Abstract
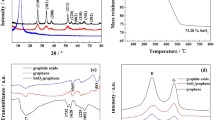
Humic acid obtained from wood, soil, and coal provides a naturally occurring highly oxidized carbonaceous two-dimensional material. Humic acid obtained from Leonardite coal and tin(II) chloride were used to synthesize graphene–SnO2 nanocomposites using scalable, thermal treatment processes. The humic acid-derived graphene–SnO2 nanocomposites showed the presence of graphene sheets with a unique crumpled and wrinkled morphology and SnO2 nanoparticles. The graphene–SnO2 nanocomposites were tested as anodes for lithium-ion batteries and showed high reversible specific capacities (641 mAh g−1). In addition, the graphene–SnO2 nanocomposites also exhibited high capacity retention upon cycling which is attributed to the interaction of SnO2 nanoparticles with the humic acid-derived graphene nanosheets that allows accommodation of highly reversible volumetric changes upon Li-ion insertion/de-insertion within the structure. In comparison, humic acid treated without the incorporation of SnCl2 during the synthesis process resulted in stacking of the nanosheets leading to low surface areas and low specific capacities. The scalable production of graphene nanocomposites from earth-abundant precursors opens up significant opportunities for low-cost and high performance materials for numerous energy storage and conversion devices.







Similar content being viewed by others
References
F. Bonaccorso, L. Colombo, G.H. Yu, M. Stoller, V. Tozzini, A.C. Ferrari, R.S. Ruoff, V. Pellegrini, Science 347, 10 (2015)
S. Zhuiykov, E. Kats, Ionics 19, 825 (2013)
N. Badi, J. Mater. Sci.: Mater. Electron. 27, 10342 (2016)
H. Liu, J. Huang, C. Xiang, J. Liu, X. Li, J. Mater. Sci.: Mater. Electron. 24, 3640 (2013)
H. Liu, J. Chen, R. Hu, X. Yang, H. Ruan, Y. Su, W. Xiao, J. Mater. Sci.: Mater. Electron. 27, 3968 (2016)
A.N. Naveen, P. Manimaran, S. Selladurai, J. Mater. Sci.: Mater. Electron. 26, 8988 (2015)
Y. Chen, J. Xu, Y. Yang, Y. Zhao, W. Yang, X. He, S. Li, C. Jia, J. Mater. Sci.: Mater. Electron. 27, 2564 (2016)
A.C. Neto, F. Guinea, N.M. Peres, K.S. Novoselov, A.K. Geim, Rev. Mod. Phys. 81, 1 (2009)
E.-S.M. Duraia, Z. Mansurov, S. Tokmoldin, Vacuum 86, 232 (2011)
F. Akbar, M. Kolahdouz, S. Larimian, B. Radfar, H. Radamson, J. Mater. Sci.: Mater. Electron. 26, 4347 (2015)
S. Bykkam, K.V. Rao, R. Naresh kumar, C.S. Chakra, T. Dayakar, J. Mater. Sci.: Mater. Electron. 27, 12574 (2016)
K. Li, M. Gao, Z. Huang, T. Pan, Y. Lin, J. Mater. Sci.: Mater. Electron. 28, 7468 (2017)
F. Belliard, P.A. Connor, J.T.S. Irvine, Solid State Ion. 135, 163 (2000)
Y. Zhao, X. Li, B. Yan, D. Li, S. Lawes, X. Sun, J. Power Sources 274, 869 (2015)
F. Ye, B. Zhao, R. Ran, Z. Shao, J. Power Sources 290, 61 (2015)
B. Huang, X. Li, Y. Pei, S. Li, X. Cao, R.C. Masse, G. Cao, Small. 12, 1945 (2016)
Y. Zhang, J. Xie, S. Zhang, P. Zhu, G. Cao, X. Zhao, Electrochim. Acta 151, 8 (2015)
S.-M. Paek, E. Yoo, I. Honma, Nano Lett. 9, 72 (2009)
P. Avouris, C. Dimitrakopoulos, Mater. Today 15, 86 (2012)
P.F. Hu, H. Wang, Y. Yang, J. Yang, J. Lin, L. Guo, Adv. Mater. 28, 3486 (2016)
H. Zhu, J. Yin, X. Zhao, C.Y. Wang, X.R. Yang, Chem. Commun. 51, 14708 (2015)
G.W. Beall, U.S. Patent Application US 8865307 B2 (2014)
E.M. Duraia, G.W. Beall, Superlatt. Microsc. 98, 379 (2016)
G.W. Beall, E.M. Duraia, Q. Yu, Z. Liu, Physica E 56, 331 (2014)
E.M. Duraia, G.W. Beall, Sens. Actuator B 220, 22 (2015)
E.M. Duraia, B. Henderson, G.W. Beall, J. Phys. Chem. Solids 85, 86 (2015)
C. Powell, G.W. Beall, Curr. Opin. Colloid Interface Sci. 20, 362 (2015)
D.R. Rolison, J.W. Long, J.C. Lytle, A.E. Fischer, C.P. Rhodes, T.M. McEvoy, M.E. Bourga, A.M. Lubers, Chem. Soc. Rev. 38, 226 (2009)
J.C. Groen, L.A.A. Peffer, J. Perez-Ramirez, Microporous Mesoporous Mater. 60, 1 (2003)
M. Sophie, A. Cellot, F. Ribot, C. Sanchez, L. Armelao, L. Gueneau, L. Delattre, J. Mater. Chem. 12, 2396 (2002)
Y.-C. Chen, J.-M. Chen, Y.-H. Huang, Y.-R. Lee, H.C. Shih, Surf. Coat. Technol. 202, 1313 (2007)
A.N. Fouda, M.E. Shazly, F. El-Tantawy, Rev. Adv. Mater. Sci. 45, 8 (2016)
S. Myungbeom, E. Park, B.M. Yoo, T.H. Han, H.B. Park, H. Kim, Carbon 110, 79 (2016)
M. Batzill, U. Diebold, Prog. Surf. Sci. 79, 47 (2005)
A.C. Ferrari, D.M. Basko, Nat. Nanotechnol. 8, 235 (2013)
E.S.M. Duraia, A. Fahami, G.W. Beall, J. Electron. Mater. 47, 1176 (2018)
A. Diéguez, A. Romano-Rodríguez, A. Vilà, J.R. Morante, J. Appl. Phys. 90, 1550 (2001)
M.-R. Yang, S.-Y. Chu, R.-C. Chang, Sens. Actuators B 122, 269 (2007)
A. Dieguez, A. Romano-Rodriguez, A. Vila, J.R. Morante, J. Appl. Phys. 90, 1550 (2001)
W. Chen, D. Ghosh, S.W. Chen, J. Mater. Sci. 43, 5291 (2008)
F. Ye, B.T. Zhao, R. Ran, Z.P. Shao, Chem.-Eur. J. 20, 4055 (2014)
Z. Du, X. Yin, M. Zhang, Q. Hao, Y. Wang, T. Wang, Mater. Lett. 64, 2076 (2010)
X. Li, X. Meng, J. Liu, D. Geng, Y. Zhang, M.N. Banis, Y. Li, J. Yang, R. Li, X. Sun, M. Cai, M.W. Verbrugge, Adv. Funct. Mater. 22, 1647 (2012)
G. Wang, X. Shen, J. Yao, J. Park, Carbon 47, 2049 (2009)
Acknowledgements
We gratefully acknowledge the National Science Foundation PREM (Grant No. DMR-1205670) for support of this work.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Duraia, ES.M., Niu, S., Beall, G.W. et al. Humic acid-derived graphene–SnO2 nanocomposites for high capacity lithium-ion battery anodes. J Mater Sci: Mater Electron 29, 8456–8464 (2018). https://doi.org/10.1007/s10854-018-8858-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-018-8858-x