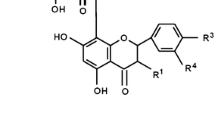
Abstract
Anthocyanins are considered as bioactive components of plant-based diets that provide protection against ischemic cardiovascular pathologies by mechanisms dependent on their antioxidant and reductive capacities. However, it is not clear whether similar anthocyanin-mediated mechanisms can provide protection against ischemia-induced brain mitochondrial injury and cell death. In this study, we compared effects of three cyanidin-3-glycosides – glucoside (Cy3G), galactoside (Cy3Gal) and rutinoside (Cy3R), with pelargonxidin-3-glucoside (Pg3G) and found that at 10–20 μM concentrations they have no direct effect on respiratory functions of mitochondria isolated from normal or ischemia-damaged rat brain slices. However, intravenous injection of Cy3Gal and Cy3G (0,025 mg/kg or 0,05 mg/kg what matches 10 μM or 20 μM respectively) but not Cy3R in rats protected against ischemia-induced caspase activation and necrotic cell death, and reduced infarct size in cerebral cortex and cerebellum. These effects correlated with cytochrome c reducing capacity of cyanidin-3-glycosides. In contrast, intravenous injection of 0,025 mg/kg Pg3G which has the lowest cytochrome c reducing capacity among investigated anthocyanins, had no effect on ischemia-induced caspase activation and necrosis but reduced brain infarct size whereas intravenous injection of 0,05 mg/kg of Pg3G slightly promoted necrosis in the brain. Our data suggest that reductive rather than antioxidant capacities of anthocyanins may be important components in providing protection against ischemic brain damage.












Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- Cy3G:
-
cyanidin-3-glucoside, anthocyanin
- Cy3Gal:
-
cyanidin-3-galactoside, anthocyanin
- Cy3R:
-
cyanidin-3-rutinoside, anthocyanin
- Pg3G:
-
pelargonidin-3-glucoside, anthocyanin
- AA:
-
ascorbic acid
- DHA:
-
dehydroascorbic acid
- LDH:
-
lactate dehydrogenase
- DEVD:
-
acetyl-Asp-Glu-Val-Asp-7-amido-4-methylcoumarin
- TTC:
-
2,3,5,-triphenyltetrazolium chloride.
References
Agus DB, Gambhir SS, Pardridge WM, Spielholz C, Baselga J, Vera JC, Golde DW (1997) Vitamin C crosses the blood-brain barrier in the oxidized form through the glucose transporters. J Clin Investig 100(11):2842–2848. https://doi.org/10.1172/JCI119832
Andres-Lacueva C, Shukitt-Hale B, Galli RL, Jauregui O, Lamuela-Raventos RM, Joseph JA (2005) Anthocyanins in aged blueberry-fed rats are found centrally and may enhance memory. Nutr Neurosci 8(2):111–120. https://doi.org/10.1080/10284150500078117
Arandarcikaite O, Jokubka R, Borutaite V (2015) Neuroprotective effects of nitric oxide donor NOC-18 against brain ischemia-induced mitochondrial damages: role of PKG and PKC. Neurosci Lett 586:65–70. https://doi.org/10.1016/j.neulet.2014.09.012
Borutaite V, Brown GC (2007) Mitochondrial regulation of Caspase activation by cytochrome oxidase and Tetramethylphenylenediamine via cytosolic cytochrome c redox state. J Biol Chem 282(43):31124–31130. https://doi.org/10.1074/jbc.M700322200
Borutaite V, Toleikis A, Brown GC (2013) In the eye of the storm: mitochondrial damage during heart and brain ischaemia. FEBS J 280(20):4999–5014. https://doi.org/10.1111/febs.12353
Davis JM, Murphy EA, Carmichael MD, Davis B (2009) Quercetin increases brain and muscle mitochondrial biogenesis and exercise tolerance. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology 296(4):R1071–R1077. https://doi.org/10.1152/ajpregu.90925.2008
Diehl KH, Hull R, Morton D, Pfister R, Rabemampianina Y, Smith D et al (2001) A good practice guide to the administration of substances and removal of blood, including routes and volumes. J Appl Toxicol 21(1):15–23. https://doi.org/10.1002/jat.727
Ereminas, G., Majiene, D., Sidlauskas, K., Jakstas, V., Ivanauskas, L., Vaitiekaitis, G., & Liobikas, J. (2017). Neuroprotective properties of anthocyanidin glycosides against H 2 O 2-induced glial cell death are modulated by their different stability and antioxidant activity in vitro. https://doi.org/10.1016/j.biopha.2017.07.077
Faria A, Pestana D, Teixeira D, Azevedo J, Freitas V, Mateus N, Calhau C (2010) Flavonoid transport across RBE4 cells: a blood-brain barrier model. Cell Mol Biol Lett 15(2):234–241. https://doi.org/10.2478/s11658-010-0006-4
Felgines, C., Talavéra, S., Texier, O., Besson, C., Fogliano, V., Lamaison, J.-L., … Galvano, F. (2006). Absorption and metabolism of red orange juice anthocyanins in rats. The British Journal of Nutrition, 95(5), 898–904. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/16611379
Fukumoto LR, Mazza G (2000) Assessing antioxidant and prooxidant activities of phenolic compounds. Journal of Agricultural and Food Chemistry 48(8):3597–3604 Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/10956156
Hancock JT, Desikan R, Neill SJ (2001) Does the redox status of cytochrome C act as a fail-safe mechanism in the regulation of programmed cell death? Free Radical Biology & Medicine 31(5):697–703 Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/11522455
Huang W, Zhu Y, Li C, Sui Z, Min W (2016) Effect of blueberry Anthocyanins Malvidin and glycosides on the antioxidant properties in endothelial cells. Oxidative Med Cell Longev 2016:1–10. https://doi.org/10.1155/2016/1591803
Ikizler, M., Erkasap, N., Dernek, S., Kural, T., & Kaygisiz, Z. (2007). Dietary polyphenol quercetin protects rat hearts during reperfusion: enhanced antioxidant capacity with chronic treatment. Anadolu Kardiyoloji Dergisi : AKD = the Anatolian Journal of Cardiology, 7(4), 404–410. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/18065337
Jackson TS, Xu A, Vita JA, Keaney JF (1998) Ascorbate prevents the interaction of superoxide and nitric oxide only at very high physiological concentrations. Circulation Research 83(9):916–922 Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/9797340
Joshi CN, Jain SK, Murthy PSR (2004) An optimized triphenyltetrazolium chloride method for identification of cerebral infarcts. Brain Res Protocol 13(1):11–17. https://doi.org/10.1016/j.brainresprot.2003.12.001
Kähkönen MP, Heinonen M (2003) Antioxidant activity of anthocyanins and their aglycons. J Agric Food Chem 51(3):628–633. https://doi.org/10.1021/jf025551i
Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, Messadeq N, Milne J, Lambert P, Elliott P, Geny B, Laakso M, Puigserver P, Auwerx J (2006) Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 127(6):1109–1122. https://doi.org/10.1016/j.cell.2006.11.013
Milbury PE, Kalt W (2010) Xenobiotic metabolism and berry flavonoid transport across the blood−brain barrier †. J Agric Food Chem 58(7):3950–3956. https://doi.org/10.1021/jf903529m
Min J, Yu S-W, Baek S-H, Nair KM, Bae O-N, Bhatt A, Kassab M, Nair MG, Majid A (2011) Neuroprotective effect of cyanidin-3-O-glucoside anthocyanin in mice with focal cerebral ischemia. Neurosci Lett 500(3):157–161. https://doi.org/10.1016/j.neulet.2011.05.048
Modun D, Music I, Katalinic V, Salamunic I, Boban M (2003) Comparison of protective effects of catechin applied in vitro and in vivo on ischemia-reperfusion injury in the isolated rat hearts. Croatian Medical Journal 44(6):690–696 Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/14652880
Pan Z, Voehringer DW, Meyn RE (1999) Analysis of redox regulation of cytochrome c-induced apoptosis in a cell-free system. Cell Death & Differentiation 6(7):683–688. https://doi.org/10.1038/sj.cdd.4400544
Sandoval-Acuña C, Ferreira J, Speisky H (2014) Polyphenols and mitochondria: an update on their increasingly emerging ROS-scavenging independent actions. Arch Biochem Biophys 559:75–90. https://doi.org/10.1016/j.abb.2014.05.017
Scarabelli TM, Mariotto S, Abdel-Azeim S, Shoji K, Darra E, Stephanou A, Chen-Scarabelli C, Marechal JD, Knight R, Ciampa A, Saravolatz L, de Prati AC, Yuan Z, Cavalieri E, Menegazzi M, Latchman D, Pizza C, Perahia D, Suzuki H (2009) Targeting STAT1 by myricetin and delphinidin provides efficient protection of the heart from ischemia/reperfusion-induced injury. FEBS Lett 583(3):531–541. https://doi.org/10.1016/j.febslet.2008.12.037
Shin W-H, Park S-J, Kim E-J (2006) Protective effect of anthocyanins in middle cerebral artery occlusion and reperfusion model of cerebral ischemia in rats. Life Sci 79(2):130–137. https://doi.org/10.1016/j.lfs.2005.12.033
Skemiene K, Jablonskiene G, Liobikas J, Borutaite V (2013a) Protecting the heart against ischemia/reperfusion-induced necrosis and apoptosis: the effect of anthocyanins. Medicina (Lithuania) 49(2):84–88
Skemiene K, Liobikas J, Borutaite V (2015) Anthocyanins as substrates for mitochondrial complex i - protective effect against heart ischemic injury. FEBS J 282(5):963–971. https://doi.org/10.1111/febs.13195
Skemiene K, Rakauskaite G, Trumbeckaite S, Liobikas J, Brown GC, Borutaite V (2013b) Anthocyanins block ischemia-induced apoptosis in the perfused heart and support mitochondrial respiration potentially by reducing cytosolic cytochrome c. Int J Biochem Cell Biol 45(1):23–29. https://doi.org/10.1016/j.biocel.2012.07.022
Stephanou A, Latchman DS (2004) STAT-1: a novel regulator of apoptosis. Int J Exp Pathol 84(6):239–244. https://doi.org/10.1111/j.0959-9673.2003.00363.x
Suto D, Sato K, Ohba Y, Yoshimura T, Fujii J (2005) Suppression of the pro-apoptotic function of cytochrome c by singlet oxygen via a haem redox state-independent mechanism. Biochem J 392(2):399–406. https://doi.org/10.1042/BJ20050580
Vera JC, Rivas CI, Fischbarg J, Golde DW (1993) Mammalian facilitative hexose transporters mediate the transport of dehydroascorbic acid. Nature 364(6432):79–82. https://doi.org/10.1038/364079a0
Vera JC, Rivas CI, Velásquez FV, Zhang RH, Concha II, Golde DW (1995) Resolution of the facilitated transport of Dehydroascorbic acid from its intracellular accumulation as ascorbic acid. J Biol Chem 270(40):23706–23712. https://doi.org/10.1074/jbc.270.40.23706
Youdim, K. A., Dobbie, M. S., Kuhnle, G., Proteggente, A. R., Abbott, N. J., & Rice-Evans, C. (2003). Interaction between flavonoids and the blood-brain barrier: in vitro studies. Journal of Neurochemistry, 85(1), 180–192. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/12641740
Ziberna L, Lunder M, Moze S, Vanzo A, Tramer F, Passamonti S, Drevensek G (2010) Acute cardioprotective and cardiotoxic effects of bilberry anthocyanins in ischemia-reperfusion injury: beyond concentration-dependent antioxidant activity. Cardiovasc Toxicol 10(4):283–294. https://doi.org/10.1007/s12012-010-9091-x
Funding
This project has received funding from European Social Fund (project No 09.3.3-LMT-K-712-01-0131) under grant agreement with the Research Council of Lithuania (LMTLT).
Author information
Authors and Affiliations
Contributions
Authors’ contributions: VB designed the research, ER carried out the procedures with animals. KP and KS performed the experiments. All authors analyzed data and contributed to the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All experimental procedures were reviewed and approved by the National Ethical Committee for Animal Care (Lisences No 0217 and No 0228) according to Directive 86/609/EEC. The rats were maintained and handled at Lithuanian University of Health Sciences animal house in agreement with the Guide for the Care and Use of Laboratory Rats.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Skemiene, K., Pampuscenko, K., Rekuviene, E. et al. Protective effects of anthocyanins against brain ischemic damage. J Bioenerg Biomembr 52, 71–82 (2020). https://doi.org/10.1007/s10863-020-09825-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-020-09825-9