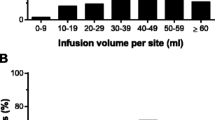
The present clinical study was designed to evaluate the efficacy, pharmacokinetics and safety of a new 10% liquid intravenous immune globulin in patients with primary immunodeficiency diseases. Sixty-one adults and children with primary immuno-deficiency diseases received doses of 300–600 mg/kg body weight every 21–28 days for 12 months. No validated acute serious bacterial infections were reported. The 95% confidence interval for the annualized rate of acute serious bacterial infections (primary endpoint) was 0–0.060. A total of four predefined validated other bacterial infections commonly occurring in primary immunodeficiency disease subjects were observed; none were serious, severe or resulted in hospitalization. The median elimination half-life of IgG was 35 days. Median total IgG trough levels varied from 9.6 to 11.2 g/L. Temporally associated adverse experiences were determined for 72 h after each infusion and the most common adverse experience was headache, which was associated with 6.9% of infusions. The study met the primary endpoint for efficacy and demonstrated excellent tolerability of the new 10% liquid intravenous imunoglobulin preparation.


Similar content being viewed by others
REFERENCES
Rosen FS, Cooper MD, Wedgwood RJ: The primary immunodeficiencies. N Engl J Med 333:431–440, 1995
IUIS Scientific Committee: Primary immunodeficiency diseases. Report of an IUIS Scientific Committee. International Union of Immunological Societies. Clin Exp Immunol 118:1–28, 1999
Cooper MD, Lanier LL, Conley ME, Puck JM: Immunodeficiency disorders. Hematology (Am Soc Hematol Educ Program):314–330, 2003
Schwartz SA: Intravenous immunoglobulin treatment of immunodeficiency disorders. Pediatr Clin North Am 47:1355–1369, 2000
Durandy A, Wahn V, Petteway S, Gelfand EW: Immunoglobulin replacement therapy in primary antibody deficiency diseases-maximizing success. Int Arch Allergy Immunol 136:217–229, 2005
World Health Organization Scientific Group: Primary Immunodeficiency Diseases. Clin Exp Immunol 99:2–24, 1995
Rosner B: One-Sample Inference for Incidence-Rate Data: Fundamentals of Biostatistics. Pacific Grove, CA, Duxbury Thompson Learning, 2000
Department of Health And Human Services, Food And Drug Administration, Center For Biologics Evaluation And Research: Blood Products Advisory Committee, 65th Meeting, Silver Spring, MD. Washington, Miller Reporting Company, Inc., March 2000
Lee ML, Poon WY, Kingdon HS: A two-phase linear regression model for biologic half-life data. J Lab Clin Med 115:745–748, 1990
Lee ML, Bray G, Mosher J, Gau J, Gomperts ED: An improved, robust method for the determination of coagulation factor half-life. Haemophilia 2 (Suppl 1):22, 1996
Ochs HD, Pinciaro PJ, The Octagam Study Group: Octagam® 5%, an intravenous IgG Product, is efficacious and well tolerated in subjects with primary immunodeficiency diseases. J Clin Immun 24:309–314, 2004
Berger M, Pinciaro PJ, Flebogamma 5% Investigators: Safety, efficacy, and pharmacokinetics of Flebogamma 5%. [immune globulin intravenous (human)] for replacement therapy in primary immunodeficiency diseases. J Clin Immunol 24(4):389–396, 2004
De Gracia J, Vendrell M, Álvarez A, Pallisa E, Rodrigo MJ, de la Rosa D, Mata F, Andreu J, Morell F: Immunoglobulin therapy to control lung damage in patients with common variable immunodeficiency. Int Immunopharmacol 4:745–753, 2004
Roifman CM, Schroeder H, Berger M, Sorensen R, Ballow M, Buckley RH, Gewurz A, Korenblat P, Sussman G, Lemm G: Comparison of the efficacy of IGIV-C, 10% (caprylate/chromatography) and IGIV-SD, 10% as replacement therapy in primary immune deficiency. A randomised double-blind trial. Int Immunopharmacol 3:1325–1333, 2003
Favre O, Leimgruber A, Nicole A, Spertini F: Intravenous immunoglobulin replacement prevents severe and lower respiratory tract infections, but not upper respiratory tract and non-respiratory infections in common variable immune deficiency. Allergy 60(3):385–390, 2005
Eijkhout HW, van Der Meer JW, Kallenberg CG, Weening RS, van Dissel JT, Sanders LA, Strengers PF, Nienhuis H, Schellekens PT, Inter-University Working Party for the Study of Immune Deficiencies: The effect of two different dosages of intravenous immunoglobulin on the incidence of recurrent infections in patients with primary hypogammaglobulinemia. A randomized, double-blind, multicenter crossover trial. Ann Intern Med 135:165–174, 2001.
Bayrakci B, Ersoy F, Sanal Ö, Kilic S, Metin A, Tezcan I: The efficacy of immunoglobulin replacement therapy in the long-term follow-up of the B-cell deficiencies (XLA, HIM, CVID). Turk J Pediatr 47:239–246, 2005
Pettit SJ, Bourne H, Spickett GP: Survey of infection in patients receiving antibody replacement treatment for immune deficiency. J Clin Pathol 55:577–580, 2002
Alyanakian MA, Bernatowska E, Scherrmann JM, Aucouturier P, Poplavsky JL: Pharmacokinetics of total immunoglobulin G and immunoglobulin G subclasses in patients undergoing replacement therapy for primary immunodeficiency syndromes. Vox Sang 84:188–192, 2003
Ballow M, Berger M, Bonilla FA, Buckley RH, Cunningham-Rundles CH, Fireman P, Kaliner M, Ochs HD, Skoda-Smith S, Sweetser MT, Taki H, Lathia C: Pharmacokinetics and tolerability of a new intravenous immunoglobulin preparation, IGIV-C, 10% (Gamunex, 10%). Vox Sang 84:202–210, 2003
Roifman CM, Gelfand EW: Replacement therapy with high dose intravenous gamma-globulin improves chronic sinopulmonary disease in patients with hypogammaglobulinemia. Pediatr Infect Dis J 7:S92–S96, 1988
Morell A: Pharmacokinetics of intravenous immunoglobulin preparations. In: Intravenous Immunoglobulins in Clinical Practice, ML Lee and V Strand (eds). New York, Basel, Hong Kong: Marcel Dekker, Inc., 1997, pp 1–18
Wolf HH, Davies SV, Borte M, Caulier MT, Williams PE, Bernuth HV, Egner W, Sklenar I, Adams C, Späth P, Morell A, Andresen I: Efficacy, tolerability, safety and pharmacokinetics of a nanofiltered intravenous immunoglobulin: Studies in patients with immune thrombocytopenic purpura and primary immunodeficiencies. Vox Sang 84:45–53, 2003
Sekul EA, Cupler EJ, Dalakas MC: Aseptic meningitis associated with high-dose intravenous immunoglobulin therapy: frequency and risk factors. Ann Intern Med 121:259–262, 1994
ACKNOWLEDGMENTS
The authors thank the following persons from Baxter (Vienna, Austria, and Westlake Village, CA, USA) for their support in the successful completion of this study, through study management, data management, biostatistics or reviewing the manuscript: Werner Engl, Lisa Grubbs, Cora Hiebinger, Bill Knowlton, Friedrich Maritsch, Queennette Santiago, Richard Schiff, Phillip Schroth, Marlies Sharkhawy and Cindy Wu.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Church, J.A., Leibl, H., Stein, M.R. et al. Efficacy, Safety and Tolerability of a New 10% Liquid Intravenous Immune Globulin [IGIV 10%] in Patients with Primary Immunodeficiency. J Clin Immunol 26, 388–395 (2006). https://doi.org/10.1007/s10875-006-9025-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-006-9025-3