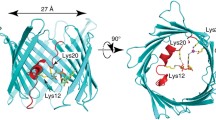
Abstract
The Voltage Dependent Anion Channel (VDAC) is the most abundant protein in the outer membrane of mitochondria. This strategic localization puts it at the heart of a great number of phenomena. Its recent implication in apoptosis is an example of the major importance of this protein and has created a surge of interest in VDAC. There is no atomic-resolution structure allowing a better understanding of the function of VDAC, so alternative techniques to X-ray diffraction have been used to study VDAC. Here we discuss structural models from folding predictions and review data acquired by Atomic Force Microscopy (AFM) imaging that allowed to observe VDAC’s structure and supramolecular organization in the mitochondrial outer membrane.
Similar content being viewed by others
References
Abrecht H, Goormaghtigh E, Ruysschaert JM, Homble F (2000) J Biol Chem 275:40992–40999
Baines CP, Kaiser RA, Sheiko T, Craigen WJ, Molkentin JD (2007) Nat Cell Biol 9:550–555
Bay DC, Court DA (2002) Biochem Cell Biol 80:551–562
Buzhynskyy B, Girmens J-F, Faigle W, Scheuring S (2007a) J Mol Biol 374:162–169
Buzhynskyy N, Hite RK, Walz T, Scheuring S (2007b) EMBO Rep 8:51–55
Colombini M (1980) Ann N Y Acad Sci 341:552–563
Colombini M, Blachly-Dyson E, Forte M (1996) Ion Channels 4:169–202
Cowan SW et al (1992) Nature 358:727–733
Dufrêne YF (2001) Micron 32:153–165
Gonçalves RP, Agnus G, Sens P, Houssin C, Bartenlian B, Scheuring S (2006) Nat Meth 3:1007–1012
Gonçalves RP, Buzhynskyy N, Prima V, Sturgis JN, Scheuring S (2007) J Mol Biol 369:413–418
Han D, Antunes F, Canali R, Rettori D, Cadenas E (2003) J Biol Chem 278:5557–5563
Hoogenboom BW, Suda K, Engel A, Fotiadis D (2007) J Mol Biol 370:246–255
Laurent V et al (2005) Biophys J 89:667–675
Mannella CA (1982) J Cell Biol 94:680–687
Mannella CA (1998) J Struct Biol 121:207–218
Mannella CA, Forte M, Colombini M (1992) J Bioenerg Biomembr 24:7–19
Mathupala SP, Ko YH, Pedersen PL (2006) Oncogene 25:4777–4786
Müller DJ, Baumeister W, Engel A (1999) Proc Natl Acad Sci USA 96:13170–13174
Rabinowitz M, Sinclair J, DeSalle L, Haselkorn R, Swift HH (1965) Proc Natl Acad Sci USA 5
Rief M, Gautel M, Oesterhelt F, Fernandez JM, Gaub HE (1997) Science 276:1109–1112
Roman I, Figys J, Steurs G, Zizi M (2006) Mol Cell Proteomics 5:1667–1680
Rostovtseva T, Colombini M (1996) J Biol Chem 271:28006–28008
Rostovtseva T, Tan W, Colombini M (2005) J Bioenerg Biomembranes 37:129–142
Rostovtseva T, Kazemi N, Weinrich M, Bezrukov SM (2006) J Biol Chem 281:37496–37506
Schatz G (1963) Biochem Biophys Res Commun 12:448–451
Schein SJ, Colombini M, Finkelstein A (1976) J Membr Biol 30:99–120
Scheuring S, Sturgis JN (2005) Science 309:484–487
Scheuring S, Müller DJ, Stahlberg H, Engel H-A, Engel A (2002) Eur Biophys J 31:172–178
Schlattner U, Dolder M, Wallimann T, Tokarska-Schlattner M (2001) J Biol Chem 276:48027–48030
Shao L, Kinnally KW, Mannella CA (1996) Biophys J 71:778–786
Shimizu S, Narita M, Tsujimoto Y (1999) Nature 399:483–487
Shimizu S, Matsuoka Y, Shinohara Y, Yoneda Y, Tsujimoto Y (2001) J Cell Biol 152:237–250
Song J, Midson C, Blachly-Dyson E, Forte M, Colombini M (1998) J Biol Chem 273:24406–24413
Tajeddine N et al. (2008) Oncogene. doi:10.1038/onc.2008.63
Vander Heiden MG, Li XX, Gottleib E, Hill RB, Thompson CB, Colombini M (2001) J Biol Chem 276:19414–19419
Wallin IE (1922) J Bacteriol 7:471–474
Young MJ, Bay DC, Hausner G, Court DA (2007) BMC Evolutionary Biology 7
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gonçalves, R.P., Buzhysnskyy, N. & Scheuring, S. Mini review on the structure and supramolecular assembly of VDAC. J Bioenerg Biomembr 40, 133–138 (2008). https://doi.org/10.1007/s10863-008-9141-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-008-9141-2