Abstract
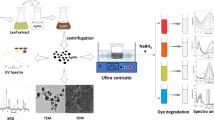
In this work, we reported preparation of the silver nanoparticles (Ag NPs) supported on natural diatomite surface as a cheap support using Alocasia macrorrhiza leaf extract. The existing phytochemicals in the A. macrorrhiza leaf extract converts the silver ions to Ag NPs on diatomite as a natural support. The green synthesized Ag/diatomite nanocomposite was characterized by using various analytical techniques such as FT-IR, XRD, FESEM, TEM, EDS. The synthesized Ag NPs were identified using FT-IR and UV–visible spectrophotometry. The Ag/diatomite nanocomposite was used as an effective nanocatalyst for the reduction of 4-nitrophenol (4-NP), 2,4-dinitrophenylhydrazine (2,4-DNPH), methyl orange (MO), Congo red (CR) and Nigrosin (NS) using sodium borohydride at ambient temperature. Furthermore, the Ag/diatomite nanocomposite can be recovered and reused five times without marked loss of its catalytic activity.


















Similar content being viewed by others
References
R. Dai, J. Chen, J. Lin, S. Xiao, S. Chen, Y. Deng, Reduction of nitro phenols using nitroreductase from E. coli in the presence of NADH. J. Hazard. Mater. 170(1), 141–143 (2009)
A.B. Prevot, C. Baiocchi, M.C. Brussino, E. Pramauro, P. Savarino, V. Augugliaro, G. Marci, L. Palmisano, Photocatalytic Degradation of Acid Blue 80 in Aqueous Solutions Containing TiO2 Suspensions. Environ. Sci. Technol. 35(5), 971–976 (2001)
M. Qin, K. Lin, Q. Shuai, H. Liang, J. Peng, C. Mao, Y. Ji, H. Wu, Facile synthesis of 2D single-phase Ni0.9Zn0.1O and its application in decolorization of dye. J. Mater. Sci. Mater. Electron. 29, 9740–9744 (2018)
U. Pagga, D. Brown, The degradation of dyestuffs: Part II Behaviour of dyestuffs in aerobic biodegradation tests. Chemosphere 15(4), 479–491 (1986)
L.A. Alfonso-Herrera, A.M. Huerta-Flores, L.M. Torres-Martínez, J.M. Rivera-Villanueva, D.J. Ramírez-Herrera, Hybrid SrZrO3-MOF heterostructure: surface assembly and photocatalytic performance for hydrogen evolution and degradation of indigo carmine dye. J. Mater. Sci. Mater. Electron. 29, 10395–10410 (2018)
F.K. Higson, Microbial degradation of nitroaromatic compounds, microbial degradation of nitroaromatic compounds. Adv. Appl. Microbiol. 37, 1–19 (1992)
F. Han, V. Kambala, M. Srinivasan, D. Rajarathnam, R. Naidu, Tailored titanium dioxide photocatalysts for the degradation of organic dyes in wastewater treatment: a review. Appl. Catal. A 359(1–2), 25–40 (2009)
H. Khojasteh, M. Salavati-Niasari, F.S. Sangsefidi, Photocatalytic evaluation of RGO/TiO2NWs/Pd-Ag nanocomposite as an improved catalyst for efficient dye degradation. J. Alloys Compds. 746, 611–618 (2018)
M. Goudarzi, M. Salavati-Niasari, Controllable synthesis of new Tl2S2O3 nanostructures via hydrothermal process; characterization and investigation photocatalytic activity for degradation of some anionic dyes. J. Mol. Liq. 219, 851–857 (2016)
M. Goudarzi, M. Mousavi-Kamazani, M. Salavati-Niasari, Zinc oxide nanoparticles: solvent-free synthesis, characterization and application as heterogeneous nanocatalyst for photodegradation of dye from aqueous phase. J. Mater. Sci. Mater. Electron. 28(12), 8423–8428 (2017)
B. Manu, S. Chaudhari, Anaerobic decolorisation of simulated textile wastewater containing azo dyes. Bioresour. Technol. 82(3), 225–231 (2002)
P. Wilhelm, D. Stephan, Photodegradation of rhodamine B in aqueous solution via SiO2@TiO2 nano-spheres. J. Photochem. Photobiol. A 185(1), 19–25 (2007)
P. Raveendran, J. Fu, S.L. Wallen, Completely “Green” synthesis and stabilization of metal nanoparticles. J. Am. Chem. Soc. 125(46), 13940–13941 (2003)
V.T.P. Vinod, P. Saravanan, B. Sreedhar, D. Keerthi Davi, R.B. Sashidhar, A facile synthesis and characterization of Ag, Au and Pt nanoparticles using a natural hydrocolloid gum kondagogu (Cochlospermum gossypium). Colloids Surf. 83(2), 291–298 (2011)
M. Nasrollahzadeh, S.M. Sajadi, A. Hatamifard, Waste chicken eggshell as a natural valuable resource and environmentally benign support for biosynthesis of catalytically active Cu/eggshell, Fe3O4/eggshell and Cu/Fe3O4/eggshell nanocomposites. Appl. Catal. B 191, 209–227 (2016)
M. Maryami, M. Nasrollahzadeh, E. Mehdipour, S.M. Sajadi, Preparation of the Ag/RGO nanocomposite by use of Abutilon hirtum leaf extract: a recoverable catalyst for the reduction of organic dyes in aqueous medium at room temperature. Int. J. Hydrogen Energy 41(46), 21236–21245 (2016)
F. Razi, S. Zinatloo-Ajabshir, M. Salavati-Niasari, Preparation, characterization and photocatalytic properties of Ag2ZnI4/AgI nanocomposites via a new simple hydrothermal approach. J. Mol. Liq. 225, 645–651 (2017)
M. Ghanbari, F. Soofivand, M. Salavati-Niasari, Simple synthesis and characterization of Ag2CdI4/AgI nanocomposite as an effective photocatalyst by co-precipitation method. J. Mol. Liq. 223, 21–28 (2016)
M. Goudarzi, Z. Zarghami, M. Salavati-Niasari, Novel and solvent-free cochineal-assisted synthesis of Ag-Al2O3 nanocomposites via solid-state thermal decomposition route: characterization and photocatalytic activity assessment. J. Mater. Sci. Mater. Electron. 27(9), 9789–9797 (2016)
S.S. Momeni, M. Nasrollahzadeh, A. Rustaiyan, Biosynthesis and application of Ag/bone nanocomposite for the hydration of cyanamides in Myrica gale L. extract as a green solvent. J. Colloid. Interface Sci. 499, 93–101 (2017)
T.T. Hanh, N.T. Thu, L.A. Quoc, N.Q. Hien, Synthesis and characterization of silver/diatomite nanocomposite by electron beam irradiation. Radiat. Phys. Chem. 139, 141–146 (2017)
P. Yuan, D. Liu, M. Fan, D. Yang, R. Zhu, F. Ge, J.X. Zhu, H. He, Removal of hexavalent chromium [Cr(VI)] from aqueous solutions by the diatomite-supported/unsupported magnetite nanoparticles. J. Hazard. Mater. 173, 614–621 (2010)
W. Zhaolun, Y. Yuxiang, Q. Xuping, Z. Jianbo, C. Yaru, N. Linxi, Decolouring mechanism of Zhejiang diatomite. Application to printing and dyeing wastewater. Environ. Chem. Lett. 3(1), 33–37 (2005)
P.R. Das, S. Akter, Md.T. Islam, M.H. Kabir, M. Haque, Z. Khatun, M. Nurunnabi, Z. Khatun, Y. Lee, R. Jahan, M. Rahmatullah, A selection of medicinal plants used for treatment of diarrhea by folk medicinal practitioners of Bangladesh. Am.-Eurasian J. Sustain. Agric. 6(3), 153–161 (2012)
A. Joshi, B.S. Karnawat, J.P. Narayan, V. Sharma, Alocasia macrorrhiza: a decorative but dangerous plant. Int. J. Sci. Stud. 3, 221–223 (2015)
S.T. Han, Medicinal Plants in the South Pacific, Western Pacific Series No. 19 (Regional Office for the Western Pacific, Geneva, 1998), pp. 9–10
H.-H. Yeoh, Y.-C. Wee, L. Watson, Taxonomic variation in total leaf protein amino acid compositions of monocotyledonous plants. Biochem. Syst. Ecol. 14(1), 91–96 (1986)
L. Nauheimer, P.C. Boyce, S.S. Renner, Giant taro and its relatives: a phylogeny of the large genus Alocasia (Araceae) sheds light on Miocene floristic exchange in the Malesian region. Mol. Phylogenet. Evol. 63(1), 43–51 (2012)
Z. Sun, X. Yang, G. Zhang, S. Zheng,, R.L. Frost, A novel method for purification of low grade diatomite powders in centrifugal fields. Int. J. Miner. Process. 125, 18–26 (2013)
M.A. Al-Ghouti, Y.S. Al-Degs, New adsorbents based on microemulsion modified diatomite and activated carbon for removing organic and inorganic pollutants from waste lubricants. Chem. Eng. J. 173(1), 115–128 (2011)
B. Sreedhar, D.K. Devi, D. Yada, Selective hydrogenation of nitroarenes using gum acacia supported Pt colloid an effective reusable catalyst in aqueous medium. Catal. Commun. 12(11), 1009–1014 (2011)
R.J. Kalbasi, A.A. Nourbakhsh, F. Babaknezhad, Synthesis and characterization of Ni nanoparticles-polyvinylamine/SBA-15 catalyst for simple reduction of aromatic nitro compounds. Catal. Commun. 12, 955–960 (2011)
M. Islam, P. Mondal, A.S. Roy, K. Tuhina, Synthesis, characterization and catalytic activities of a reusable polymer-anchored palladium(II) complex: effective catalytic hydrogenation of various organic substrates. Transition Met. Chem. 35(4), 427–435 (2010)
D. Shah, H. Kaur, Resin-trapped gold nanoparticles: an efficient catalyst for reduction of nitro compounds and Suzuki-Miyaura coupling. J. Mol. Catal. A 381, 70–76 (2014)
N. Sahiner, H. Ozay, O. Ozay, N. Aktas, New catalytic route: Hydrogels as templates and reactors for in situ Ni nanoparticle synthesis and usage in the reduction of 2- and 4-nitrophenols. Appl. Catal. A 385, 201–207 (2010)
C. Xu, Y. Yuan, R. Yuan, X. Fu, Enhanced photocatalytic performances of TiO2-graphene hybrids on nitro-aromatics reduction to amino-aromatics. RSC Adv. 3(39), 18002–18008 (2013)
M. Xie, F. Zhang, Y. Long, J. Ma, Pt nanoparticles supported on carbon coated magnetic microparticles: an efficient recyclable catalyst for hydrogenation of aromatic nitro-compounds. RSC Adv. 3, 10329–10334 (2013)
P. Wang, F. Zhang, Y. Long, M. Xie, R. Li, J. Ma, Stabilizing Pd on the surface of hollow magnetic mesoporous spheres: a highly active and recyclable catalyst for hydrogenation and Suzuki coupling reactions. Catal. Sci. Technol. 3(6), 1618–1624 (2013)
A.K. Shil, D. Sharma, N.R. Guha, P. Das, Solid supported Pd (0): an efficient recyclable heterogeneous catalyst for chemoselective reduction of nitroarenes. Tetrahedron Lett. 53(36), 4858–4861 (2012)
H. Yang, S. Li, X. Zhang, X. Wang, J. Ma, Imidazolium ionic liquid-modified fibrous silica microspheres loaded with gold nanoparticles and their enhanced catalytic activity and reusability for the reduction of 4-nitrophenol. J. Mater. Chem. A 2, 12060–12067 (2014)
X. Wang, J. Fu, M. Wang, Y. Wang, Z. Chen, J. Zhang, J. Chen, Q. Xu, Facile synthesis of Au nanoparticles supported on polyphosphazene functionalized carbon nanotubes for catalytic reduction of 4-nitrophenol. J. Mater. Sci. 49(14), 5056–5065 (2014)
Z. Duan, G. Ma, W. Zhang, Preparation of copper nanoparticles and catalytic properties for the reduction of aromatic nitro compounds. Bull. Korean Chem. Soc. 33(12), 4003–4006 (2012)
Z. Wang, C. Xu, G. Gao, X. Li, Facile synthesis of well-dispersed Pd–graphene nanohybrids and their catalytic properties in 4-nitrophenol reduction. RSC Adv. 4, 13644–13651 (2014)
N. Sahiner, Soft and flexible hydrogel templates of different sizes and various functionalities for metal nanoparticle preparation and their use in catalysis. Prog. Polym. Sci. 38(9), 1329–1356 (2013)
K.-L. Wu, R. Yu, X.-W. Wei, Monodispersed FeNi2 alloy nanostructures: solvothermal synthesis, magnetic properties and size-dependent catalytic activity. CrystEngComm 14(22), 7626–7632 (2012)
T.B. Devi, M. Ahmaruzzaman, S. Begum, A rapid, facile and green synthesis of Ag@AgCl nanoparticles for the effective reduction of 2,4-dinitrophenyl hydrazine. New J. Chem. 40, 1497–1506 (2016)
A. Rostami-Vartooni, M. Nasrollahzadeh, M. Alizadeh, Green synthesis of seashell supported silver nanoparticles using Bunium persicum seeds extract: application of the particles for catalytic reduction of organic dyes. J. Colloid Interface Sci. 470, 268–275 (2016)
B.K. Ghosh, S. Hazra, B. Nak, N.N. Ghosh, Preparation of Cu nanoparticle loaded SBA-15 and their excellent catalytic activity in reduction of variety of dyes. Powder Technol. 269, 371–378 (2015)
M. Atarod, M. Nasrollahzadeh, S.M. Sajadi, Euphorbia heterophylla leaf extract mediated green synthesis of Ag/TiO2 nanocomposite and investigation of its excellent catalytic activity for reduction of variety of dyes in water. J. Colloid Interface Sci. 462, 272–279 (2016)
P. Zhang, Y. Sui, C. Wang, Y. Wang, G. Cui, C. Wang, B. Liu, B. Zou, A one-step green route to synthesize copper nanocrystals and their applications in catalysis and surface enhanced Raman scattering. Nanoscale 6, 5343–5350 (2014)
B.R. Ganapuram, M. Alle, R. Dadigala, A. Dasari, V. Maragoni, V. Guttena, Catalytic reduction of methylene blue and Congo red dyes using green synthesized gold nanoparticles capped by salmalia malabarica gum. Int. Nano Lett. 5(4), 215–222 (2015)
Acknowledgements
We gratefully acknowledge the Iranian Nano Council and the University of Qom for the support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nasrollahzadeh, M., Mehdipour, E. & Maryami, M. Efficient catalytic reduction of nitroarenes and organic dyes in water by synthesized Ag/diatomite nanocomposite using Alocasia macrorrhiza leaf extract. J Mater Sci: Mater Electron 29, 17054–17066 (2018). https://doi.org/10.1007/s10854-018-9802-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-018-9802-9