Abstract
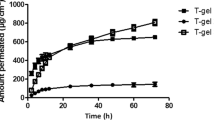
Lipid (L-α-phosphatidylcholine) was used in liposome-ketoprofen formulation to obtain vesicles systems characterized by a net positive charge along the liposomal surface. A careful analysis of vesicles formation and systems stability was made. Dynamic stability and specificity of liposomes disruption and prolonged release of ketoprofen was provided by steric effect accomplished on the vesicle surface by chitosan molecules, which were introduced into the system additionally. The retardation effect of the liposomes containing ketoprofen was tested in vitro and in vivo. The studies have shown that the liposomes containing ketoprofen obtained are characterized by a net positive charge and an average diameter of 1,287 nm for dialyzed solutions (pH 7.40). This formulation presents in vivo significant antinociceptive effects starting at 90 min, with a maximum intensity between 2 and 8 h, prolonged more than 10 h, and an analgesic activity within 3–4 h.





Similar content being viewed by others
References
Dudley GA, Czerkawski J, Meinrod A, Gillis G, Baldwin A, Scarpone M. Efficacy of naproxen sodium for exercise induced dysfunction muscle injury and soreness. Clin J Sport Med. 1997;10:3–10.
Predel HG, Koll R, Pabst H, Dieter R, Gallacchi G, Giannetti B, Bulitta M, Heidecker JL, Mueller EA. Diclofenac patch for topical treatment of acute impact injuries: a randomised, double blind, placebo controlled, multicentre study. Br J Sports Med. 2004;38:318–23.
Iwama T. NSAIDs and colorectal cancer prevention. J Gastroenterol. 2009;44:72–6.
McCarthy M. Comparative toxicity of nonsteroidal anti-inflammatory drugs. Am J Med. 2000;107:37–46.
Lemke TL, Williams DA, Roche VF, Zito SW. Foye’s principles of medical chemistry. 6th ed. Philadelphia: Wolter Kluwer; 2008.
Mathew ST, Devi SG, Prasanth VV, Vinod B. Formulation and in vitro–in vivo evaluation of ketoprofen-loaded albumin microspheres for intramuscular administration. J Microencapsul. 2009;26:456–69.
Del Gaudio P, Russo P, Lauro MR, Colombo P, Aquino RP. Encapsulation of ketoprofen and ketoprofen lysinate by prilling for controlled drug release. AAPS PharmSciTech. 2009;10:1178–85.
Rassu G, Gavini E, Spada G, Giunchedi P, Marceddu S. Ketoprofen spray-dried microspheres based on Eudragit® RS and RL: study of the manufacturing parameters. Drug Develop Ind Pharm. 2008;34:1178–87.
Rajesh KS, Bhowmik BB, Sa B. Effect of polyvinylpyrrolidone on physical characteristics of ketoprofen-loaded polystryrene microparticles. Indian J Pharm Sci. 2003;65:526–9.
Tan TW, Hu B, Jin XH, Zhang M. Release behavior of ketoprofen from chitosan/alginate microcapsules. J Bioact Compat Polym. 2003;18:207–18.
Barauskas J, Johnsson M, Tiberg F. Self-assembled lipid superstructures: beyond vesicles and liposomes. Nano Lett. 2005;5:1615–9.
Discher DE, Eisenberg A. Polymer vesicles. Science. 2002;29:7967–73.
Soussan E, Cassel S, Blanzat M, Rico-Lattes I. Drug delivery by soft matter: matrix and vesicular carriers. Angew Chem Int Ed. 2008;48:274–88.
Garlea A, Popa MI, Pohoata V, Melnig V. Ibuprofen/ketoprofen entrapment in chitosan based vesicle carrier. Rom J Biophys. 2007;17:157–68.
Garlea A, Melnig V, Popa MI. Nanostructured chitosan-surfactant matrices as polyphenols nanocapsules template with zero order release kinetics. J Mater Sci, Mater Med. 2010;21:1211–23.
Tiyaboonchai W. Chitosan nanoparticles: a promising system for drug delivery. Naresuan Univ J. 2003;11:51–66.
Riddick TM. Control of colloid stability through zeta potential. New York: Zetameter Inc; 1968.
Keefe FJ, Fillingim RB, Williams DA. Behavioral assessment of pain: nonverbal measures in animals and humans. ILAR J. 1991;33:3–13.
Walker EA, Butelman ER, DeCosta BR, Woods JH. Opioid thermal antinociception in rhesus monkeys: receptor mechanisms and temperature dependency. J Pharmacol Exp Ther. 1993;267:280–6.
Shin JW, Hwang KS, Kim YK, Leem JG, Lee C. Nonsteroidal an-tiinflammatory drugs suppress pain-related behaviors, but not referred hyperalgesia of visceral pain in mice. Anaesth Analg. 2006;102:195–200.
Winters WD, Hance AJ, Cadd GG, Quam DD. Acetic acid for analgesic screening. J Pharmacol Exp Therapeut. 2003;244:51–7.
Hamza M, Dionne RA. Mechanisms of non-opioid analgesics beyond cyclooxygenase enzyme inhibition. Curr Mol Pharmacol. 2009;2:1–14.
Pinardi G, Sierralta F, Miranda HF. Adrenergic mechanisms in antinociceptive effects of non steroidal anti-inflammatory drugs in acute thermal nociception in mice. Inflamm Res. 2002;51:219–22.
Mishra S, Suryawanshi R, Chawla V, Saraf S. Fabrication and characterization of solid lipid microparticles of ketoprofen. Ars Pharm. 2011;52:12–5.
Girard P, Verniers D, Coppé MC, Pansart Y. Nefopam and ketoprofen synergy in rodent models of antinociception. Eur J Pharmacol. 2008;584:263–71.
Acknowledgments
This work was financial supported by Internal Research Grant Nr. 16407/2009 of the Gr.T. Popa University of Medicine and Pharmacy, Iasi, Romania, and 195CPI/2008 PN II Capacitati MEC Grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tarţău, L., Cazacu, A. & Melnig, V. Ketoprofen-liposomes formulation for clinical therapy. J Mater Sci: Mater Med 23, 2499–2507 (2012). https://doi.org/10.1007/s10856-012-4712-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-012-4712-5