Abstract
Background
Chronic granulomatous disease (CGD) is a primary immunodeficiency disease that is characterized by susceptibility to bacterial and fungal infections. CGD patients also suffer from immune regulatory disorders, such as CGD-associated bowel inflammation with granuloma, which could be caused by excessive inflammation without demonstrable infection.
Purpose
We investigated the clinical manifestation of interstitial lung disease (ILD) resulting from excessive inflammation in X-linked CGD patients.
Methods
Pulmonary CT images and testing of serum KL-6 levels were performed to assess ILD in the patients. For this study, patients with pulmonary lesions due to demonstrable infections were excluded from among ILD patients.
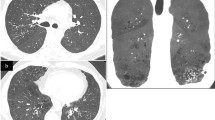
Results
Among 33 CGD patients, four developed ILD; they had increased reticulo-nodular opacities on CT images and elevated serum KL-6 levels. Histopathological examinations revealed multiple homogeneous microgranulomas in the lesions of inflammatory cell infiltration. Mononuclear cells obtained from their pulmonary lesions produced higher amounts of inflammatory cytokines than the peripheral blood mononuclear cells of CGD patients, suggesting that the only infiltrating cells in the pulmonary lesions were activated and produced large amounts of inflammatory cytokines in ILD patients. Interestingly, an anti-inflammatory drug, such as a corticosteroid or thalidomide, but not anti-bacterial or anti-fungal drugs, improved CT image findings and reduced their KL-6 levels.
Conclusions
CGD patients’ daily exposures to inhaled antigens may induce excessive reactions with the production of inflammatory cytokines leading to the development of ILD with multiple microgranulomas, which could be due to an inadequate production of reactive oxygen species in CGD.






Similar content being viewed by others
References
Kang EM, Marciano BE, DeRavin S, Zarember KA, Holland SM, Malech HL. Chronic granulomatous disease: overview and hematopoietic stem cell transplantation. J Allergy Clin Immunol. 2011;127(6):1319–26.
Kawai T, Kusakabe H, Seki A, Kobayashi S, Onodera M. Osteomyelitis due to trimethoprim/sulfamethoxazole-resistant Edwardsiella tarda infection in a patient with X-linked chronic granulomatous disease. Infection. 2011;39(2):171–3.
Kobayashi S, Murayama S, Takanashi S, Takahashi K, Miyatsuka S, Fujita T, et al. Clinical features and prognoses of 23 patients with chronic granulomatous disease followed for 21 years by a single hospital in Japan. Eur J Pediatr. 2008;167(12):1389–94.
Holland SM. Chronic granulomatous disease. Clin Rev Allergy Immunol. 2010;38(1):3–10.
De Ravin SS, Naumann N, Cowen EW, Friend J, Hilligoss D, Marquesen M, et al. Chronic granulomatous disease as a risk factor for autoimmune disease. J Allergy Clin Immunol. 2008;122(6):1097–103.
van de Veerdonk FL, Smeekens SP, Joosten LA, Kullberg BJ, Dinarello CA, van der Meer JW, et al. Reactive oxygen species-independent activation of the IL-1beta inflammasome in cells from patients with chronic granulomatous disease. Proc Natl Acad Sci U S A. 2010;107(7):3030–3.
Kawai T, Watanabe N, Yokoyama M, Arai K, Oana S, Harayama S, et al. Thalidomide attenuates excessive inflammation without interrupting lipopolysaccharide-driven inflammatory cytokine production in chronic granulomatous disease. Clin Immunol. 2013;147(2):122–8.
Fernandez-Boyanapalli R, Frasch SC, Riches DW, Vandivier RW, Henson PM, Bratton DL. PPARgamma activation normalizes resolution of acute sterile inflammation in murine chronic granulomatous disease. Blood. 2010;116(22):4512–22.
Seger RA. Modern management of chronic granulomatous disease. Br J Haematol. 2008;140(3):255–66.
Uzel G, Orange JS, Poliak N, Marciano BE, Heller T, Holland SM. Complications of tumor necrosis factor-alpha blockade in chronic granulomatous disease-related colitis. Clin Infect Dis Off Publ Infect Dis Soc Am. 2010;51(12):1429–34.
Kohno N, Kyoizumi S, Awaya Y, Fukuhara H, Yamakido M, Akiyama M. New serum indicator of interstitial pneumonitis activity. Sialylated carbohydrate antigen KL-6. Chest. 1989;96(1):68–73.
Senn L, Robinson JO, Schmidt S, Knaup M, Asahi N, Satomura S, et al. 1,3-Beta-D-glucan antigenemia for early diagnosis of invasive fungal infections in neutropenic patients with acute leukemia. Clin Infect Dis. 2008;46(6):878–85.
Persat F, Ranque S, Derouin F, Michel-Nguyen A, Picot S, Sulahian A. Contribution of the (1–>3)-beta-D-glucan assay for diagnosis of invasive fungal infections. J Clin Microbiol. 2008;46(3):1009–13.
Meissner F, Seger RA, Moshous D, Fischer A, Reichenbach J, Zychlinsky A. Inflammasome activation in NADPH oxidase defective mononuclear phagocytes from patients with chronic granulomatous disease. Blood. 2010;116(9):1570–3.
Bogaert P, Tournoy KG, Naessens T, Grooten J. Where asthma and hypersensitivity pneumonitis meet and differ: noneosinophilic severe asthma. Am J Pathol. 2009;174(1):3–13.
Khaldi H, Marchand-Adam S, Kannengiesser C, Fabre A, Debray MP, Danel C, et al. Diffuse interstitial pneumonia and pulmonary hypertension: a novel manifestation of chronic granulomatous disease. Eur Respir J. 2009;33(6):1498–502.
Shah A. Aspergillus-associated hypersensitivity respiratory disorders. Indian J Chest Dis Allied Sci. 2008;50(1):117–28.
Van Hoeyveld E, Dupont L, Bossuyt X. Quantification of IgG antibodies to Aspergillus fumigatus and pigeon antigens by ImmunoCAP technology: an alternative to the precipitation technique? Clin Chem. 2006;52(9):1785–93.
Lacasse Y, Selman M, Costabel U, Dalphin JC, Morell F, Erkinjuntti-Pekkanen R, et al. Classification of hypersensitivity pneumonitis: a hypothesis. Int Arch Allergy Immunol. 2009;149(2):161–6.
Buchvald F, Petersen BL, Damgaard K, Deterding R, Langston C, Fan LL, et al. Frequency, treatment, and functional outcome in children with hypersensitivity pneumonitis. Pediatr Pulmonol. 2011;46(11):1098–107.
Dostert C, Petrilli V, Van Bruggen R, Steele C, Mossman BT, Tschopp J. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science. 2008;320(5876):674–7.
Segal BH, Han W, Bushey JJ, Joo M, Bhatti Z, Feminella J, et al. NADPH oxidase limits innate immune responses in the lungs in mice. PLoS One. 2010;5(3):e9631.
Takeuchi O, Akira S. Toll-like receptors; their physiological role and signal transduction system. Int Immunopharmacol. 2001;1(4):625–35.
Olsson S, Sundler R. The macrophage beta-glucan receptor mediates arachidonate release induced by zymosan: essential role for Src family kinases. Mol Immunol. 2007;44(7):1509–15.
Marciano BE, Rosenzweig SD, Kleiner DE, Anderson VL, Darnell DN, Anaya-O’Brien S, et al. Gastrointestinal involvement in chronic granulomatous disease. Pediatrics. 2004;114(2):462–8.
Palumbo A, Facon T, Sonneveld P, Blade J, Offidani M, Gay F, et al. Thalidomide for treatment of multiple myeloma: 10 years later. Blood. 2008;111(8):3968–77.
Meierhofer C, Wiedermann CJ. New insights into the pharmacological and toxicological effects of thalidomide. Curr Opin Drug Discov Dev. 2003;6(1):92–9.
Marks DJ, Miyagi K, Rahman FZ, Novelli M, Bloom SL, Segal AW. Inflammatory bowel disease in CGD reproduces the clinicopathological features of Crohn’s disease. Am J Gastroenterol. 2009;104(1):117–24.
Keller M, Sollberger G, Beer HD. Thalidomide inhibits activation of caspase-1. J Immunol. 2009;183(9):5593–9.
Eski M, Sahin I, Sengezer M, Serdar M, Ifran A. Thalidomide decreases the plasma levels of IL-1 and TNF following burn injury: is it a new drug for modulation of systemic inflammatory response. Burns. 2008;34(1):104–8.
Yasui K, Yashiro M, Tsuge M, Manki A, Takemoto K, Yamamoto M, et al. Thalidomide dramatically improves the symptoms of early-onset sarcoidosis/Blau syndrome: its possible action and mechanism. Arthritis Rheum. 2010;62(1):250–7.
Acknowledgments
We are grateful to the CGD patients and blood donors for their participation in this study. The manuscript was proofread and edited by Dr. Julian Tang of the Department of Clinical Research Education, National Center for Child Health and Development. This study was supported by a Grant from the National Center for Child Health and Development.
Conflict of Interest
The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kawai, T., Watanabe, N., Yokoyama, M. et al. Interstitial Lung Disease with Multiple Microgranulomas in Chronic Granulomatous Disease. J Clin Immunol 34, 933–940 (2014). https://doi.org/10.1007/s10875-014-0089-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-014-0089-1