Abstract
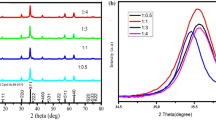
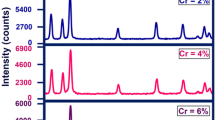
ZnAl2O4 nanoparticles (NPs) doped with Cr3+ (1.0 mol%) were prepared by citric acid assisted sol–gel method at pH values of 3, 5, 7, 9, 11 and 13 with the use of ammonia as pH controller. Analysis of x-ray diffraction (XRD) spectra show that the synthesized ZnAl2O4 NPs are cubic structured and the pH did not affect the structure significantly. The average crystallite size increases with an increase in pH. The photoluminescence (PL) intensity was found to be minimum and maximum for the sample synthesized at pH 3 and pH 13, respectively. The PL emission intensity was found to depend on the synthesis pH conditions. The scanning electron microscope (SEM) revealed that the morphology of ZnAl2O4:Cr3+ NPs changed with the pH values of the solution. Ultraviolet–visible (UV–Vis) measurements show that reflectance is maximum in UV and visible region for sample synthesized under acidic pH of 5. The energy band gap energy was tuned from 3.10 to 3.88 eV with increase in pH value from 3 to 13. It was observed that a strong alkaline nature of the solution enhances the structural and optical properties of ZnAl2O4:Cr3+ nanostructures.








Similar content being viewed by others
References
T.T. Loan, N.T. Thuy, N.N. Long, Synthesis and optical properties of ZnAl2O4:Cr3+, Tb3+ powders. Eur. Phys. J. Appl. Phys. 64, 10404 (2013)
M. Zawadzki, Microwave hydrothermal synthesis of ZnAl2O4. Solid State Sci. 8, 14–18 (2006)
S.G. Menon, D.N. Hebbar, S.D. Kulkarni, K.S. Choudhari, C. Santhosh, Facile synthesis and luminescence studies of nanocrystalline red emitting Cr:ZnAl2O4 phosphor. Mater. Res. Bull. 86, 63–71 (2017)
S.V. Motloung, F.B. Dejene, O.M. Ntwaeaborwa, H.C. Swart, Effects of catalyst/zinc mole fraction on ZnAl2O4:0.01% Cr3+ nanocrystals synthesized using sol–gel process. Mater. Res. Express 1, 045029 (2014)
W. Nie, F.M. Michel-Calendini, C. Linares, G. Boulon, C. Daul, New results on optical properties and term-energy calculations in Cr3+-doped ZnAl2O4. J. Lumin. 46(3), 177–190 (1990)
W. Mikenda, N-lines in the luminescence spectra of Cr3+-doped spinels (I) identification of N-lines. J. Lumin. 26, 53–66 (1981)
G. Samvit, S.G. Menon, K.S. Choudhari, S.A. Shivashankar, C. Santhosh, S.D. Kulkarni, Rapid annealing: a novel processing technique for Cr:ZnAl2O4 nanoparticles. J. Alloys Compd. 728, 484–489 (2017)
R. Wahab, S.G. Ansari, Y.S. Kim, M. Song, H.-S. Shin, The role of pH variation on the growth of zinc oxide nanostructures. Appl. Surf. Sci. 255, 4891–4896 (2009)
S.S. Alias, A.B. Ismail, A.A. Mohamad, Effect of pH on ZnO nanoparticle properties synthesized by sol–gel centrifugatio. J. Alloys Compd. 499, 231–237 (2010)
P. Sagar, P.K. Shishodia, R.M. Mehra, Influence of pH value on the quality of sol–gel derived ZnO films. Appl. Surf. Sci. 253, 5419–5424 (2007)
J. Ungula, B.F. Dejene, H.C. Swart, Effect of pH on the structural, optical and morphological properties of Ga-doped ZnO nanoparticles by reflux precipitation method. Phys. B 535, 251–257 (2018)
G. Lakshminarayana, L. Wondraczek, Photoluminescenceandenergy transfer in Tb3+/Mn2+ co-doped ZnAl2O4 glass ceramics. J. Solid State Chem. 184, 1931–1938 (2011)
J. Wrzyszcz, M. Zawadzki, J. Trawczynski, H. Grabowska, W. Mista, Some catalytic properties of hydrothermally synthesised zinc aluminate spinel. Appl. Catal. A 210, 263–269 (2001)
J.J. Kingsley, K. Suresh, K.C. Patil, Combustion synthesis of fine-particle metal aluminates. J. Mater. Sci. 25, 1305–1312 (1990)
S.K. Sharma, D. Gourier, T. Maldiney, B. Viana, E. Teston, D. Scherman, C. Richard, Persistent luminescence of AB2O4:Cr3+ (A = Zn, Mg, B = Ga, Al) spinels: new biomarkers for in vivo imaging. Opt. Mater. 36, 1901–1906 (2014)
S.V. Motloung, F.B. Dejene, O.M. Ntwaeaborwa, H.C. Swart, Effects of catalyst/zinc mole fraction on ZnAl2O4:0.01% Cr3+ nanocrystals synthesized using sol–gel process. Mater. Res. Express 1, 045029 (2014)
M.K. Hussen, F.B. Dejene, G.G. Gonfa, Effect of citric acid on material properties of ZnGa2O4:Cr3+ nanopowder prepred by sol-gel method. Appl. Phys. A 124, 390 (2018)
S. Safa, R. Azimirad, S.S. Moghaddamc, M. Rabbanic, Investigating on performance of CuO micro and nanostructures prepared by different precursors. Desalin. Water Treat. 57, 6723–6731 (2016)
M. Tsega, F.B. Dejene, Influence of acidic pH on the formulation ofTiO2 nanocrystalline powders with enhanced photoluminescence property. Heliyon 3, e00246 (2017)
S.V. Motloung, F.B. Dejene, H.C. Swart, O.M. Ntwaeaborwa, Effects of Cr3+mol% on the structure and optical properties of the ZnAl2O4:Cr3+nanocrystals Synthesized using sol–gel process. Ceram. Int. 41, 6776–6783 (2015)
L. F. Koao, F. B. Dejene, H. C. Swart Effect of pH on the properties of ZnO nanostructures prepared by chemical bath deposition method. Proceedings of SAIP2015, (2015) pp. 43–46. ISBN: 978-0-620-707145
G. Rani, Annealing effect on the structural, optical and thermoluminescent properties of ZnAl2O4:Cr3+. Powder Technol. 312, 354–359 (2017)
Miron and Grozescu, Hydrothermal synthesis of ZnAl2O4:Cr3+nanocrystals. Optoelectron. Adv. Mater. Rapid Commun. 6, 673–675 (2012)
M.K. Hussen, F.B. Dejene, Influence of annealing temprature on material properties of red emmiting ZnGa2O4:Cr3+ nano structures. J. Sol-Gel. Sci. Technol. 88, 454–464 (2018)
M.K. Hussen, F.B. Dejene, Effect of Cr3+ doping on structural and optical property of ZnGa2O4 synthesized by sol gel method. Optik 181, 514–523 (2018)
Acknowledgement
The authors would like to acknowledge and appreciate the financial support from the Ministry of science and higher education (Ethiopia) and National Research Foundation (NRF) South Africa.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hussen, M.K., Dejene, F.B. & Tsega, M. Effect of pH on material properties of ZnAl2O4:Cr3+ nano particles prepared by sol–gel method. J Mater Sci: Mater Electron 30, 10191–10201 (2019). https://doi.org/10.1007/s10854-019-01355-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-019-01355-4