Abstract
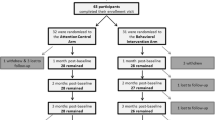
Between 30 and 50 % of MS patients may prematurely discontinue disease modifying therapies. Little research has examined how to best talk with patients who have discontinued treatment against medical advice. The aim of this pilot study was to determine whether telephone counseling increases disease modifying therapy (DMT) re-initiation among nonadherent patients with multiple sclerosis (MS). Participants were eligible if they had relapsing-remitting disease, had stopped taking a DMT, and had no plan to re-initiate treatment despite a provider recommendation. Following a baseline assessment, 81 patients were randomly assigned to either five 20 min, weekly sessions of Motivational Interviewing/Cognitive Behavioral Therapy (MI-CBT) or Treatment as Usual (TAU) with brief education. At 10 weeks, patients initially assigned to TAU switched over to MI-CBT. Compared to patients in the TAU group, patients undergoing MI-CBT were significantly more likely to indicate they were re-initiating DMT (41.7 vs. 14.3 %). These significant results were replicated among patients crossing over from TAU to MI-CBT. Treatment satisfaction was high, with 97 % of participants reporting that they would recommend MI-CBT to other patients with MS. Results of this pilot study provide initial support for the use of MI-CBT among MS patients who have discontinued treatment against medical advice.
Clinicaltrials.gov: NCT01925690.





Similar content being viewed by others
References
Amrhein, P. C., Miller, W. R., Yahne, C. E., Palmer, M., & Fulcher, L. (2003). Client commitment language during motivational interviewing predicts drug use outcomes. Journal of Consulting and Clinical Psychology, 71, 862–878.
Berger, B. A., Liang, H., & Hudmon, K. S. (2005). Evaluation of software-based telephone counseling to enhance medication persistency among patients with multiple sclerosis. Journal of the American Pharmacists Association, 45, 466–472.
Bruce, J. M., Hancock, L. M., & Lynch, S. G. (2010). Objective adherence monitoring in multiple sclerosis: Initial validation and association with self-report. Multiple Sclerosis, 16, 112–120.
Castro-Borrero, W., Graves, D., Frohman, T. C., Flores, A. B., Hardeman, P., Logan, D., et al. (2012). Current and emerging therapies in multiple sclerosis: A systematic review. Therapeutic Advances in Neurological Disorders, 5, 205–220.
Compston, A., & Coles, A. (2002). Multiple sclerosis. Lancet, 359, 1221–1231.
Ebers, G. C. (1998). Randomised double-blind placebo-controlled study of interferon beta-1a in relapsing/remitting multiple sclerosis. PRISMS (Prevention of Relapses and Disability by Interferon beta-1a Subcutaneously in Multiple Sclerosis) Study Group. The Lancet, 352, 1498–504.
Finkelsztejn, A. (2014). Multiple sclerosis: Overview of disease-modifying agents. Perspectives in Medicinal Chemistry, 6, 65–72.
Freedman, M. S., Hughes, B., Mikol, D. D., Bennett, R., Cuffel, B., Divan, V., et al. (2008). Efficacy of disease-modifying therapies in relapsing remitting multiple sclerosis: A systematic comparison. European Neurology, 60, 1–11.
Gerkovich, M., Williams, K., Catley, D., & Goggin, K. (2008). Development and validation of a scale to measure motivation to adhere to HIV medication. Poster presented at the 3rd international conference on HIV treatment adherence.
Goggin, K., Gerkovich, M., Williams, K., Banderas, J., Catley, D., Berkley-Patton, J., et al. (2013). A randomized controlled trial examining the efficacy of motivation counseling with observed therapy for anti-retroviral therapy adhrence. AIDS and Behavior, 17, 1992–2001.
Halpern, R., Agarwal, S., Dembek, C., Borton, L., & Lopez-Bresnahan, M. (2011). Comparison of adherence and persistence among multiple sclerosis patients treated with disease-modifying therapies: A retrospective administrative claims analysis. Patient Prefer Adherence, 5, 73–84.
Heesen, C., Kasper, J., Segal, J., Kopke, S., & Muhlhauser, I. (2004). Decision role preferences, risk knowledge, and information interests in patients with multiple sclerosis. Multiple Sclerosis, 10, 643–650.
Heesen, C., Kiopke, S., Solari, A., Geiger, F., & Kasper, J. (2013). Patient autonomy in multiple sclerosis—Possible goals and assessment strategies. Journal of the Neurological Sciences, 331, 2–9.
Hettema, J. E., & Hendricks, P. S. (2010). Motivational interviewing for smoking cessation: A meta-analytic review. Journal of Consulting and Clinical Psychology, 78, 868–884.
Kleinman, N. L., Beren, I. A., Rajagopalan, K., & Brook, R. A. (2010). Medication adherence with disease modifying treatments for multiple sclerosis among US employees. Journal of Medical Economics, 13, 633–640.
Kurtzke, J. F. (1983). Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology, 33, 1444–1452.
Miller, W., & Rollnick, S. (2013). Motivational interviewing: Helping people change (3rd ed.). New York: The Guilford Press.
Mohr, D., Boudewyn, A. C., Likosky, W., Levine, E., & Goodkin, D. E. (2001). Injectable medication for the treatment of multiple sclerosis: The influence of self-efficacy expectations and injection anxiety on adherence and ability to self-inject. Annals of Behavioral Medicine, 23, 125–132.
Mohr, D. C., Burke, H., Beckner, V., & Merluzzi, N. (2005). A preliminary report on a skills-based telephone-administered peer support programme for patients with multiple sclerosis. Multiple Sclerosis, 11, 222–226.
Mohr, D., Goodkin, D. E., Likosky, W., Gatto, N., Baumann, K., & Rudick, R. (1997). Treatment of depression improves adherence to interferon beta-1b therapy for multiple sclerosis. Archives of Neurology, 54, 531–533.
National Multiple Sclerosis Society. (2015a). MS prevalence [cited February 2, 2015]. http://www.nationalmssociety.org/About-the-Society/MS-Prevalence
National Multiple Sclerosis Society. (2015b). MS symptoms [cited February 2, 2015]. http://www.nationalmssociety.org/Symptoms-Diagnosis/MS-Symptoms
National Multiple Sclerosis Society. (2015c). Secondary-progressive MS (SPMS) [cited February 3, 2015]. http://www.nationalmssociety.org/What-is-MS/Types-of-MS/Secondary-progressive-MS
Parsons, J., Rosof, E., Punzalan, J., & Di Maria, L. (2005). Patient care and STDs. Aids, 19, 31–39.
Pietrolongo, E., Giordano, A., Kleinefeld, M., Confalonieri, P., Lugaresi, A., & Tortorella, C. (2013). Decision-making in multiple sclerosis consultations in Italy: Third oberserve and patient assessments. PLoS ONE, 8, e60721.
Pozzilli, C., Schweikert, B., Ecari, U., & Oentrich, W. (2011). Supportive strategies to improve adherence to IFN Beta-1b in multiple sclerosis—Results of the BPlus observational cohort study. Journal of Neurological Sciences, 307, 120–126.
Reynolds, M. W., Stephen, R., Seaman, C., & Rajagopalan, K. (2010). Persistence and adherence to disease modifying drugs among patients with multiple sclerosis. Current Medical Research and Opinion, 26, 663–674.
Simon, J. H., Jacobs, L. D., Campion, M., Wende, K., Simonian, N., Cookfair, D. L., et al. (1998). Magnetic resonance studies of intramuscular interferon beta-1a for relapsing multiple sclerosis. The Multiple Sclerosis Collaborative Research Group. Annals of Neurology, 43, 79–87.
Tullman, M. J. (2013). Overview of the epidemiology, diagnosis, and disease progression associated with multiple sclerosis. The American Journal of Managed Care, 19, S15–S20.
West, D., DiLillo, V., Bursac, Z., Gore, S., & Greene, P. (2007). Motivationa interviewing improves weight loss in women with type 2 diabetes. Diabetes Care, 30, 1081–1087.
Wong, J., Gomes, T., Mamdani, M., Manno, M., & O’Conner, P. (2011). Adherence to multiple sclerosis disease-modifying therapies in Ontario is low. Canadian Journal of Neurological Sciences, 38, 429–433.
Acknowledgments
This work was funded by a grant from the National Multiple Sclerosis Society (HC 0138) to the first author.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Jared Bruce provides unbranded talks as a member of the Novartis Speaker’s Bureau. He has also received research funding from Cephalon and is a paid consultant to the National Hockey League. Amanda Bruce, Sharon Lynch, Lauren Strober, Sean O’Bryan, Deborah Sobotka, Joan Thelen, Abigail Ness, Morgan Glusman, Kathy Goggin, Andrea Bradley-Ewing and Delwyn Catley declare that they have no conflict of interest.
Human and animal rights and Informed consent
All procedures followed were in accordance with ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients included in the study.
Rights and permissions
About this article
Cite this article
Bruce, J., Bruce, A., Lynch, S. et al. A pilot study to improve adherence among MS patients who discontinue treatment against medical advice. J Behav Med 39, 276–287 (2016). https://doi.org/10.1007/s10865-015-9694-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10865-015-9694-6