Abstract
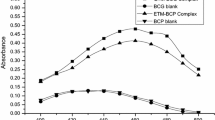
Simple and sensitive spectrophotometric and spectrofluorimetric methods have been developed for the determination of hydrochlorothiazide (I), indapamide (II) and xipamide(III) based on ternary complex formation with eosin and lead (II) in the presence of methylcellulose as surfactant. The methods do not involve solvent extraction. For spectrophotometric method, the ternary complex showed an absorption maximum at 543 nm. The factors affecting the formation of ternary complex were studied and optimized. The method obeys Beer’s law over concentration range of 8–40 μg mL−1. A fluorescence quenching method for the determination of the cited drugs by forming this ternary complex was also investigated for the purpose of enhancing the sensitivity of the determination. The analytical performance of both methods was fully validated, and the results were satisfactory. The methods have been successfully applied for the determination of the studied drugs in their pharmaceutical tablets and the results obtained ware in good agreement with those obtained by the reference method. Common excipients used as additives in tablets do not interfere with the proposed methods.


Similar content being viewed by others
References
Manfred EW (1994) Burger’s medicinal chemistry and drug discovery, 5th edn. John Wiley & Sons, New York, NY, USA, pp 386–387
Bedair MM, Korany MA, Ebdel-Hay MA, Gazy AA (1990) Analyst 115(4):449–453
Panderi I, Parissi-Poulou M (1994) J Pharm Biomed Anal 12(2):151–156
Ferraro MC, Castellano PM, Kaufman TS (2002) J Pharm Biomed Anal 30(4):1121–1131
Dince E, Baleanu D (2002) J Pharm Biomed Anal 30(3):715–723
Albero I, Rodenos V, Garcia S, Sanchez-Pedreno C (2002) J Pharm Biomed Anal 29(1–2):299–305
Youssef NF (2003) J AOAC Int 86(5):935–940
Agrawal YK, Majumdar FD (1995) Anal Lett 28(9):1619–1627
Sastry CSP, Suryanarayana MV, Tipirneni ASRP (1989) Indian Drugs 26(6):304–306
Sastry CSP, Suryanarayana MV, Tipirneni ASRP (1989) Talanta 36(4):491–494
El-Kommos ME, Ahmad A, Salem H, Omar MA (2006) Bull Pharm Sci 29(1):33–58
Garg G, Saraf S, Saraf S (2008) J AOAC Int 91(5):1045–1050
Brown SM, Busch KL (1991) J PlanarChromatogr Mod TLC 4(3):189–193
Carda-Broch S, Esteve-Romero JS, Garcia-Alvarez-Coque MC (1998) Anal Chem Acta 375(1–2):143–154
Frontini R, Mielck JBJ (1992) Liq Chromatogr 15(14):2519–2528
Torres-Lapasio JR, Baeza-Baeza JJ, Garcia-Alvarez-Coque MC (1997) J Chromatogr A 769(2):155–168
Dadgar D, Kelly M (1988) Analyst 113(2):229–231
Deventer K, Pozo OJ, Van Eenoo P, Delbeke FT (2009) J Chromatogr A 20(12):2466–2473
Sultana N, Arayne MS, Ali SS, Sajid S (2008) Se Pu 26(5):544–549
Obando MA, Estela JM, Cerdà V (2008) Anal Bioanal Chem 391(6):2349–2356
Rane VP, Sangshetti JN, Shinde DB (2008) J Chromatogr Sci 46(10):887–891
Yan T, Li H, Deng L, Guo Y, Yu W, Fawcett JP, Zhang D, Cui Y, Gu J (2008) J Pharm Biomed Anal 48(4):1225–1229
Gonzalez E, Becerra A, Laserna JJ (1996) J Chromatogr B Biomed-Appl 687(1):145–150
Quaglia MG, Donati E, Carlucci G, Mazzeo P, Fanali S (2002) J Pharm Biomed Anal 29(6):981–987
Gonzalez E, Montes R, Laserna J (1993) J Anal Chem Acta 282(2):687–693
Zheng X, Lu M, Zhang L, Chi Y, Zheng L, Chen G (2008) Talanta 76(1):15–20
Liu X, Song Y, Yue Y, Zhang J, Chen X (2008) Electrophoresis 29(13):2876–2883
Sirén H, Shimmo R, Sipola P, Abenet S, Riekkola ML (2008) J Chromatogr A 1198–1199:215–219
Zhou N, Liang YZ, Wang B, Wang P, Chen X, Zeng MM (2008) Biomed Chromatogr 22(3):223–231
Mohamed ME, Aboul-Enein HY (1985) Anal Lett 18(20):2591–2603
Legorburu MJ, Alonso RM, Jimenez RM (1993) Bioelectrochem-Bioenerg 32(1):57–66
Legurburu MJ, Alonso RM, Jimenez RM (1996) Electroanalysis 8(3):280–284
Hillaert S, De Grauwe K, Van den Bossche W (2001) J Chromatogr A 249(1–2):439–449
Abdel Razak O (2004) J Pharm Biomed Anal 34(2):433–440
Sakai T (1984) Dojin News 30:1
Abdellatef HE, Ayad MM, Taha EA (1999) J Pharm Biomed Anal 18:1021–1027
Ayad MM, Shalaby AA, Abdellatef HE, Hosny MM (2002) J Pharm Biomed Anal 28:311–321
Abdellatef HE (2007) Spectrochimica Acta Part A 66:701–706
Kelani K, Bebawy LI, Abdel-Fattah L (1999) J Pharm Biomed Anal 18:985–992
ICH Harmonized Tripartie Guidline Q2A: “Text on validation of analytical procedures”. (available at http://www.ich.org): Incorporated in November (2005)
ICH Harmonized Tripartie Guidline Q2B (R1): “ Validation of Analytical Proceudures: Text and Methodology”. (available at http://www.ich.org): Incorporated in November 2005
Smith GP, Zui LC (1977) Analytical profiles of drug substances and excipients, Vol. 23. Academic Press, p 572, 575
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Omar, M.A. Spectrophotometric and Spectrofluorimetric Determination of Certain Diuretics Through Ternary Complex Formation with Eosin and Lead (II). J Fluoresc 20, 275–281 (2010). https://doi.org/10.1007/s10895-009-0551-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-009-0551-2