Abstract
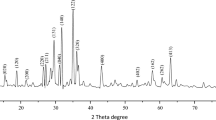
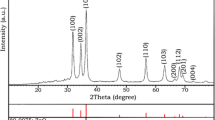
In this study, ZnO nanoparticles were synthesized using metal acetate, egg white as bio-template in water under microwave irradiation followed by calcination at 700 °C. The obtained ZnO nanoparticles with hexagonal wurtzite structure were characterized by XRD, SEM, TEM, TGA/DSC, FT-IR techniques. The photocatalytic activity of ZnO nanoparticles was evaluated by photocatalytic degradation of Congo red as organic dye pollutant under UV-light irradiation within 40 min. It was found that, the kinetic of photocatalytic behaviour of ZnO is first order. The stability and reusability of the prepared catalyst was considerable.








Similar content being viewed by others
References
C. Galindo, P. Jacques, and A. Kalt (2001). Chemosphere 45, 997–1005.
J.-M. Herrmann, M. Vautier, and C. Guillard (2001). J. Catal. 201, 46–59.
Z. Karim, A. P. Mathew, M. Grahn, J. Mouzon, and K. Oksman (2014). Carbohydr. Polym. 112, 668–676.
K. L. Yeap, T. T. Teng, B. T. Poh, N. Morad, and K. E. Lee (2014). Chem. Eng. J. 243, 305–314.
A. A. P. Mansur, H. S. Mansur, F. P. Ramanery, L. C. Oliveira, and P. P. Souza (2014). Appl. Catal. B 158–159, 269–279.
S. Wijannarong, S. Aroonsrimorakot, P. Thavipoke, A. Kumsopa, and S. Sangjan (2013). APCBEE Procedia. 5, 279–282.
Y. Zheng, G. Yao, Q. Cheng, S. Yu, M. Liu, and C. Gao (2013). Desalination 328, 42–50.
S. M. de A. G. U. de Souza, K. A. S. Bonilla, and A. A. U. de Souza (2010). J. Hazard. Mater. 179, 35–42.
P. Banerjee, S. DasGupta, and S. De (2007). J. Hazard. Mater. 140, 95–103.
C. Biswas, Z. Ma, X. Zhu, T. Kawaharamura, and K. L. Wang (2016). Sol. Energy Mater. Sol. Cells. 157, 1048–1056.
J. Wang, Y. Xia, Y. Dong, R. Chen, L. Xiang, and S. Komarneni (2016). Appl. Catal. B 192, 8–16.
Y. Zeng, Y. Zhao, and Y. Jiang (2016). J. Alloys Compd. 671, 579–584.
V. L. Patil, S. A. Vanalakar, P. S. Patil, and J. H. Kim (2017). Sens. Actuators B 239, 1185–1193.
B. E. Filali, T. V. Torchynska, G. Polupan, and L. Shcherbyna (2017). Mater. Res. Bull. 85, 161–167.
B. Astinchap, R. Moradian, and M. Nasseri Tekyeh (2016). Optik. 127, 9871–9877.
L. I. Ali, S. A. El-Molla, M. M. Ibrahim, H. R. Mahmoud, and M. A. Naghmash (2016). Opt. Mater. 58, 484–490.
A. Phuruangrat, T. Thongtem, and S. Thongtem (2014). Ceram. Int. 40, 9069–9076.
Y. Zhou, C. Liu, X. Zhong, H. Wu, M. Li, and L. Wang (2014). Ceram. Int. 40, 10415–10421.
Y. Xua, J. Jin, X. Li, Y. Han, H. Meng, T. Wang, and X. Zhang (2016). Mater. Res. Bull. 76, 235–239.
K. N. Abbas, N. Bidin, and R. S. Sabry (2016). Ceram. Int. 42, 13535–13546.
R. Pandimurugan and S. Thambidurai (2016). Adv. Powder Technol. 27, 1062–1072.
S. Maensiri, C. Masingboon, P. Lackal, W. Jareonboon, V. Promarak, P. L. Anderson, and S. Seraphin (2007). Cryst. Growth Des. 7, 950–955.
J. Li, X. Zhao, C. Yan, and L. Yijing (2008). Mater. Chem. Phys. 107, 177–182.
Q. Li, V. Kumar, Y. Li, H. Zhang, T. J. Marks, and R. P. H. Chang (2005). Chem. Mater. 17, 1001–1006.
F. Nouroozi and F. Farzaneh (2011). J. Braz. Chem. Soc. 22, 484–488.
J. J. Wu and S. C. Liu (2002). Adv. Mater. 14, 215–218.
K. Vanheusden, W. L. Warren, C. H. Seager, D. R. Tallant, J. A. Voigt, and B. E. Gnade (1996). J. Appl. Phys. 79, 7983–7990.
P. Yang, H. Yan, S. Mao, R. Russo, J. Johnson, R. Saykally, N. Morris, J. Pham, R. He, and H. J. Choi (2002). J. Adv. Funct. Mater. 12, 323–331.
P. Jiang, J. J. Zhou, H. F. Fang, C. Y. Wang, Z. L. Wang, and S. S. Xie (2007). Adv. Funct. Mater. 17, 1303–1310.
J. B. De Heredia, J. Torregrosa, J. R. Dominguez, and J. A. Peres (2001). J. Hazard. Mater. 83, 255–264.
C. S. Chiou, J. L. Shie, C. Y. Chang, C. C. Liu, and C. T. Chang (2006). J. Hazard. Mater. 137, 1123–1129.
J. Liqiang, Q. Yichun, W. Baiqi, L. Shudan, J. Baojiang, Y. Libin, F. Wei, F. Honggang, and S. Jiazhong (2006). Sol. Energy. Mater. Sol. Cells. 90, 1773–1787.
M. Pera-Titus, V. G. Molina, M. A. Banos, J. Gimenz, and S. Esplugas (2004). Appl. Catal. B Environ. 47, 219–256.
Z. Zhu, D. Yang, and H. Liu (2011). Adv. Powder Technol. 22, 493–497.
Acknowledgements
The financial support from the Alzahra University is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farzaneh, F., Asgharpour, Z., Nouroozi, F. et al. Rapid Synthesis and Characterization of Zinc Oxide Nanoparticles with Albumen as Photodegradation of Congo Red Under Microwave Irradiation. J Clust Sci 28, 1637–1646 (2017). https://doi.org/10.1007/s10876-017-1175-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-017-1175-3