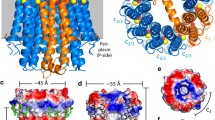
ATPases with unusual membrane-embedded rotor subunits were found in both F1F0 and A1A0 ATP synthases. The rotor subunit c of A1A0 ATPases is, in most cases, similar to subunit c from F0. Surprisingly, multiplied c subunits with four, six, or even 26 transmembrane spans have been found in some archaea and these multiplication events were sometimes accompanied by loss of the ion-translocating group. Nevertheless, these enzymes are still active as ATP synthases. A duplicated c subunit with only one ion-translocating group was found along with “normal” F0 c subunits in the Na+ F1F0 ATP synthase of the bacterium Acetobacterium woodii. These extraordinary features and exceptional structural and functional variability in the rotor of ATP synthases may have arisen as an adaptation to different cellular needs and the extreme physicochemical conditions in the early history of life.
Similar content being viewed by others
References
Aufurth, S., Schägger, H., and Müller, V. (2000). J. Biol. Chem. 275, 33297–33301.
Becher, B., and Müller, V. (1994). J. Bacteriol. 176, 2543–2550.
Coskun, Ü., Grüber, G., Koch, M. H., Godovac-Zimmermann, J., Lemker, T., and Müller, V. (2002). J. Biol. Chem. 277, 17327–17333.
Coskun, Ü., Radermacher, M., Müller, V., Ruiz, T., and Grüber, G. (2004). J. Biol. Chem. 279, 22759–22764.
Cross, R. L., and Müller, V. (2004). FEBS Lett. 576, 1–4.
Forster, A., Daniel, R., and Müller, V. (1995). Biochim. Biophys. Acta. 1229, 393–397.
Grüber, G., Svergun, D. I., Coskun, Ü., Lemker, T., Koch, M. H., Schägger, H., and Müller, V. (2001a). Biochemistry 40, 1890–1896.
Grüber, G., Wieczorek, H., Harvey, W. R., and Müller, V. (2001b). J. Exp. Biol. 204, 2597–2605.
Hendrickson, E. L., Kaul, R., Zhou, Y., Bovee, D., Chapman, P., Chung, J., Conway de Macario, E., Dodsworth, J. A., Gillett, W., Graham, D. E., Hackett, M., Haydock, A. K., Kang, A., Land, M. L., Levy, R., Lie, T. J., Major, T. A., Moore, B. C., Porat, I., Palmeiri, A., Rouse, G., Saenphimmachak, C., Soll, D., Van Dien, S., Wang, T., Whitman, W. B., Xia, Q., Zhang, Y., Larimer, F. W., Olson, M. V., and Leigh, J. A. (2004). J. Bacteriol. 186, 6956–6969.
Hilario, E., and Gogarten, J. P. (1998). J. Mol. Evol. 46, 703–715.
Ihara, K., Watanabe, S., Sugimura, K., and Mukohata, Y. (1997). Arch. Biochem. Biophys. 341, 267–272.
Inatomi, K. I., Maeda, M., and Futai, M. (1989). Biochem. Biophys. Res. Commun. 162, 1585–1590.
Kane, P. M., and Smardon, A. M. (2003). J. Bioenerg. Biomembr. 35, 313–321.
Kawasaki-Nishi, S., Nishi, T., and Forgac, M. (2001). Proc. Natl. Acad. Sci. U.S.A. 98, 12397–12402.
Lemker, T., Ruppert, C., Stöger, H., Wimmers, S., and Müller, V. (2001). Eur. J. Biochem. 268, 3744–3750.
Lemker, T., Grüber, G., Schmid, R., and Müller, V. (2003). FEBS Lett. 544, 206–209.
Lingl, A., Huber, H., Stetter, K. O., Mayer, F., Kellermann, J., and Müller, V. (2003). Extremophiles 7, 249–257.
Müller, V., Ruppert, C., and Lemker, T. (1999). J. Bioenerg. Biomembr. 31, 15–28.
Müller, V., Aufurth, S., and Rahlfs, S. (2001). Biochim. Biophys. Acta. 1505, 108–120.
Müller, V. (2003). Appl. Environ. Microbiol. 69, 6345–6353.
Müller, V., and Grüber, G. (2003). Cell. Mol. Life Sci. 60, 474–494.
Müller, V. (2004). J. Bioenerg. Biomembr. 36, 115–125.
Nelson, N., and Taiz, L. (1989). Trends Biochem. Sci. 14, 113–116.
Rahlfs, S., Aufurth, S., and Müller, V. (1999). J. Biol. Chem. 274, 33999–34004.
Reidlinger, J., and Müller, V. (1994). Eur. J. Biochem. 223, 275– 283.
Robb, F. T., Maeder, D. L., Brown, J. R., DiRuggiero, J., Stump, M D., Yeh, R. K., Weiss, R. B., and Dunn, D. M. (2001). Methods Enzymol. 330, 134–157.
Ruppert, C., Wimmers, S., Lemker, T., and Müller, V. (1998). J. Bacteriol. 180, 3448–3452.
Ruppert, C., Kavermann, H., Wimmers, S., Schmid, R., Kellermann, J., Lottspeich, F., Huber, H., Stetter, K. O., and Müller, V. (1999). J. Biol. Chem. 274, 25281–25284.
Sapra, R., Bagramyan, K., and Adams, M. W. (2003). Proc. Natl. Acad. Sci. U.S.A. 100, 7545–7550.
Schäfer, G., and Meyering-Vos, M. (1992). Biochim. Biophys. Acta 1101, 232–235.
Schäfer, G., Engelhard, M., and Müller, V. (1999). Microbiol. Mol. Biol. Rev. 63, 570–620.
Schemidt, R. A., Qu, J., Williams, J. R., and Brusilow, W. S. (1998). J. Bacteriol. 180, 3205–3208.
Slesarev, A. I., Mezhevaya, K. V., Makarova, K. S., Polushin, N. N., Shcherbinina, O. V., Shakhova, V. V., Belova, G. I., Aravind, L., Natale, D. A., Rogozin, I. B., Tatusov, R. L., Wolf, Y. I., Stetter, K. O., Malykh, A. G., Koonin, E. V., and Kozyavkin, S. A. (2002). Proc. Natl. Acad. Sci. U.S.A. 99, 4644– 4649.
Steinert, K., Wagner, V., Kroth-Pancic, P. G., and Bickel-Sandkötter, S. (1997). J. Biol. Chem. 272, 6261–6269.
Wilms, R., Freiberg, C., Wegerle, E., Meier, I., Mayer, F., and Müller, V. (1996). J. Biol. Chem. 271, 18843– 18852.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Müller, V., Lingl, A., Lewalter, K. et al. ATP Synthases With Novel Rotor Subunits: New Insights into Structure, Function and Evolution of ATPases. J Bioenerg Biomembr 37, 455–460 (2005). https://doi.org/10.1007/s10863-005-9491-y
Issue Date:
DOI: https://doi.org/10.1007/s10863-005-9491-y