Abstract
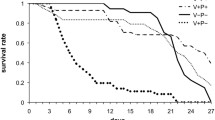
Growing evidence suggests that the freely accessible pollen of some plants is chemically protected against pollen-feeding flower visitors. For example, a diet of pollen from buttercup plants (Ranunculus) recently was shown to have a deleterious effect on developing larvae of several bee species not specialized on Ranunculus. Numerous Ranunculus species contain ranunculin, the glucosyl hydrate form of the highly reactive and toxic lactone protoanemonin, that causes the toxicity of these plants. We tested whether the presence of ranunculin is responsible for the lethal effects of R. acris pollen on the larvae of two bee species that are not Ranunculus specialists. To investigate the effect on bee larval development, we added ranunculin to the pollen provisions of the Campanula specialist bee Chelostoma rapunculi and the Asteraceae specialist bee Heriades truncorum, and allowed the larvae to feed on these provisions. We quantified ranunculin in pollen of R. acris and in brood cell provisions collected by the Ranunculus specialist bee Chelostoma florisomne. We demonstrated that although ranunculin was lethal to both tested bee species in high concentrations, the concentration in the pollen of R. acris was at least fourfold lower than that tolerated by the larvae of C. rapunculi and H. truncorum in the feeding experiments. Ranunculin concentration in the brood cells of C. florisomne was on average even twentyfold lower than that in Ranunculus pollen, suggesting that a mechanism different from ranunculin intoxication accounts for the larval mortality reported for bees not specialized on Ranunculus pollen.


Similar content being viewed by others
References
Bai, Y., Benn, M. H., Majak, W., and McDiarmid, R. 1996. Extraction and HPLC determination of ranunculin in species of the buttercup family. J. Agr. Food Chem. 44:2235–2238.
Benn, M. H. and Yelland, L. J. 1968. Ranunculin. Can. J. Chem. 46:729–732.
Bergström, G., Dobson, H. E. M., and Groth, I. 1995. Spatial fragrance patterns within the flowers of Ranunculus acris (Ranunculaceae). Plant. Syst. Evol. 195:221–242.
Bhattacharya, P. R., Nath, S. C., and Bordoloi, D. N. 1993. Insecticidal activity of Ranunculus sceleratus (L.) against Drosophila melanogaster and Tribolium castaneum. Indian J. Exp. Biol. 31:85–86.
Bonora, A., Botta, B., Menziani-Andreoli, E., and Bruni, A. 1988. Organ-specific distribution and accumulation of protoanemonin in Ranunculus ficaria L. Biochem. Physiol. Pfl. 183:443–447.
Campbell, W. E., Cragg, G. M. L., and Powrie, A. H. 1979. Anemonin, protoanemonin and ranunculin from Knowltonia capensis. Phytochemistry 18:323–324.
Castellanos, M. C., Wilson, P., Keller, S. J., Wolfe, A. D., and Thomson, J. D. 2006. Anther evolution: pollen presentation strategies when pollinators differ. Am. Nat. 167:288–296.
de Carvalho, A. C. P. and Message, D. 2004. A scientific note on the toxic pollen of Stryphnodendron polyphyllum (Fabaceae, Mimosoideae) which causes sacbrood-like symptoms. Apidologie 35:89–90.
Dobson, H. E. M. and Peng, Y. S. 1997. Digestion of pollen components by larvae of the flower-specialist bee Chelostoma florisomne (Hymenoptera: Megachilidae). J. Insect Physiol. 43:89–100.
Harder, L. D. and Barclay, R. M. R. 1994. The functional significance of poricidal anthers and buzz pollination: controlled pollen removal from Dodecatheon. Funct. Ecol. 8:509–517.
Hill, R. and van Heyningen, R. 1951. Ranunculin: the precursor of the vesicant substance of the buttercup. Biochem. J. 49:332–335.
Jürgens, A. and Dötterl, S. 2004. Chemical composition of anther volatiles in Ranunculaceae: genera-specific profiles in Anemone, Aquilegia, Caltha, Pulsatilla, Ranunculus, and Trollius species. Am. J. Bot. 91:1969–1980.
Kingsbury, J. M. 1964. Poisonous Plants of the United States and Canada. Prentice-Hall, Englewood Cliffs, NJ.
Kremen, C., Williams, N. M., Aizen, M. A., Gemmill-Herren, B., Lebuhn, G., Minckley, R., Packer, L., Potts, C. G., Roulston, T., Steffan-Dewenter, I., Vazquez, P., Winfree, R., Adams, L., Crone, E. E., Greenleaf, S. S., Keitt, T. H., Klein, A., Regetz, J., and Ricketts, T. H. 2007. Pollination and other ecosystem services produced by mobile organisms: a conceptual framework for the effects of land-use change. Ecol. Let. 10:299–314.
Lee, E. T. and Wang, J. W. 2003. Statistical Methods for Survival Data Analysis. Wiley and Sons, New York, NY.
Majak, W. 2001. Review of toxic glycosides in rangeland and pasture forages. J Range Manage 54:494–498.
Mares, D. 1987. Antimicrobial activity of protoanemonin, a lactone from ranunculaceous plants. Mycopathologia 98:133–140.
Martin, M. L., Roman, L. S., and Dominguez, A. 1990. In vitro activity of protoanemonin, an antifungal agent. Planta Med. 56:66–69.
Mauch, F. and Staehelin, L. A. 1989. Functional implications of the subcellular localization of ethylene-induced chitinase and β-1,3-glucanase in bean leaves. Plant Cell 1:447–457.
Morgenthaler, O. and Maurizio, A. 1941. Die “Bettlacher Maikrankheit”, eine Vergiftung durch Hahnenfuss-Pollen. Schweizerische Bienenzeitung 1:447–457.
Müller, A. 1996. Host-plant specialization in western palearctic anthidiine bees (Hymenoptera: Apoidea: Megachilidae). Ecol. Monogr. 66:235–257.
Müller, A., Diener, S., Schnyder, S., Stutz, K., Sedivy, C., and Dorn, S. 2006. Quantitative pollen requirements of solitary bees: Implications for bee conservation and the evolution of bee-flower relationships. Biol. Conserv. 130:604–615.
Piskorski, R., Ineichen, S., and Dorn, S. 2011a. Ability of the oriental fruit moth Grapholita molesta (Lepidoptera: Tortricidae) to detoxify juglone, the main secondary metabolite in the non-host plant walnut. J. Chem. Ecol. 37:1110–1116.
Piskorski, R., Kroder, S., and Dorn, S. 2011b. Can pollen headspace volatiles and pollenkitt lipids serve as reliable chemical cues for bee pollinators? Chem. Biodivers. 8:577–586.
Pitkin, B., Ellis, W., Plant, C., and Edmunds, R. 2010. The leaf and stem mines of British flies and other insects. ‹http://www.ukflymines.co.uk›.
Praz, C. J., Müller, A., and Dorn, S. 2008a. Specialized bees fail to develop on non-host pollen: do plants chemically protect their pollen? Ecology 89:795–804.
Praz, C. J., Müller, A., and Dorn, S. 2008b. Host recognition in a pollen-specialist bee: evidence for a genetic basis. Apidologie 39:547–557.
Ruijgrok, H. W. L. 1966. The distribution of ranunculin and cyanogenetic compounds in the Ranunculaceae. Meeting of the Phytochemical Group. Comparative Phytochemistry, Cambridge UK, 1965:175-186.
Sedivy, C., Praz, C. J., Müller, A., Widmer, A., and Dorn, S. 2008. Patterns of host-plant choice in bees of the genus Chelostoma: the constraint hypothesis of host-range evolution in bees. Evolution 62:2487–2507.
Sedivy, C., Müller, A., and Dorn, S. 2011. Closely related pollen generalist bees differ in their ability to develop on the same pollen diet: evidence for physiological adaptations to digest pollen. Funct. Ecol. 25:718–725.
Spencer, K. A. 1990. Host Specialization in the World Agromyzidae (Diptera). Kluwer Academic Publishers, Dordrecht, Netherlands.
Varitimidis, C., Petrakis, P. V., Vagias, C., and Roussis, V. 2006. Secondary metabolites and insecticidal activity of Anemone pavonina. J. Biosciences 61:521–526.
Vogel, S. 1993. Betrug bei Pflanzen: Die Täuschblumen. Abhandlungen der Akademie der Wissenschaften und der Literatur in Mainz 1:5–48.
Westerkamp, C. 1997. Keel blossoms: bee flowers with adaptations against bees. Flora 192:125–132.
Westerkamp, C. and Classen-Bockhoff, R. 2007. Bilabiate flowers: the ultimate response to bees? Ann. Bot. 100:361–374.
Westrich, P. 1989. Die Wildbienen Baden-Württembergs. Ulmer, Stuttgart, Germany.
Acknowledgments
We thank Michael H. Benn (University of Calgary, Canada) for the gift of a substantial sample of ranunculin; Renato Zenobi and Louis Bertschi (MS service of the Laboratory of Organic Chemistry, ETH Zurich) for support and chemical analyses; the staff of the Neolithic village of Gletterens for providing bee nests; Bettina Gutbrodt and Mare Haider for technical support, and Heather Kirk and three anonymous reviewers for useful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sedivy, C., Piskorski, R., Müller, A. et al. Too Low to Kill: Concentration of the Secondary Metabolite Ranunculin in Buttercup Pollen does not Affect Bee Larval Survival. J Chem Ecol 38, 996–1002 (2012). https://doi.org/10.1007/s10886-012-0153-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-012-0153-3