Abstract
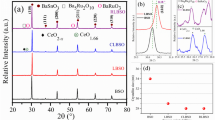
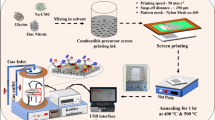
We report a comparative study on enhancing and inhibiting the sensing performance of Sr-doped ZnO (Sr0.01 Zn0.99O) and RuO2-activated Sr-doped ZnO heterostructured sensors towards the low concentration (≤ 50 ppm) of ammonia gas at ambient. Sub-microns sized with high specific surface area, high reactive, oxygen-deficient Sr-doped ZnO particles were synthesized at low temperature (196 °C) through facile glycine–nitrate solution combustion synthesis (SCS) method. Porous, adhered screen-printed film of Sr-doped ZnO with optical bandgap (3.22 eV) was dip-coated using 0.02 M RuCl3 aqueous solution to obtain RuO2 activation. Smaller crystallite size and lesser lattice distortion obtained with Sr-doping in ZnO enhance the gas response (S = 71) towards the 50 ppm of ammonia gas at room temperature. RuO2-activated Sr-doped ZnO sensor associated with lesser oxygen vacancies and a lower concentration of chemisorbed oxygen species due to passivation layer and no-spill-over activity of RuO2, which inhibits the gas response from 71 to 3. Sr-doped ZnO-based sensor shows high selectivity towards ammonia against 50 ppm of volatile organic compound (VOCs) vapor. Expeditious sensor kinetics (response time and recovery time) in the Sr-doped ZnO sensor was observed, in which smaller crystallite size offers a shorter distance for the diffusion of oxygen vacancies (Vo). Ultra-high-sensitive and selective sensors with ease and economical fabrication offer feasibility in industries and domestic applications where detection of the less concentration ammonia vapor is crucial.









Similar content being viewed by others
References
S. Cui et al., Fast and selective room-temperature ammonia sensors using silver nanocrystal-functionalized carbon nanotubes. ACS Appl. Mater. Interfaces. 4(9), 4898–4904 (2012)
B. Timmer et al., Ammonia sensors and their applications—a review. Sensors Actuators B 107(2), 666–677 (2005)
K.-I. Shimizu et al., Doped-vanadium oxides as sensing materials for high temperature operative selective ammonia gas sensors. Sensors Actuators B 141(2), 410–416 (2009)
C. Liu et al., The effect of noble metal (Au, Pd and Pt) nanoparticles on the gas sensing performance of SnO2-based sensors: a case study on the 221 high-index faceted SnO2 octahedra. CrystEngComm 17(33), 6308–6313 (2015)
Z. Pang et al., Fabrication of PA6/TiO2/PANI composite nanofibers by electrospinning–electrospraying for ammonia sensor. Colloids Surf. A 461, 113–118 (2014)
W. Liu et al., Highly sensitive room temperature ammonia gas sensor based on Ir-doped Pt porous ceramic electrodes. Appl. Surf. Sci. 390, 929–935 (2016)
Y.-F. Sun et al., Metal oxide nanostructures and their gas sensing properties: a review. Sensors 12(3), 2610–2631 (2012)
L. Zhu, W. Zeng, Room-temperature gas sensing of ZnO-based gas sensor: a review. Sensors Actuators A 267, 242–261 (2017)
G. Korotcenkov, The role of morphology and crystallographic structure of metal oxides in response of conductometric-type gas sensors. Mater. Sci. Eng. 61(1–6), 1–39 (2008)
S. Ansari et al., Grain size effects on H2 gas sensitivity of thick film resistor using SnO2 nanoparticles. Thin Solid Films 295(1–2), 271–276 (1997)
P. Sahay, R. Nath, Al-doped ZnO thin films as methanol sensors. Sensors Actuators B 134(2), 654–659 (2008)
H.-B. Lin et al., Mechanical reinforcement and piezoelectric properties of nanocomposites embedded with ZnO nanowhiskers. Scripta Mater. 59(7), 780–783 (2008)
Y. Chen et al., Enhanced photoelectrochemical properties of ZnO/ZnSe/CdSe/Cu2-xSe core–shell nanowire arrays fabricated by ion-replacement method. Appl. Catal. B 209, 110–117 (2017)
P.S. Venkatesh et al., Point defects assisted NH3 gas sensing properties in ZnO nanostructures. Sensors Actuators B 212, 10–17 (2015)
A.J. Kulandaisamy et al., Room temperature ammonia sensing properties of ZnO thin films grown by spray pyrolysis: effect of Mg doping. J. Alloy. Compd. 688, 422–429 (2016)
K. Lokesh et al., Effective ammonia detection using n-ZnO/p-NiO heterostructured nanofibers. IEEE Sens. J. 16(8), 2477–2483 (2016)
S.-W. Fan et al., UV-activated room-temperature gas sensing mechanism of polycrystalline ZnO. Appl. Phys. Lett. 95(14), 142106 (2009)
A. Dey, Semiconductor metal oxide gas sensors: a review. Mater. Sci. Eng. B 229, 206–217 (2018)
A. Renitta, K. Vijayalakshmi, Highly sensitive hydrogen safety sensor based on Cr incorporated ZnO nano-whiskers array fabricated on ITO substrate. Sensors Actuators B 237, 912–923 (2016)
K. Ravichandran et al., Effect of tungsten doping on the ammonia vapour sensing ability of ZnO thin films prepared by a cost effective simplified spray technique. Surf. Interfaces 18, 100412 (2019)
G.T. Rao, D.T. Rao, Gas sensitivity of ZnO based thick film sensor to NH3 at room temperature. Sensors Actuators B 55(2–3), 166–169 (1999)
R.S. Ganesh et al., Tuning the selectivity of NH3 gas sensing response using Cu-doped ZnO nanostructures. Sensors Actuators A 269, 331–341 (2018)
D.R. Miller et al., Nanoscale metal oxide-based heterojunctions for gas sensing: a review. Sensors Actuators B 204, 250–272 (2014)
M. Poloju et al., Improved gas sensing performance of Al doped ZnO/CuO nanocomposite based ammonia gas sensor. Mater. Sci. Eng. B 227, 61–67 (2018)
H. Tang et al., A selective NH3 gas sensor based on Fe2O3–ZnO nanocomposites at room temperature. Sensors Actuators B 114(2), 910–915 (2006)
G.H. Mhlongo et al., Room temperature ferromagnetism and gas sensing in ZnO nanostructures: influence of intrinsic defects and Mn Co, Cu doping. Appl. Surf. Sci. 390, 804–815 (2016)
D. Patil et al., Cr2O3-activated ZnO thick film resistors for ammonia gas sensing operable at room temperature. Sensors Actuators B 126(2), 368–374 (2007)
J. Iqbal et al., Effect of Co doping on morphology, optical and magnetic properties of ZnO 1-D nanostructures. J. Mater. Sci.: Mater. Electron. 24(11), 4393–4398 (2013)
R.S. Ganesh et al., Controlled synthesis of Ni-doped ZnO hexagonal microdiscs and their gas sensing properties at low temperature. Chem. Phys. Lett. 689, 92–99 (2017)
W. Wen et al., Gas-sensing property of a nitrogen-doped zinc oxide fabricated by combustion synthesis. Sensors Actuators B 184, 78–84 (2013)
M. Graf et al., Microfabricated gas sensor systems with sensitive nanocrystalline metal-oxide films. J. Nanopart. Res. 8(6), 823–839 (2006)
M. Zhang et al., Electromagnetic functions of patterned 2d materials for micro–nano devices covering ghz, thz, and optical frequency. Adv. Opt. Mater. 7(19), 1900689 (2019)
G. Manjunath et al., A scalable screen-printed high performance ZnO-UV and gas sensor: effect of solution combustion. Mater. Sci. Semicond. Process. 107, 104828 (2020)
T. Vijayan et al., Comparative investigation on nanocrystal structure, optical, and electrical properties of ZnO and Sr-doped ZnO thin films using chemical bath deposition method. J. Mater. Sci. 43(6), 1776–1782 (2008)
R. Heller et al., Precision determination of the lattice constants of zinc oxide. J. Appl. Phys. 21(12), 1283–1284 (1950)
K.P. Raj et al., Structural, optical, photoluminescence and photocatalytic assessment of Sr-doped ZnO nanoparticles. Mater. Chem. Phys. 183, 24–36 (2016)
J. Shen et al., An ESCA study of the interaction of oxygen with the surface of ruthenium. Appl. Surf. Sci. 51(1–2), 47–60 (1991)
G. Lemay et al., Synthesis of some ring-substituted ruthenocenes and their use in the preparation of Ru/ZSM-5 catalysts. Can. J. Chem. 64(9), 1943–1948 (1986)
A. Iriondo et al., Butyraldehyde production by butanol oxidation over Ru and Cu catalysts supported on ZrO2, TiO2 and CeO2. Studies in surface science and catalysis (Elsevier, Amsterdam, 2010), pp. 453–456
M. Navaneethan et al., Influence of Al doping on the structural, morphological, optical, and gas sensing properties of ZnO nanorods. J. Alloy. Compd. 698, 555–564 (2017)
J. Liao et al., The influence of post-annealing on the chemical structures and dielectric properties of the surface layer of Ba0.6Sr0.4TiO3 films. J. Phys. D 39(11), 2473 (2006)
M. Sosulnikov, Y.A. Teterin, X-ray photoelectron studies of Ca, Sr and Ba and their oxides and carbonates. J. Electron Spectrosc. Relat. Phenom. 59(2), 111–126 (1992)
W. Wang et al., Hydrous ruthenium oxide nanoparticles anchored to graphene and carbon nanotube hybrid foam for supercapacitors. Sci. Rep. 4, 4452 (2014)
Q. Zhu et al., Selectively enhanced UV and NIR photoluminescence from a degenerate ZnO nanorod array film. J. Mater. Chem. C 2(23), 4566–4580 (2014)
M. Wagh et al., Modified zinc oxide thick film resistors as NH3 gas sensor. Sensors Actuators B 115(1), 128–133 (2006)
C. Xu et al., Grain size effects on gas sensitivity of porous SnO2-based elements. Sensors Actuators B 3(2), 147–155 (1991)
N. Izu et al., The effects of the particle size and crystallite size on the response time for resistive oxygen gas sensor using cerium oxide thick film. Sensors Actuators B 94(2), 222–227 (2003)
A.K. Bal et al., Characterization and ammonia sensing properties of pure and ámodified ZnO films. Appl. Phys. A 103(2), 497–503 (2011)
G.K. Mani, J.B.B. Rayappan, Selective detection of ammonia using spray pyrolysis deposited pure and nickel doped ZnO thin films. Appl. Surf. Sci. 311, 405–412 (2014)
G.K. Mani, J.B.B. Rayappan, A highly selective and wide range ammonia sensor—nanostructured ZnO: Co thin film. Mater. Sci. Eng. B 191, 41–50 (2015)
Acknowledgements
Financial support from Science and Engineering Research Board (SERB), Department of Science and Technology, India (Grant No. ECR/2015/000339) is gratefully acknowledged. One of the author P. Nagaraju would like to thank DST-SERB for financial support to carry out the present work (Grant No. ECR/2016/000534).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Manjunath, G., Nagaraju, P. & Mandal, S. A comparative study on enhancer and inhibitor of glycine–nitrate combustion ZnO screen-printed sensor: detection of low concentration ammonia at room temperature. J Mater Sci: Mater Electron 31, 10366–10380 (2020). https://doi.org/10.1007/s10854-020-03584-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-020-03584-4