Abstract
Patients with Down syndrome (DS) are characterized by increased susceptibility to autoimmunity and respiratory tract infections that are suggestive of humoral immunity impairment. Here, we sought to determine the follicular helper (Tfh) and follicular regulatory (Tfr) T cell profile in the blood of children with DS. Blood was collected from 24 children with DS, nine of which had autoimmune diseases. Children with DS showed skewed Tfh differentiation towards the CXCR3+ phenotype: Tfh1 and Tfh1/17 subsets were increased, while Tfh2 and Tfh17 subsets were reduced. While no differences in the percentage of Tfr cells were seen, the ratio of Tfh1 and CXCR3+PD-1+ subsets to Tfr cells was significantly increased in the affected children. The excessive polarization towards a CXCR3+ phenotype in children with DS suggests that re-calibration of Tfh subset skewing could potentially offer new therapeutic opportunities for these patients.



Similar content being viewed by others
Abbreviations
- DS:
-
Down syndrome
- AI:
-
Autoimmune
- Tfh:
-
Follicular T helper cell
- Tfr:
-
Follicular T regulatory cell
- Treg:
-
Regulatory T cell
- GC:
-
Germinal center
- FOXP3:
-
Forkhead box P3
- CCR6:
-
Chemokine receptor 6
- CVID:
-
Common variable immunodeficiency
- CXCL13:
-
CXC motif chemokine ligand 13
- CXCR3:
-
C-X-C motif chemokine receptor 3
- CXCR5:
-
C-X-C motif chemokine receptor 5
References
Kusters MAA, Verstegen RHJ, Gemen EFA, de Vries E. Intrinsic defect of the immune system in children with Down syndrome: a review. Clin Exp Immunol. 2009;156:189–93.
Kusters MA, Verstegen RH, de Vries E. Down syndrome: is it really characterized by precocious immunosenescence? Aging Dis. 2011;2:538–45.
Kusters MAA, Gemen EFA, Verstegen RHJ, Wever PC, de Vries E. Both normal memory counts and decreased naive cells favor intrinsic defect over early senescence of Down syndrome T lymphocytes. Pediatr Res. 2010;67:557–62.
Bloemers BLP, van Furth AM, Weijerman ME, Gemke RJBJ, Broers CJM, van den Ende K, et al. Down syndrome: a novel risk factor for respiratory syncytial virus bronchiolitis a prospective birth-cohort study. PEDIATRICS. 2007;120:e1076–81.
Pierce MJ, LaFranchi SH, Pinter JD. Characterization of thyroid abnormalities in a large cohort of children with Down syndrome. Horm Res Paediatr. 2017;87:170–8.
Mårild K, Stephansson O, Grahnquist L, Cnattingius S, Söderman G, Ludvigsson JF. Down syndrome is associated with elevated risk of celiac disease: a nationwide case-control study. J Pediatr. 2013;163:237–42.
de Hingh YCM, van der Vossen PW, Gemen EFA, Mulder AB, Hop WCJ, Brus F, et al. Intrinsic abnormalities of lymphocyte counts in children with Down syndrome. J Pediatr. 2005;147:744–7.
Schoch J, Rohrer TR, Kaestner M, Abdul-Khaliq H, Sester U, Sester M, et al. Quantitative, phenotypical and functional characterization of cellular immunity in pediatric individuals with Down syndrome. J Infect Dis. 2017;215:1619–38.
Ram G, Chinen J. Infections and immunodeficiency in Down syndrome: immunodeficiency in Down syndrome. Clin Exp Immunol. 2011;164:9–16.
Verstegen RHJ, Kusters MAA, Gemen EFA, De Vries E. Down syndrome B-lymphocyte subpopulations, intrinsic defect or decreased T-lymphocyte help. Pediatr Res. 2010;67:563–9.
Carsetti R, Valentini D, Marcellini V, Scarsella M, Marasco E, Giustini F, et al. Reduced numbers of switched memory B cells with high terminal differentiation potential in Down syndrome: clinical immunology. Eur J Immunol. 2015;45:903–14.
Valentini D, Marcellini V, Bianchi S, Villani A, Facchini M, Donatelli I, et al. Generation of switched memory B cells in response to vaccination in Down syndrome children and their siblings. Vaccine. 2015;33:6689–96.
Breitfeld D, Ohl L, Kremmer E, Ellwart J, Sallusto F, Lipp M, et al. Follicular B helper T cells express cxc chemokine receptor 5, localize to B cell follicles, and support immunoglobulin production. J Exp Med. 2000;192:1545–52.
Crotty S. T follicular helper cell differentiation, function, and roles in disease. Immunity. 2014;41:529–42.
Crotty S. Follicular helper CD4 T cells (T FH ). Annu Rev Immunol. 2011;29:621–63.
Kim CH, Rott LS, Clark-Lewis I, Campbell DJ, Wu L, Butcher EC. Subspecialization of CXCR5+ T cells: B helper activity is focused in a germinal center–localized subset of CXCR5+ T cells. J Exp Med. 2001;193:1373–81.
Ansel KM, McHeyzer-Williams LJ, Ngo VN, McHeyzer-Williams MG, Cyster JG. In vivo–activated Cd4 T cells upregulate cxc chemokine receptor 5 and reprogram their response to lymphoid chemokines. J Exp Med. 1999;190:1123–34.
Morita R, Schmitt N, Bentebibel S-E, Ranganathan R, Bourdery L, Zurawski G, et al. Human blood CXCR5+CD4+ T cells are counterparts of T follicular cells and contain specific subsets that differentially support antibody secretion. Immunity. 2011;34:108–21.
Schmitt N, Bentebibel S-E, Ueno H. Phenotype and functions of memory Tfh cells in human blood. Trends Immunol. 2014;35:436–42.
Locci M, Havenar-Daughton C, Landais E, Wu J, Kroenke MA, Arlehamn CL, et al. Human circulating PD-1+CXCR3−CXCR5+ memory Tfh cells are highly functional and correlate with broadly neutralizing HIV antibody responses. Immunity. 2013;39:758–69.
Bentebibel S-E, Lopez S, Obermoser G, Schmitt N, Mueller C, Harrod C, et al. Induction of ICOS+CXCR3+CXCR5+ TH cells correlates with antibody responses to influenza vaccination. Sci Transl Med. 2013;5:176ra32.
Obeng-Adjei N, Portugal S, Tran TM, Yazew TB, Skinner J, Li S, et al. Circulating Th1-cell-type Tfh cells that exhibit impaired B cell help are preferentially activated during acute malaria in children. Cell Rep. 2015;13:425–39.
Bowyer G, Grobbelaar A, Rampling T, Venkatraman N, Morelle D, Ballou RW, et al. CXCR3+ T follicular helper cells induced by co-administration of RTS,S/AS01B and viral-vectored vaccines are associated with reduced immunogenicity and efficacy against malaria. Front Immunol. 2018;9:1660.
Shi J, Hou S, Fang Q, Liu X, Liu X, Qi H. PD-1 controls follicular T helper cell positioning and function. Immunity. 2018;49:264–74 e4.
Craft JE. Follicular helper T cells in immunity and systemic autoimmunity. Nat Rev Rheumatol. 2012;8:337–47.
Le Coz C, Joublin A, Pasquali J-L, Korganow A-S, Dumortier H, Monneaux F. Circulating TFH Subset distribution is strongly affected in lupus patients with an active disease. Bayry J, editor. PLoS ONE. 2013;8:e75319.
Li X, Wu Z, Ding J, Zheng Z, Li X, Chen L, et al. Role of the frequency of blood CD4+ CXCR5+ CCR6+ T cells in autoimmunity in patients with Sjögren’s syndrome. Biochem Biophys Res Commun. 2012;422:238–44.
Unger S, Seidl M, van Schouwenburg P, Rakhmanov M, Bulashevska A, Frede N, et al. The T H 1 phenotype of follicular helper T cells indicates an IFN-γ–associated immune dysregulation in patients with CD21low common variable immunodeficiency. J Allergy Clin Immunol. 2018;141:730–40.
Ma CS, Wong N, Rao G, Avery DT, Torpy J, Hambridge T, et al. Monogenic mutations differentially affect the quantity and quality of T follicular helper cells in patients with human primary immunodeficiencies. J Allergy Clin Immunol. 2015;136:993–1006 e1.
Gerosa J, Lougaris V, Baronio M, Plebani A, Cicalese MP, Fousteri G. Beta2 integrins are required for follicular helper T cell differentiation in humans. Clin Immunol. 2017;180:60–2.
Cicalese MP, Gerosa J, Baronio M, Montin D, Licciardi F, Soresina A, et al. Circulating follicular helper and follicular regulatory T cells are severely compromised in human CD40 deficiency: a case report. Front Immunol. 2018;9:1761.
Linterman MA, Hill DL. Can follicular helper T cells be targeted to improve vaccine efficacy? F1000Research. 2016;5:88.
Farroni C, Marasco E, Marcellini V, Giorda E, Valentini D, Petrini S, et al. Dysregulated miR-155 and miR-125b are related to impaired B-cell responses in Down syndrome. Front Immunol. 2018;9:2683.
Fonseca VR, Agua-Doce A, Maceiras AR, Pierson W, Ribeiro F, Romão VC, et al. Human blood T fr cells are indicators of ongoing humoral activity not fully licensed with suppressive function. Sci Immunol. 2017;2:eaan1487.
Shan Y, Qi C, Zhao J, Liu Y, Gao H, Zhao D, et al. Higher frequency of peripheral blood follicular regulatory T cells in patients with new onset ankylosing spondylitis. Clin Exp Pharmacol Physiol. 2015;42:154–61.
Chung Y, Tanaka S, Chu F, Nurieva RI, Martinez GJ, Rawal S, et al. Follicular regulatory T cells expressing Foxp3 and Bcl-6 suppress germinal center reactions. Nat Med. 2011;17:983–8.
Shearer WT, Rosenblatt HM, Gelman RS, Oyomopito R, Plaeger S, Stiehm ER, et al. Lymphocyte subsets in healthy children from birth through 18 years of age: the pediatric AIDS Clinical Trials Group P1009 study. Basic Clin Immunol. 2003;112:8.
Italian primary immunodeficiencies strategic scientific committee. Transient hypogammaglobulinaemia of infancy: Recommendations for Diagnosis and Treatment. 2004;
Miles J, Riches P. The determination of IgG subclass concentrations in serum by enzyme-linked immunosorbent assay: establishment of age-related reference ranges for cord blood samples, children aged 5-13 years and adults. Ann Clin Biochem. 1994;31:245–8.
Lima FA, Moreira-Filho CA, Ramos PL, Brentani H, Lima LDA, Arrais M, et al. Decreased AIRE expression and global thymic hypofunction in Down syndrome. J Immunol. 2011;187:3422–30.
Choubey D, Moudgil KD. Interferons in autoimmune and inflammatory diseases: regulation and roles. J Interf Cytokine Res. 2011;31:857–65.
Waugh KA, Araya P, Pandey A, Jordan KR, Smith KP, Granrath RE, et al. Mass cytometry reveals global immune remodeling with multi-lineage hypersensitivity to type I interferon in Down syndrome. Cell Rep. 2019;29:1893–908 e4.
Pallikkuth S, Parmigiani A, Silva SY, George VK, Fischl M, Pahwa R, et al. Impaired peripheral blood T-follicular helper cell function in HIV-infected nonresponders to the 2009 H1N1/09 vaccine. Blood. 2012;120:985–93.
Long S, Ma L, Wang D, Shang X. High frequency of circulating follicular helper T cells is correlated with B cell subtypes in patients with ankylosing spondylitis. ExpTher Med. 2018;15:4578–86.
Coraglia A, Galassi N, Fernández Romero DS, Juri MC, Felippo M, Malbrán A, et al. Common variable immunodeficiency and circulating T FH. J Immunol Res. 2016;2016:1–10.
Vaziri H, Uchida I, Cohen D, Harley CB. Loss of telomeric DNA during aging of Normal and trisomy 21 human lymphocytes. Am J Hum Genet. 1993;52:661–7.
Koutsakos M, Nguyen THO, Kedzierska K. With a little help from T follicular helper friends: humoral immunity to influenza vaccination. J Immunol. 2019;202:360–7.
Avanzini MA, Monafo V, De Amici M, Burgio GR, Plebani A, Ugazio AG, et al. Letter to the editor: Humoral immunodeficiencies in Down syndrome: serum IgG subclass and antibody response to hepatitis B vaccine. Am J Med Genet. 2005;37:231–3.
Joshi AY, Abraham RS, Snyder MR, Boyce TG. Immune evaluation and vaccine responses in Down syndrome: evidence of immunodeficiency? Vaccine. 2011;29:5040–6.
Acknowledgments
We thank patients and parents for their participation in this study.
Funding
This work was supported with funds from OSR Pilot & Seed Grant to MPC and GF and the Italian Ministry of Health Young Researchers Award (GR-2011-02348732) to GF.
Author information
Authors and Affiliations
Contributions
JG, PDL, AV, and TJ performed experiments and analyzed data. MS and MM provided samples, analyzed data, and corrected the manuscript. MA, CT, MDP, and AA provided samples and corrected the manuscript. GO, MPC, and GF analyzed data and wrote the manuscript. All authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Fig. S1
Immunoglobulin levels and lymphocyte counts in children with Down syndrome. Panel a represents the distribution of IgG (A), IgM (B) and IgA (C) values in DS children (n = 24) according to age. Dashed lines represent +2SD and -2SD normal range limits for age. Panel b shows the comparison of the total CD3+CD4+ between DS children (n = 24) and HD (n = 30). Panel c shows the distribution of CD19+ B cells in DS children according to age. Dashed line represents +2 standard deviation (SD) and -2SD from normal range limit for age [37, 38]. (PPTX 89 kb)
Fig. S2
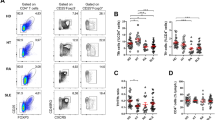
Gating strategy for Tfh (CXCR5+FOXP3−), Tfr (CXCR5+FOXP3+), CXCR5− Treg (CXCR5−FOXP3+). Arrows indicate gating strategy: singlets → lymphocytes → CD3+CD19−CD14−CD8− → CD3+CD4+. (PPTX 124 kb)
Fig. S3
Gating strategy for Tfh1 (CCR6−CXCR3+), Tfh2 (CCR6−CXCR3−), Tfh17 (CCR6+CXCR3−), Tfh1/17 (CCR6+CXCR3+) and CXCR3-PD-1+ and CXCR3+PD-1+. Arrows indicate gating strategy: singlets → lymphocytes →CD3+CD4+ → CD4+CXCR5+. (PPTX 123 kb)
Fig. S4
Tfh subset and Tfr cell ratios in DS children. Panel a shows (Tfh2 + Tfh17):Tfh1 ratio in HD and DS children. b. Ratios Tfh1:Tfr and Tfh1/17:Tfr in HD and DS children. Each dot represents one patient. Asterisks indicate statistical significance calculated by Mann Whitney test. **p < 0.01; ****p < 0.0001 (PPTX 64 kb)
Fig. S5
Tfh subsets in DS children with or without autoimmunity. Panel a shows the frequencies of Tfh1, Tfh2, Tfh17, Tfh1/17 in DS children without autoimmunity (n = 15) and DS children with autoimmunity (n = 9) compared to age-matched HD (n = 30). b. (Tfh2 + Tfh17):Tfh1 ratio in HD and DS children with or without autoimmunity. c. Ratios Tfh1:Tfr and Tfh1/17:Tfr in HD and DS children with or without autoimmunity. Each dot represents one patient. Asterisks indicate statistical significance calculated by Mann Whitney test. *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001. (PPTX 122 kb)
Fig. S6
The proportions of CXCR3−PD-1+ and CXCR3+PD-1+ Tfh cells in DS children with or without autoimmunity. Panel a shows the percentage of CXCR3−PD-1+ and CXCR3+PD-1+ in peripheral blood of DS children without autoimmunity (n = 15) and DS children with autoimmunity (n = 9) compared to age-matched HD. Panel b shows ratio CXCR3+PD1+: Tfr in DS children with or without autoimmunity as compared to age-matched HD. Panel c represents CXCL13 levels in the plasma of DS children with autoimmunity vs. DS children without autoimmunity and age-matched HD. Each dot represents one patient. Asterisks indicate statistical significance as calculated by Mann Whitney test. *p < 0.05; **p < 0.01. (PPTX 90 kb)
ESM 7
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Ottaviano, G., Gerosa, J., Santini, M. et al. A Prevalent CXCR3+ Phenotype of Circulating Follicular Helper T Cells Indicates Humoral Dysregulation in Children with Down Syndrome. J Clin Immunol 40, 447–455 (2020). https://doi.org/10.1007/s10875-020-00755-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-020-00755-0