Chem Soc 126(9):2912–2922. doi:10.1021/ja0386593
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega, N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA, Gaussian 03, Revision B.03.
Zgiershi MZ, Grabowska A (2000) Photochromism of salicylideneaniline (SA). How the photochromic transient is created: a theoretical approach. J Chem Phys 112(14):6329–69367. doi:10.1063/1.481194
Liang YP, Cao YH (2005) Intramolecular proton or hydrogen-atom transfer in the ground- and excited-states of 2-hydroxybenzophenone: a theoretical study. Chem Phys 315(3):297–302. doi:10.1016/j.cplett.2007.03.010
Liang YH, Yi PG (2007) Theoretical studies on structure, energetic and intramolecular proton transfer of alkannin. Chem Phys Lett 438(4–6):173–177. doi:10.1016/j.cplett.2007.03.010
Yang ZN, Yang SY, Zhang JP (2007) Ground- and excited-state proton transfer and rotamerism in 2-(2-Hydroxyphenyl)-5-phenyl-1, 3, 4-oxadiazole and its O/“NH or S”-substituted derivatives. J Phys Chem A 111(28):6354–6360. doi:10.1021/jp068589x
Gaenko AV, Devarajan A, Tselinskii IV, Ryde U (2006) Structural and photoluminescence properties of excited state intramolecular proton transfer capable compounds—potential emissive and electron transport materials. J Phys Chem A 110 (25): 7935–7942 doi:10.1021/jp060646z
Gorelsky SI Swizard program, revision XX, http://www.sg-chem.net/
Wang Y, Wu G (2008) Electronic structure characteristics of ESIPT and TICT fluorescence emissions and calculations of emitting energies. Acta Phys–Chim Sim 24:552–560, in Chinese
Acknowledgements
The authors would thank National Natural Science Foundation of China (Nos. 20776165, 20702065 and 20872184) and Natural Science Foundation of CQ CSTC (Nos. CSTC, 2010BB0216) for financial supports. The authors appreciate for the support from the Key Foundation of Chongqing Science and Technology Commission (CSTC 2008BA4020). We appreciate financial supports from the Fundamental Research Funds for the Central Universities (No.CDJZR10220006, CDJXS10221137, CDJZR10225501).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, H., Niu, L., Xu, X. et al. A Comprehensive Therotical Investigation of Intramolecular Proton Transfer in the Excited States for Some Newly-designed Diphenylethylene Derivatives Bearing 2-(2-Hydroxy-Phenyl)-Benzotriazole Part. J Fluoresc 21, 1721–1728 (2011). https://doi.org/10.1007/s10895-011-0867-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-011-0867-6