Abstract
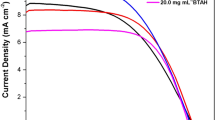
A series of thieno[3,2-b]indole-based dyes (IS 1–10) was readily synthesized in three steps from 2-(thien-2-yl)thieno[3,2-b]indole as the key precursor, and further applied as photosensitizers for dye-sensitized solar cells (DSSCs). In general, the prepared dyes have a push–pull (donor-π-linker-acceptor) structure, included thieno[3,2-b]indole ring system, bearing different aliphatic substituents at the nitrogen atom, as an electron-donating part, single thiophene unit as π-linker, and 2-cyanoacrylic acid (IS 1–5), or 5-(methylene)barbituric acid (IS 6–10) as an acceptor-anchoring group. The DSSC devices based on IS 1–10 dyes were fabricated using commercially available TiO2-coated photoanodes, and their photovoltaic characteristics were investigated. The DSSCs based on IS 1–5 dyes exhibited values of power conversion efficiency (PCE) in the range of 2.25–3.02%, while the DSSCs based on IS 6–10 dyes showed significantly low values of PCE in the range of 0.20–0.32% under AM 1.5G illumination (100 mW cm−2). The highest PCE value of 3.02% (Jsc = 7.59 mA cm−2, Voc = 0.62 V, FF = 0.64) was achieved for DSSC based on dye IS 4, bearing 2-cyanoacrylic acid as an acceptor-anchoring group among all dyes IS 1–10, whereas IS 9, bearing 5-(methylene)barbituric acid displays best PCE of 0.32% (Jsc = 0.87 mA cm−2, Voc = 0.53 V and FF = 0.68) among dyes IS 6–10. Therefore, additional studies, including thermogravimetric analysis, UV–Vis, and FTIR-measurements on the TiO2 surface, cyclic voltammograms, and photoelectrochemical measurements, were carried out for these two dyes.














Similar content being viewed by others
Data availability
Supplementary data to this article can be found online at https://doi.org/10.1007/s10854-022-07805-w.
Code availability
Not applicable.
References
L. Fraas, L. Partain (eds.), Solar Cells and Their Applications, 2nd edn. (Wiley, Chichester, 2010)
B. O’Regan, M. Grätzel, Nature 353, 737 (1991)
M. Grätzel, J. Photochem. Photobiol. C Photochem. Rev. 4, 145 (2003)
A. Błaszczyk, in Chem. Solut. Synth. Mater. Des. Thin Film Device Appl. (Elsevier, 2021), pp. 509–544
C.P. Lee, C.T. Li, K.C. Ho, Mater. Today 20, 267 (2017)
K. Sharma, V. Sharma, S.S. Sharma, Nanoscale Res. Lett. 13, 1 (2018)
G. Boschloo, Front. Chem. 7, 77 (2019)
H.A. Maddah, V. Berry, S.K. Behura, Renew. Sustain. Energy Rev. 121, 109–678 (2020)
Z. Yao, M. Zhang, H. Wu, L. Yang, R. Li, P. Wang, J. Am. Chem. Soc. 137, 3799 (2015)
N. Órdenes-Aenishanslins, G. Anziani-Ostuni, M. Vargas-Reyes, J. Alarcón, A. Tello, J.M. Pérez-Donoso, J. Photochem. Photobiol. B Biol. 162, 707 (2016)
H. Jiang, Y. Wu, A. Islam, M. Wu, W. Zhang, C. Shen, H. Zhang, E. Li, H. Tian, W.H. Zhu, ACS Appl. Mater. Interfaces 10, 13635 (2018)
Y. Mu, H. Wu, G. Dong, Z. Shen, S. Li, M. Zhang, J. Mater. Chem. A 6, 21493 (2018)
P. Kumaresan, S. Vegiraju, Y. Ezhumalai, S. Yau, C. Kim, W.-H. Lee, M.-C. Chen, P. Kumaresan, S. Vegiraju, Y. Ezhumalai, S.L. Yau, C. Kim, W.-H. Lee, M.-C. Chen, Polymers (Basel). 6, 2645 (2014)
S. Chaurasia, J.T. Lin, Chem. Rec. 16, 1311 (2016)
Y. Li (ed.), Organic Optoelectronic Materials (Springer, Cham, 2015)
M. Liang, J. Chen, Chem. Soc. Rev. 42, 3453 (2013)
Y. Wu, W. Zhu, Chem. Soc. Rev. 42, 2039 (2013)
T.N. Murakami, N. Koumura, Adv. Energy Mater. 9, 1802967 (2019)
A.S. Steparuk, R.A. Irgashev, G.L. Rusinov, E.V. Krivogina, P.I. Lazarenko, S.A. Kozyukhin, Russ. Chem. Bull. 68, 1208 (2019)
R.A. Irgashev, G.A. Kim, G.L. Rusinov, V.N. Charushin, ARKIVOC 2014, 123 (2014)
X.-H. Zhang, Y. Cui, R. Katoh, N. Koumura, K. Hara, J. Phys. Chem. C 114, 18283 (2010)
R. Kesavan, F. Attia, R. Su, P. Anees, A. El-Shafei, A.V. Adhikari, J. Phys. Chem. C 123, 24383 (2019)
L. Zhang, J.M. Cole, J. Mater. Chem. A 5, 19541 (2017)
X. Yang, M. Yanagida, L. Han, Energy Environ. Sci. 6, 54 (2012)
F. Wu, X. Li, Y. Tong, T. Zhang, J. Power Sources 342, 704 (2017)
P. Liu, W. Wang, S. Liu, H. Yang, Z. Shao, Adv. Energy Mater. 9, 1803017 (2019)
S. Sarker, H.W. Seo, Y.K. Jin, K.S. Lee, M. Lee, D.M. Kim, Electrochim. Acta 182, 493 (2015)
H. Elbohy, H. El-Mahalawy, N.A. El-Ghamaz, H. Zidan, Electrochim. Acta 319, 110 (2019)
H.J. Snaith, A. Abate, J.M. Ball, G.E. Eperon, T. Leijtens, N.K. Noel, S.D. Stranks, J.T.-W. Wang, K. Wojciechowski, W. Zhang, J. Phys. Chem. Lett. 5, 1511 (2014)
P.I. Lazarenko, S.A. Kozyukhin, A.I. Mokshina, A.A. Sherchenkov, T.N. Patrusheva, R.A. Irgashev, E.A. Lebedev, V.V. Kozik, Russ. Phys. J. 61, 196 (2018)
Acknowledgements
Analytical studies were carried out using equipment of the Center for Joint Use «Spectroscopy and Analysis of Organic Compounds» at the Postovsky Institute of Organic Synthesis of the Ural Branch of the Russian Academy of Sciences. This work was financially supported by the Russian Foundation for Basic Research (Project No. 18-29-11037 mk). A.S.S. is grateful to the partial financial support from the Ministry of Education and Science of the Russian Federation within the framework of the State Assignment for Research (Project No. AAAA-A19-119012490006-1). V.V.E. and V.A.G. are grateful to the partial financial support from the Ministry of Science and Higher Education of the Russian Federation in the frame of the State Task for 2021 IPCE RAS. E.V.K. is grateful to the partial financial support from the Foundation for Assistance to Small Innovative Enterprises the UMNIK (Project No. 14534GU/2019).
Author information
Authors and Affiliations
Contributions
Conceptualization: VVE, VAG, GLR, and SAK; device fabrication: ASS and EVK; methodology: ASS and RAI; validation: EFZ, EVB, and PIL; writing—original draft preparation: ASS and RAI; writing—review and editing: RAI and PIL; supervision: GLR and SAK.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Steparuk, A.S., Irgashev, R.A., Zhilina, E.F. et al. Performance evaluation of dye-sensitized solar cells (DSSCs) based on metal-free thieno[3,2-b]indole dyes. J Mater Sci: Mater Electron 33, 6307–6317 (2022). https://doi.org/10.1007/s10854-022-07805-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-022-07805-w