Abstract
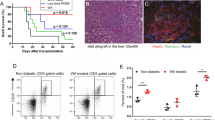
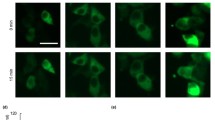
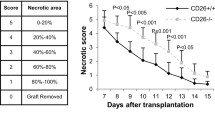
Core 1 beta 1,3-galactosyltransferase also known as T-antigen-synthase or T-synthase is a key enzyme for the synthesis of the common core 1 O-glycan structure (T-antigen). Although T-synthase is known to be important in human immune-related diseases, the effects of T-synthase and T-antigen on host immune responses remain poorly defined. In this study, a T-synthase-specific short hairpin RNA (shRNA) was transfected into murine colon carcinoma CT26 cells or mouse muscle tissues via intramuscular electroporation to assess the effects of T-synthase on T cells and cytokines. T-synthase knockdown significantly induced galectin-1 secretion both in vivo and in vitro and strongly enhanced Th2 cytokine (IL-10 and IL-4) production in vivo. Further, the increased production of galectin-1 induced by T-synthase knockdown promoted CD8+ T-cell apoptosis, which, when combined with the increased production of CD4+ T cell-derived Th2 cytokines prolonged the survival of skin allografts in mice. Our data suggest core 1 beta 1,3-galactosyltransferase-shRNA could serve not only as a useful tool in organ transplantation but also as a powerful tool for investigating O-glycans and glycoprotein synthesis and function.










Similar content being viewed by others
Abbreviations
- shRNA:
-
Short hairpin RNA
- T synthase or C1GalT:
-
Core 1 beta 1, 3-galactosyltransferase
- CsA:
-
Cyclosporin A
- T-antigen:
-
Thomsen-Friedenreich antigen
- MTT:
-
3- (4,5-cimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
- FCM:
-
Flow Cytometry
- PNA:
-
Peanut agglutinin
- PI:
-
Propidium iodide
- CFSE:
-
Carboxyfluorescein succinimidyl ester
- ECL:
-
Enhanced chemiluminescence
- ELISA:
-
Antigen-capture enzyme-linked immunosorbent assay
References
Halloran PF. Immunosuppressive drugs for kidney transplantation. N Engl J Med. 2004;351:2715–29.
Starzl TE, Murase N, Abu-Elmagd K, Gray EA, Shapiro R, Eghtesad B, et al. Tolerogenic immunosuppression for organ transplantation. Lancet. 2003;361:1502–10.
Nielsen FT, Ottosen P, Starklint H, Dieperink H. Kidney function and morphology after short-term combination therapy with cyclosporine A, tacrolimus and sirolimus in the rat. Nephrol Dial Transplant. 2003;18:491–6.
Peter-Katalinic J. Methods in enzymology: O-glycosylation of proteins. Methods Enzymol. 2005;405:139–71.
Ju T, Brewer K, D’Souza A, Cummings RD, Canfield WM. Cloning and expression of human core 1 beta1,3-galactosyltransferase. J Biol Chem. 2002;277:178–86.
Ju T, Cummings RD, Canfield WM. Purification, characterization, and subunit structure of rat core 1 Beta1,3-galactosyltransferase. J Biol Chem. 2002;277:169–77.
Ju T, Aryal RP, Stowell CJ, Cummings RD. Regulation of protein O-glycosylation by the endoplasmic reticulum-localized molecular chaperone Cosmc. J Cell Biol. 2008;182:531–42.
Itzkowitz SH, Bloom EJ, Lau TS, Kim YS. Mucin associated Tn and sialosyl-Tn antigen expression in colorectal polyps. Gut. 1992;33:518–23.
Conze T, Carvalho AS, Landegren U, Almeida R, Reis CA, David L, et al. MUC2 mucin is a major carrier of the cancer-associated sialyl-Tn antigen in intestinal metaplasia and gastric carcinomas. Glycobiology. 2010;20:199–206.
Freire T, Bay S, von Mensdorff-Pouilly S, Osinaga E. Molecular basis of incomplete O-glycan synthesis in MCF-7 breast cancer cells: putative role of MUC6 in Tn antigen expression. Cancer Res. 2005;65:7880–7.
Julian BA, Novak J. IgA nephropathy: an update. Curr Opin Nephrol Hypertens. 2004;13:171–9.
Xia L, Ju T, Westmuckett A, An G, Ivanciu L, McDaniel JM, et al. Defective angiogenesis and fatal embryonic hemorrhage in mice lacking core 1-derived O-glycans. J Cell Biol. 2004;164:451–9.
Xia L, McEver RP. Targeted disruption of the gene encoding core 1 beta1-3-galactosyltransferase (T-synthase) causes embryonic lethality and defective angiogenesis in mice. Methods Enzymol. 2006;416:314–31.
Van Dyken SJ, Green RS, Marth JD. Structural and mechanistic features of protein O glycosylation linked to CD8+ T-cell apoptosis. Mol Cell Biol. 2007;27:1096–111.
Valenzuela HF, Pace KE, Cabrera PV, White R, Porvari K, Kaija H, et al. O-glycosylation regulates LNCaP prostate cancer cell susceptibility to apoptosis induced by galectin-1. Cancer Res. 2007;67:6155–62.
Mathieu V, Le Mercier M, De Neve N, Sauvage S, Gras T, Roland I, et al. Galectin-1 knockdown increases sensitivity to temozolomide in a B16F10 mouse metastatic melanoma model. J Invest Dermatol. 2007;127:2399–410.
Liu M, Chen H, Luo F, Li P, Pan Q, Xia B, et al. Deletion of N-glycosylation sites of hepatitis C virus envelope protein E1 enhances specific cellular and humoral immune responses. Vaccine. 2007;25:6572–80.
Ma Y, Chen H, Wang Q, Luo F, Yan J, Zhang XL. IL-24 protects against Salmonella typhimurium infection by stimulating early neutrophil Th1 cytokine production, which in turn activates CD8+ T cells. Eur J Immunol. 2009;39:3357–68.
Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65:55–63.
Lowe JB, Marth JD. A genetic approach to Mammalian glycan function. Annu Rev Biochem. 2003;72:643–91.
Toscano MA, Bianco GA, Ilarregui JM, Croci DO, Correale J, Hernandez JD, et al. Differential glycosylation of TH1, TH2 and TH-17 effector cells selectively regulates susceptibility to cell death. Nat Immunol. 2007;8:825–34.
Rudd PM, Elliott T, Cresswell P, Wilson IA, Dwek RA. Glycosylation and the immune system. Science. 2001;291:2370–6.
Helenius A, Aebi M. Intracellular functions of N-linked glycans. Science. 2001;291:2364–9.
Daniels MA, Hogquist KA, Jameson SC. Sweet ‘n’ sour: the impact of differential glycosylation on T cell responses. Nat Immunol. 2002;3:903–10.
Zhang XL. Roles of glycans and glycopeptides in immune system and immune-related diseases. Curr Med Chem. 2006;13:1141–7.
Brockhausen I. The role of galactosyltransferases in cell surface functions and in the immune system. Drug News Perspect. 2006;19:401–9.
Rabinovich GA, Daly G, Dreja H, Tailor H, Riera CM, Hirabayashi J, et al. Recombinant galectin-1 and its genetic delivery suppress collagen-induced arthritis via T cell apoptosis. J Exp Med. 1999;190:385–98.
Santucci L, Fiorucci S, Rubinstein N, Mencarelli A, Palazzetti B, Federici B, et al. Galectin-1 suppresses experimental colitis in mice. Gastroenterology. 2003;124:1381–94.
Motran CC, Molinder KM, Liu SD, Poirier F, Miceli MC. Galectin-1 functions as a Th2 cytokine that selectively induces Th1 apoptosis and promotes Th2 function. Eur J Immunol. 2008;38:3015–27.
de Waal MR, Yssel H, de Vries JE. Direct effects of IL-10 on subsets of human CD4+ T cell clones and resting T cells. Specific inhibition of IL-2 production and proliferation. J Immunol. 1993;150:4754–65.
Sembeil R, Sanhadji K, Vivier G, Chargui J, Touraine JL. Prolonged survival of mouse skin allografts after transplantation of fetal liver cells transduced with hIL-10 gene. Transpl Immunol. 2004;13:1–8.
Comelli EM, Sutton-Smith M, Yan Q, Amado M, Panico M, Gilmartin T, et al. Activation of murine CD4+ and CD8+ T lymphocytes leads to dramatic remodeling of N-linked glycans. J Immunol. 2006;177:2431–40.
Van Dyken SJ, Green S, Marth JD. Structural and mechanistic features of protein O glycosylation linked to CD8+ T-cell apoptosis. Mol Cell Biol. 2007;27:1096–111.
Blasko A, Fajka-Boja R, Ion G, Monostori E. How does it act when soluble? Critical evaluation of mechanism of galectin-1 induced T-cell apoptosis. Acta Biologica Hungarica. 2011;62:106–11.
Ion G, Fajka-Boja R, Toth GK, Caron M, Monostori E. Role of p56lck and ZAP70-mediated tyrosine phosphorylation in galectin-1-induced cell death. Cell Death Differ. 2005;12:1145–7.
Matarrese P, Tinari A, Mormone E, Bianco GA, Toscano MA, Ascione B, et al. Galectin-1 sensitizes resting human T lymphocytes to Fas (CD95)-mediated cell death via mitochondrial hyperpolarization, budding, and fission. J Biol Chem. 2005;280:6969–85.
Perillo NL, Pace KE, Seilhamer JJ, Baum LG. Apoptosis of T cells mediated by galectin-1. Nature. 1995;378:736–9.
Pace KE. Cutting edge: CD7 delivers a pro-apoptotic signal during galectin-1-induced T cell. J Immunol. 2000;165:2331–4.
Vespa GN. Galectin-1 specifically modulates TCR signals to enhance TCR apoptosis but inhibit IL-2 production and proliferation. J Immunol. 1999;162:799–806.
Liu SD. Galectin-1 tunes TCR binding and signal transduction to regulate CD8 burst size. J Immunol. 2009;182:5283–95.
Stillman BN, Hsu DK, Pang M, Brewer CF, Johnson P, Liu FT, et al. Galectin-3 and galectin-1 bind distinct cell surface glycoprotein receptors to induce T cell death. J Immunol. 2006;176:778–89.
Xu G, Tu W, Xu C. Immunological tolerance induced by galectin-1 in rat allogeneic renal transplantation. Int Immunopharmacol. 2010;10:643–6.
Acknowledgments
The plasmid pPCR-hC1GalT was kindly provided by Dr. Narimatsu H and Dr. Ju T.
This work was supported by grants from the National Grand Program on Key Infectious Disease, National Natural Science Foundation of China (30921001, 81000714 and 30800038), and National Outstanding Youth Foundation of China (81025008), the 973 Program of China (2009CB522507), 211 program (303-581045), the program for Innovative Research Team of University, the Science and Technology Program of Wuhan (#201150530141) and DFG Transregio 60.
Competing Interests
The authors have dedared that no competing interests exist.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, HD., Zhou, X., Yu, G. et al. Knockdown of Core 1 Beta 1, 3-galactosyltransferase Prolongs Skin Allograft Survival with Induction of Galectin-1 Secretion and Suppression of CD8+T Cells. J Clin Immunol 32, 820–836 (2012). https://doi.org/10.1007/s10875-012-9653-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-012-9653-8