Abstract
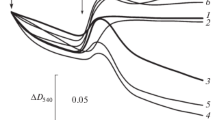
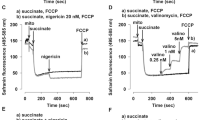
Superoxide anion generation and the impairment of oxidative phosphorylation yield were studied in rat skeletal muscle mitochondria submitted to anoxia/reoxygenationk in vitro. Production of superoxide anion was detected after several cycles of anoxia/reoxygenationk. Concomitantly, a decrease of state 3 respiration and phosphorylation yield (ADP/O) were observed. The latter resulted from a proton leak. The presence of palmitic acid during anoxia/reoxygenationk cycles led to a dose-dependent inhibition of superoxide anion production together with a partial protection of the ADP/O ratio measured after anoxia/reoxygenationk. The ADP/O decrease was shown to be due to a permeability transition pore-sustained proton leak, as it was suppressed by cyclosporine A. The permeability transition pore activation was induced during anoxia/reoxygenationk by superoxide anion, as it was cancelled by the spin trap (POBN), which scavenges superoxide anion and by palmitic acid, which induces mitochondrial uncoupling. It can be proposed that the palmitic acid-induced proton leak cancels the production of superoxide anion by mitochondria during anoxia/reoxygenationk and therefore prevents the occurrence of the superoxide anion-induced permeability transition pore-mediated proton leak after anoxia/reoxygenationk.
Similar content being viewed by others
Abbreviations
- A/R:
-
anoxia/reoxygenationk
- BSA:
-
bovine serum albumin
- CsA:
-
cyclosporine A
- EPR:
-
electron paramagnetic resonance
- FFA:
-
free fatty acids
- Jo:
-
respiratory rate in phosphorylating state 3
- Jp:
-
rate of ATP synthesis
- nBM:
-
n-butyl malonate
- O2 − :
-
superoxide anion
- OH:
-
hydroxyl radical
- OXPHOS:
-
oxidative phosphorylation
- PA:
-
palmitic acid
- POBN:
-
α-(4-pyridyl-1-oxide)-N-tert-butylnitrone
- PTP:
-
permeability transition pore
- ROS:
-
reactive oxygen species
- SOD:
-
superoxide dismutase
- UCP:
-
uncoupling protein
- UQ:
-
ubiquinone
- ΔμH+ :
-
proton electrochemical gradient
REFERENCES
Arsenijevic D, Onuma H, Pecqueur C, et al (2000) Nat Genet 26:435–439
Bernardi P, Petronilli V, Di Lisa, F, et al (2001) Trends Biochem Sci 26:112–127
Brand MD, Affourtit C, Esteves TC, et al (2004) Free Radic Biol Med 37:755–767
Britigan BE, Ratcliffe HR, Buettner GR, et al (1996) Biochem Biophys Acta 1290:231–240
Brookes PS (2005) Free Radic Biol Med 38:12–23
Czarna M, Jarmuszkiewicz W (2005) FEBS Lett 579:3136–31430
Di Lisa F, Bernardi P (1998) Mol Cell Biochem 184:379–391
Du G, Mouithys-Myckalad A, Sluse FE (1998) Free Radic Biol Med 25:1066–1074
Du G, Willet K, Jarmuszkiewicz W, Sluse-Goffart CM, et al (2004) Toxicol Mech Methods 14:97–101
Echtay KS, Esteves TC, Pakay JL, et al (2003) EMBO J 22:4103–4110
Echtay KS, Roussel D, St-Pierre J, et al (2002) Nature 15:96–99
Green DR, Kroemer G (2005) Science 305:626–629
Halestrap AP, Connern CP, Griffiths EJ, et al (1997) Mol Cell Biochem 174:167–172
Hollander J, Fiebig R, Gore M, et al (1999) Am J Physiol 277:856–862
Jarmuszkiewicz W, Navet R, Alberici LC, et al (2004) J Bioenergy Biomembr 36:493–502
Jezek P, Engstova H, Zackova M, et al (1998) Biochim Biophys Acta 1365:319–327
Klingenberg M, Echtay KS (2001) Biochim Biophys Acta 1504:128–143
Kowaltowski AJ, Costa ADT, Vercesi AE (1998) FEBS Lett 425:213–216
Mattiasson G, Shamloo M, Gido G, et al (2003) Nat Med 9:1062–1068
Morel I, Sergent O, Cogrel P, et al (1995) Free Radic Biol Med 18:303–310
Negre-Salvayre A, Hirtz C, Carrera G, et al FASEB J 11:809–815
Pou S, Ramos CL, Gladwell T, et al (1994) Anal. Biochem 217:76–83
Rosen GM, Cohen MS, Britigan BE, et al (1990) Free Radic Res Commun 9:187–195
Skulachev VP (1996) Q Rev Biophys 29:169–202
Talbot DA, Lambert AJ, Brand MD (2004) FEBS Lett 556:111–115
Tonkonogi M, Salhin K (1997) Acta Physiol Scand 161:345–353
Turrens JF, Boveris A (1980) Biochem J 191:421–427
Vasquez-Vivar J, Kalyanaraman B, Kennedy MC (2000) J Biol Chem 275:14064–14069
Vidal-Puig AJ, Grujic D, Zhang C-Y, et al (2000) J Biol Chem 275:16258–16266
Willet K, Detry O, Lambermont B, et al (2000) Transplantation 69:582–588
Wilson DO, Johnson P (2000) J Appl Physiol 88:1791–1796
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Navet, R., Mouithys-Mickalad, A., Douette, P. et al. Proton leak induced by reactive oxygen species produced during in vitro anoxia/reoxygenation in rat skeletal muscle mitochondria. J Bioenerg Biomembr 38, 23–32 (2006). https://doi.org/10.1007/s10863-006-9002-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-006-9002-9