Abstract
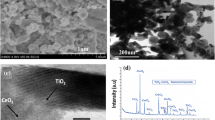
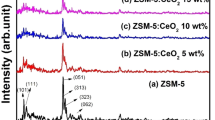
CeO2/zeolite nanocomposite was successfully prepared by the mixing-calcination method. The structural characteristics of photocatalyst were investigated by XRD, SEM, TEM and EDX. Photocatalytic degradation experiments were carried out with varying amounts of the CeO2/zeolite, the ratio of 3:1 (CeO2/zeolite) was exhibited excellent photocatalytic activity towards dye degradation. Synergistic effect of CeO2/zeolite played a key role in photocatalytic degradation. The main reactive oxygen species was determined by trapping experiments. Additionally, the recyclability was tested up to the fourth cycle. The CeO2/zeolite nanocomposite is a promising photocatalyst for removing trace and unprocessed organic contaminants in the industrial dye waste water treatment. The efficiency of CeO2/zeolite nanocomposite offers a potential economical route to degrade organic contaminants and recovering photocatalyst simultaneously.

















Similar content being viewed by others
References
M. Zhou, Q. Yu, L. Lei, and G. Barton (2007). Sep. Purif. Technol. 57, 380.
H. Zangeneh, A. A. L. Zinatizadeh, M. Habibi, M. Akia, and M. H. Isa (2015). J. Ind. Eng. Chem. 26, 1.
W. Li, S. Zhao, B. Qi, Y. Du, X. Wang, and M. Huo (2009). Appl. Catal. B. 92, 333.
H. S. Rai, M. S. Bhattacharyya, J. Singh, T. K. Bansal, P. Vats, and U. C. Banerjee (2005). Crit. Rev. Environ. Sci. Technol. 35, 219.
S. Song, L. Xu, Z. He, H. Ying, J. Chen, X. Xiao, and B. Yan (2008). J. Hazard. Mater. 152, 1301.
U. G. Akpan and B. H. Hameed (2009). J. Hazard. Mater. 170, 520.
K. H. Kim and S. K. Ihm (2011). J. Hazard. Mater. 186, 16.
F. Han, V. S. R. Kambala, M. Srinivasan, D. Rajarathnam, and R. Naidu (2009). Appl. Catal. A 359, 25.
A. Hatamifard, M. Nasrollahzadeh, and J. Lipkowski (2015). RSC Adv. 5, 91372.
J. Angelo, P. Magalhães, L. Andrade, and A. Mendes (2016). Appl. Surf. Sci. 387, 183.
T. K. Maji, D. Bagchi, P. Kar, D. Karmakar, and S. K. Pal (2017). J. Photochem. Photobiol. A Chem. 332, 391.
B. Li, H. Yuan, P. Yang, B. Yi, Y. Zhong, and Z. Hu (2016). Ceram. Int. 42, 17405.
P. Zhou, Z. Le, Y. Xie, J. Fang, and J. Xu (2017). J. Alloy. Compd. 692, 170.
Q. Cheng, W. Shi, and L. Duan (2015). B. sun, X. Li, A. Xu. J. Rare Earths 33, 249.
A. A. Ansari, A. Kaushik, P. R. Solanki, and B. D. Malhotra (2008). Electrochem. Commun. 10, 1246.
S. A. Ansari, M. M. Khan, M. O. Ansari, S. Kalathil, J. Lee, and M. H. Cho (2014). RSC Adv. 4, 16782.
I. Celardo, J. Z. Pedersen, E. Traversa, and L. Ghibelli (2011). Nanoscale 3, 1411.
S. Gangopadhyay, D. D. Frolov, A. E. Masunov, and S. Seal (2014). J. Alloys. Compd. 584, 199.
N. S. Arul, D. Mangalaraj, and T. W. Kim (2015). Appl. Surf. Sci. 349, 459.
H. I. Chen and H. Y. Chang (2004). Colloids. Surf. A. 242, 61.
Q. Dai, S. Bai, H. Li, W. Liu, X. Wang, and G. Lu (2014). Cryst. Eng. Commun. 16, 9817.
D. Zhang, X. Du, L. Shia, and R. Gao (2012). Dalton Trans. 41, 14455.
M. Mureseanu, V. Parvulescu, T. Radu, M. Filip, and G. Carja (2015). J. Alloys. Compd. 648, 864.
E. K. Goharshadi, S. Samiee, and P. Nancarrow (2011). J. Colloid Interface Sci. 356, 473.
R. J. Tayade, R. G. Kulkarni, and R. V. Jasra (2007). Ind. Eng. Chem. Res. 46, 369.
L. Y. Ji and C. Wei (2011). Catal. Sci. Technol. 1, 802.
H. B. Hadjltaief, M. B. Zina, M. E. Galvez, and P. D. Costa (2016). J. Photochem. Photobiol. A 315, 25.
B. Neppolian, S. Mine, Y. Horiuchi, C. L. Bianchi, M. Matsuoka, D. D. Dionysiou, and M. Anpo (2016). Chemosphere 153, 237.
K. Zhou, X. Y. Hu, B. Y. Chen, C. C. Hsueh, Q. Zhang, J. Wang, Y. J. Lin, and C. T. Chang (2016). Appl. Surf. Sci. 383, 300.
W. Zhang, X. Xiao, L. Zheng, and C. Wan (2015). Appl. Surf. Sci. 358, 468.
M. N. Chong, Z. Y. Tneu, P. E. Poh, B. Jin, and R. Aryal (2015). J. Taiwan Inst. Chem. Eng. 50, 288.
C. L. Marchena, L. Lerici, S. Renzini, L. Pierella, and L. Pizzio (2016). Appl. Catal. B 188, 23.
P. Latha, R. Dhanabackialakshmi, P. Senthil Kumar, and S. Karuthapandian (2016). Sep. Purif. Technol. 168, 124.
K. Guesh, C. M. Álvarez, and Y. Chebude (2016). Appl. Surf. Sci. 378, 473.
I. D. D. Kanakaraju, J. Kockler, C. A. Motti, B. D. Glass, and M. Oelgemöller (2015). Appl. Catal. B 166–167, 45.
X. Xing, P. H. Chang, G. Lv, W. T. Jiang, J. S. Jean, L. Liao, and Z. Li (2016). J. Taiwan Inst. Chem. Eng. 59, 237.
M. Lafjah, F. Djafri, A. Bengueddach, N. Keller, and V. Keller (2011). J. Hazard. Mater. 186, 1218.
A. N. Okte and O. Yılmaz (2009). Micropor. Mesopor. Mater. 126, 245.
P. Wang, K. Ueno, H. Takigawa, and K. Kobiro (2013). J. of Supercrit. Fluids. 78, 124.
A. Corma and H. Garcia (2004). Chem. Commun. doi:10.1039/B400147H.
C. T. Chang, J. J. Wang, T. Ouyangb, Q. Zhang, and Y. H. Jing (2015). Mater. Sci. Eng. B 196, 53.
J. Xiao, X. Wang, M. Fujii, Q. Yang, and C. Song (2013). AIChE 59, 1441.
G. Sudha and E. Subramanian (2015). J. Adv. Chem. Sci. 1, (3), 117–120.
J. K. Reddy, G. Suresh, C. H. Hymavathi, V. D. Kumari, and M. Subrahmanyam (2009). Catal. Today 141, 89.
T. Cai, Y. Liao, Z. Peng, Y. Long, Z. Wei, and Q. Deng (2009). J. Environ. Sci. 21, 997.
L. Wang, J. Ding, Y. Chai, Q. Liu, J. Ren, X. Liu, and W. L. Dai (2015). Dalton Trans. 44, 11223.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Latha, P., Karuthapandian, S. Novel, Facile and Swift Technique for Synthesis of CeO2 Nanocubes Immobilized on Zeolite for Removal of CR and MO Dye. J Clust Sci 28, 3265–3280 (2017). https://doi.org/10.1007/s10876-017-1292-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-017-1292-z