Abstract
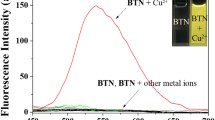
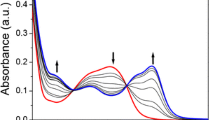
A new 7-nitrobenz-2-oxa-1,3-diazole (NBD) derived fluorescent probe (1) exhibiting high selectivity for Cu2+ detection, produced significant fluorescence quenching in the presence of Cu2+ ion, while the metal ions Ca2+, Cd2+, Co2+, Fe2+, Hg2+, Mg2+, Mn2+, Ni2+ and Zn2+ produced only minor changes in fluorescence. The apparent association constant (K a) for Cu2+ binding in chemosensor 1 was found to be 1.22 × 103 M−1. The maximum fluorescence quenching activity caused by Cu2+ binding to 1 was observed over the pH range 6–10.












Similar content being viewed by others
References
Cowan JA (1997) Inorganic biochemistry: an introduction. Wiley-VCH, New York, pp 133–134
Barnham KJ, Masters CL, Bush AI (2004) Neurodegenerative diseases and oxidative stress. Nat Rev Drug Discovery 3:205–214
Waggoner DJ, Bartnikas TB, Gitlin JD (1999) The role of copper in neurodegenerative disease. Neurobiol Dis 6:221–230
Vulpe C, Levinson B, Whitney S, Packman S, Gitschier J (1993) Isolation of a candidate gene for Menkes disease and evidence that it encodes a copper-transporting ATPase. Nat Genet 3:7–13
Gonzales APS, Firmino MA, Nomura C, Rocha FRP, Oliveira PV, Gaubeur I (2009) Peat as a natural solid-phase for copper preconcentration and determination in a multicommuted flow system coupled to flame atomic absorption spectrometry. Anal Chim Acta 636:198–204
Becker JS, Matusch A, Depboylu C, Dobrowolska J, Zoriy MV (2007) Quantitative imaging of selenium, copper, and zinc in thin sections of biological tissues (Slugs-Genus Arion) measured by laser ablation inductively coupled plasma mass spectrometry. Anal Chem 79:6074–6080
Liu Y, Liang P, Guo L (2005) Nanometer titanium dioxide immobilized on silica gel as sorbent for preconcentration of metal ions prior to their determination by inductively coupled plasma atomic emission spectrometry. Talanta 68:25–30
Ensafi AA, Khayamian T, Benvidi A (2006) Simultaneous determination of copper, lead and cadmium by cathodic adsorptive stripping voltammetry using artificial neural network. Anal Chim Acta 561:225–232
Torrado A, Walkup GK, Imperiali B (1998) Exploiting polypeptide motifs for the design of selective Cu(II) ion chemosensors. J Am Chem Soc 120:609–610
Zheng Y, Huo Q, Kele P, Andreopoulos FM, Pham SM, Leblanc RM (2001) A new fluorescent chemosensor for copper ions based on tripeptide glycyl−histidyl−lysine (GHK). Org Lett 3:3277–3280
Zeng L, Miller EW, Pralle A, Isacoff EY, Chang CJ (2006) A selective turn-on fluorescent sensor for imaging copper in living cells. J Am Chem Soc 128:10–11
Qi X, Jun EJ, Xu L, Kim S, Hong JSJ, Yoon YJ, Yoon J (2006) New BODIPY derivatives as off-on fluorescent chemosensor and fluorescent chemodosimeter for Cu2+: cooperative selectivity enhancement toward Cu2+. J Org Chem 71:2881–2884
Xiang Y, Tong A, Jin P, Ju Y (2006) New fluorescent rhodamine hydrazone chemosensor for Cu(II) with high selectivity and sensitivity. Org Lett 8:2863–2866
Yang H, Liu Z, Zhou Z, Shi E, Li F, Du Y, Yi T, Huang C (2006) Highly selective ratiometric fluorescent sensor for Cu(II) with two urea groups. Tetrahedron Lett 47:2911–2914
Martinez R, Zapata F, Caballero A, Espinosa A, Tarraga A, Molina P (2006) Rhodamine-diacetic acid conjugate 2-aza-1, 3-butadiene derivatives featuring an anthracene or pyrene unit: highly selective colorimetric and fluorescent signaling of Cu2+ cation. Org Lett 8:3235–3238
Zhang X, Shiraishi Y, Hirai T (2007) Cu(II)-selective green fluorescence of a rhodamine-diacetic acid conjugate. Org Lett 9:5039–5042
Li G, Xu Z, Chen C, Huang Z (2008) A highly efficient and selective turn-on fluorescent sensor for Cu2+ ion based on calix[4]arene bearing four iminoquinoline subunits on the upper rim. Chem Comm 1774–1776
Wu S, Liu S (2009) A new water-soluble fluorescent Cu(II) chemosensor based on tetrapeptide histidyl-glycyl-glycyl-glycine (HGGG). Sens Actuators B 141:187–191
Ballesteros E, Moreno D, Gomez T, Rodriguez T, Rojo J, Garcia-Valverde M, Torroba T (2009) A new selective chromogenic and turn-on fluorogenic probe for copper(II) in water-acetonitrile 1:1 solution. Org Lett 11:1269–1272
Jung HS, Kwon PS, Lee JW, Kim J, Hong CS, Kim JW, Yan S, Lee JY, Lee JW, Joo T, Kim S (2009) Coumarin-derived Cu2+-selective fluorescence sensor:synthesis, mechanisms, and applications in living cells. J Am Chem Soc 131:2008–2012
Zhou Y, Wang F, Kim Y, Kim S, Yoon J (2009) Cu2+-selective ratiometric and “off-on” sensor based on the rhodamine derivative bearing pyrene group. Org Lett 11:4442–4445
He G, Zhao X, Zhang X, Fan H, Wu S, Li H, He C, Duan C (2010) A turn-on PET fluorescence sensor for imaging Cu2+ in living cells. New J Chem 34:1055–1058
Goswami S, Sen D, Das NK (2010) A new highly selective, ratiometric and colorimetric fluorescence sensor for Cu2+ with a remarkable red shift in absorption and emission spectra based on internal charge transfer. Org Lett 12:856–859
Bukowski MR, Zhu S, Koehntop KD, Brennessel WW, Que LJ (2004) Characterization of an FeIII-OOH species and its decomposition product in a bleomycin model system. J Biol Inorg Chem 9:39–48
Senthilvelan A, Ho I, Chang K, Lee G, Liu Y, Chung W (2009) Cooperative recognition of a copper cation and anion by a calix[4]arene substituted at the lower rim by a β-Amino-α, β-unsaturated ketone. Chem Eur J 15:6152–6160
Benesi HA, Hildebrand JH (1949) A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J Am Chem Soc 71:2703–2707
Singh RB, Mahanta S, Guchhait N (2010) Spectral modulation of charge transfer fluorescence probe encapsulated inside aqueous and non-aqueous β-cyclodextrin nanocavities. J Mol Struct 963:92–97
Acknowledgements
We gratefully acknowledge the financial supports of the National Science Council (ROC) and National Chiao Tung University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, SR., Wu, SP. An NBD-based Sensitive and Selective Fluorescent Sensor for Copper(II) Ion. J Fluoresc 21, 1599–1605 (2011). https://doi.org/10.1007/s10895-011-0848-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-011-0848-9