Abstract
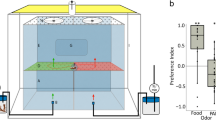
Mate location responses of male and female solitary-reared locusts that had either experienced no crowding or that had been crowded for varying periods were studied in a flatbed wind tunnel. Two hypotheses were explored: that both sexes of this phase of the locust participate in locating the other by using a combination of chemical and visual signals, and that individuals that experience some crowding (i.e., undergo varying levels of phase shift) can compete effectively with their solitary counterparts in mate location and mating. Our results confirm that both male and female solitarious locusts actively participate in mate location, although the former is the more aggressive partner. The responses of the insects are stronger when a visual cue is provided with the olfactory signal. Crowding of solitary-reared adults enhances their responsiveness to the other sex in the absence and presence of the visual cue. This phenomenon may constitute one of several mechanisms that are involved in recruiting solitary individuals into gregarizing groups and facilitating the spread of gregarious characters across a reproductively active solitarious population.




Similar content being viewed by others
References
Ameismeier, F. 1987. Ultra structure of the chemosensitive basiconic single-walled wall-pore sensilla on the antennae in adults and embryonic stages of Locusta migratoria L. (Insecta, Orthoptera). Cell Tissue Res. 247:605–612.
Bashir, M. O., Hassanali, A., Rai, M. M., and Saini, R. K. 2000. Changing oviposition preferences of the desert locust Schistocerca gregaria (Forskål) suggest a strong species predisposition for gregarisation. J. Chem. Ecol. 26:1721–1733.
Bouaichi, A., Roessingh, P., and Simpson, S. J. 1995. An analysis of behavioral effects of crowding and re-isolation on solitary reared adult desert locusts (Schistocerca gregaria) and their offspring. Physiol. Entomol. 20:199–208.
Bouaichi, A., Simpson, S. J., and Roessingh, P. 1996. The effluence of environmental microstructure on the behavioral phase state and distribution of the desert locust Schistocerca gregaria. Physiol. Entomol. 21:247–256.
Byer, J. A. 1991. Pheromones and chemical ecology of locusts. Biol. Rev. 66:347–378.
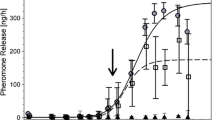
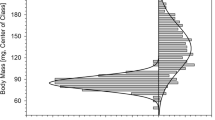
Deng, A. L., Torto, B., Hassanali, A., and Ali, E. E. 1996. Effects of shifting to crowded or solitary conditions on pheromone release and morphometrics of the desert locust Schistocerca gregaria (Forskål) (Orthoptera: Acrididae). J. Insect Physiol. 42:771–776.
El Bashir, S., Inayatullah, C., Ahmed, A. O., and Abdel Rahman, H. E. 1993. Models for estimation of population density of late instar nymphs and fledglings of the desert locust, Schistocerca gregaria (Forskål). Int. J. Pest Manag. 39:467–470.
Greenwood, M. and Chapman, R. F. 1984. Differences in numbers of sensilla on the antennae of solitarious and gregarious Locusta migratoria L. (Orthoptera: Acrididae). Int. J. Insect Morphol. Embryol. 13:295–301.
Hassanali, A., Njagi, P. G. N., and Bashir, M. O. 2005. Chemical ecology of locusts related acridids. Annu. Rev. Entomol. 50:223–245.
Inayatullah, C., El Bashir, S., and Hassanali, A. 1994. Sexual behavior and communication in the desert locust Schistocerca gregaria (Orthoptera: Acrididae): sex pheromone in solitaria. Environ. Entomol. 23:1544–1551.
Malual, A. G., Hassanali, A., Torto, B., Assad, Y. O. H., and Njagi, P. G. N. 2001. The nature of gregarizing signal responsible for maternal transfer of phase to the offspring in the desert locust Schistocerca gregaria. J. Chem. Ecol. 27:1423–1435.
McCaffery, A. R., Simpson, S. J., Saiful Islam, M., and Roessingh, P. 1998. A gregarising factor present in the egg pod of the desert locust Schistocerca gregaria. J. Exp. Biol. 201:347–363.
Njagi, P. G. N., Torto, B., Obeng-Ofori, D., and Hassanali, A. 1996. Phase-independent responses to phase-specific aggregation pheromone in adult desert locust Schistocerca gregaria (Forskål) (Orthoptera: Acrididae). Physiol. Entomol. 21:131–137.
Ochieng', S. A., Hallberg, E., and Hansson, B. S. 1998. Fine structure and distribution of antennal sensilla of the desert locust Schistocerca gregaria (Forskål) (Orthoptera: Acrididae). Cell Tissue Res. 291:525–536.
Ochieng-Odero, J. P. R., Ndugo, S. M., El Bashir, S., and Capstick, P. B. 1994. A method of rearing crowded and isolated locusts (Orthoptera: Acrididae) in the laboratory, pp. 33–44, in P. G. N. Njagi and M. F. B. Chaudhury (eds.). Proceedings of the Workshop on Effective Networking of Research and Development on Environmentally Sustainable Locust Control Methods Among Locust-Affected Countries. ICIPE Science Press, Nairobi, Kenya.
Popov, G. B. 1958. Ecological studies on oviposition by swarms of the desert locust Schistocerca gregaria (Forskål). Entomol. Exp. Appl. 6:279–303.
Riede, K. 1987. A comparative study of mating behavior in some neotropical grasshoppers (Acridoidea). Ethology 76:265–296.
Roessingh, P. and Simpson, S. J. 1994. The time course of behavioral phase change in nymphs of the desert locust Schistocerca gregaria. Physiol. Entomol. 19:191–197.
Roffey, J. and Popov G. 1968. Environmental and behavioural processes in a desert locust outbreak. Nature 219:446–450.
Siddiqui, J. I. and Khan, M. A. 1981. The secretion and perception of a sex pheromone in the grasshopper Hieroglyphus nigrorepletus Bolivar (Orthoptera: Acrididae). Acrida 10:233–242.
Stower, W. J., Popov, B., and Greathead, D. J. 1958. Oviposition behavior and egg mortality of the desert locust (Schistocerca gregaria Forskål) on the coast of the Eritrea. Anti-Locust Bull. 30:1–33.
Uvarov, B. P. 1977. Grasshoppers and Locusts: A Handbook of General Acridology, Vol. 3. Centre for Overseas Pest Research, Cambridge, UK.
Wallace, G. K. 1959. Visual scanning in the desert locust Schistocerca gregaria (Forskål). J. Exp. Biol. 36:512–525.
Whitman, D. W. 1982. Grasshopper sexual pheromone: A component of the defensive secretion in Taeniopoda eques. Physiol. Entomol. 7:111–115.
Whitman, D. W. 1990. Grasshopper chemical communication, pp. 357–391, in R. F. Chapman and A. Joern (eds.). Biology of Grasshoppers. John Wiley and Sons, New York.
Acknowledgments
This work was supported by The Program for Co-operation with International Institutes (SII), formerly DSO, and The Swiss Agency for Development and Cooperation (SDC) through FAO, to which we are most grateful. The assistance provided by Messrs. M. Miti and the late J. Ongudha, ICIPE's Animal Rearing Unit, in rearing the locusts is appreciated.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ould Ely, S., Mahamat, H., Njagi, P.G.N. et al. Mate Location Mechanism and Phase-Related Mate Preferences in Solitarius Desert Locust, Schistocerca gregaria . J Chem Ecol 32, 1057–1069 (2006). https://doi.org/10.1007/s10886-006-9045-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-006-9045-8