Abstract
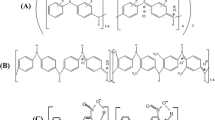
Poly(aniline-co-o-aminophenol) copolymer (PANoAP) is synthesized based on modified chemical route in highly acidic medium by using ferric chloride as an oxidizing agent in the presence of polyethylene glycol with molecular weight 200 as a surfactant. The chemical structure of resulting PANoAP is characterized by Fourier transform infrared FT-IR and 1HNMR spectroscopy. The morphological analysis was carried out by scanning electron microscope. The resulting copolymer exhibits nanostructured polydispersity. The electrochemical behavior of the copolymer is studied by cyclic voltammetry in 0.2 M H2SO4 solution, at a scan rate of 50 mV s−1. The potentials of oxidative signals of the PANoAP are found to be 0.141, 0.086, − 0.0083 and − 0.176 eV respectively. The reduction of quinoid structure in the copolymer backbone is detected by a broadly cathodic peak at 0.144 V and anodic signal at 0.141 V. Thin film of PANoAP is fabricated by spin coating with a thickness of 70 ± 3 nm. Based on the X-ray diffraction analysis, the PANoAP has crystalline nature depending on the strong interchain hydrogen bonding, and the electrostatic (dipole–dipole) interactions between the copolymer chains. The onset energy band value of the PANoAP thin film is found to be 2.19 eV. In addition, the electrical conductivity of the PANoAP thin film was found to be 6 × 10−2 S cm−1 at room temperature.












Similar content being viewed by others
References
B.C. Sherman, W.B. Euler, R.R. Force, The modern student laboratory: polyaniline-A conducting polymer: electrochemical synthesis and electrochromic properties. J. Chem. Educ. 71, A94 (1994)
H.S. Nalwa, Handbook of Advanced Electronic and Photonic Materials and Devices: Semiconductors, vol. 1 (Academic Press, Cambridge, 2001)
R. Holze, H. Nalwa, Advanced Functional Molecules and Polymers, vol. 2 (Gordon & Breach, Amsterdam, 2001), p. 171
M.K. Roković, K. Kvastek, V. Horvat-Radošević, L. Duić, Poly(ortho-ethoxyaniline) in corrosion protection of stainless steel. Corros. Sci. 49, 2567–2580 (2007)
P. Manisankar, C. Vedhi, G. Selvanathan, R. Somasundaram, Electrochemical and electrochromic behavior of novel poly(aniline-co-4, 4ʹ-diaminodiphenyl sulfone. Chem. Mater. 17, 1722–1727 (2005)
C.Y. Wang, V. Mottaghitalab, C.O. Too, G.M. Spinks, G.G. Wallace, Polyaniline and polyaniline–carbon nanotube composite fibres as battery materials in ionic liquid electrolyte. J. Power Sources. 163, 1105–1109 (2007)
K. Ghanbari, M. Mousavi, M. Shamsipur, Preparation of polyaniline nanofibers and their use as a cathode of aqueous rechargeable batteries. Electrochim. Acta 52, 1514–1522 (2006)
Y. Zou, L.-X. Sun, F. Xu, Biosensor based on polyaniline–Prussian blue/multi-walled carbon nanotubes hybrid composites. Biosens. Bioelectron. 22, 2669–2674 (2007)
M. Gerlache, S. Girousi, G. Quarin, J.-M. Kauffmann, Pulsed electrochemical detection of H2O2 on gold. Electrochim. Acta 43, 3467–3473 (1998)
H. Hu, B.E. Ortíz-Aguilar, L. Hechavarría, Effect of pH value of poly(ethylenimine)–H2SO4 electrolyte on electrochromic response of polyaniline thin films. Opt. Mater. 29, 579–584 (2007)
D. Maia, S. Das Neves, O. Alves, M.-A. De Paoli, Photoelectrochemical measurements of polyaniline growth in a layered material. Electrochim. Acta 44, 1945–1952 (1999)
H. Huang, Z. Zheng, J. Luo, H. Zhang, L. Wu, Z. Lin, Internal photoemission in polyaniline revealed by photoelectrochemistry. Synth. Met. 123, 321–325 (2001)
D. Gonçalves, R. Faria, M. Yonashiro, L. Bulhoes, Electrochemical oxidation of o-aminophenol in aqueous acidic medium: formation of film and soluble products. J. Electroanal. Chem. 487, 90–99 (2000)
R. Holze, In situ UV–Vis spectroelectrochemical studies of the copolymerization of o-aminophenol and aniline. Synth. Met. 156, 566–575 (2006)
A.G.C. Márquez, L.M.T. Rodríguez, A.M. Rojas, Synthesis of fully and partially sulfonated polyanilines derived from ortanilic acid: an electrochemical and electromicrogravimetric study. Electrochim. Acta 52, 5294–5303 (2007)
W.-C. Chen, T.-C. Wen, A. Gopalan, Role of anions to influence inductive behavior for poly(2-amino diphenylamine-co-aniline)—an electrochemical impedance spectroscopic analysis. Synth. Met. 130, 61–71 (2002)
A.H. Gemeay, R.G. El-Sharkawy, I.A. Mansour, A.B. Zaki, Preparation and characterization of polyaniline/manganese dioxide composites and their catalytic activity. J. Colloid Interface Sci. 308, 385–394 (2007)
A. Karyakin, A. Strakhova, A. Yatsimirsky, Self-doped polyanilines electrochemically active in neutral and basic aqueous solutions: electropolymerization of substituted anilines. J. Electroanal. Chem. 371, 259–265 (1994)
A.A. Karyakin, I.A. Maltsev, L.V. Lukachova, The influence of defects in polyaniline structure on its electroactivity: optimization of ‘self-doped’polyaniline synthesis. J. Electroanal. Chem. 402, 217–219 (1996)
Y. Wei, R. Hariharan, S.A. Patel, Chemical and electrochemical copolymerization of aniline with alkyl ring-substituted anilines. Macromolecules 23, 758–764 (1990)
M. Sato, S. Yamanaka, J. Nakaya, K. Hyodo, Electrochemical copolymerization of aniline with o-aminobenzonitrile. Electrochim. Acta 39, 2159–2167 (1994)
Q. Yang, Y. Zhang, H. Li, Y. Zhang, M. Liu, J. Luo, L. Tan, H. Tang, S. Yao, Electrochemical copolymerization study of o-toluidine and o-aminophenol by the simultaneous EQCM and in situ FTIR spectroelectrochemisty. Talanta 81, 664–672 (2010)
M. Hosny, N. Nowesser, A.S. Al Hussaini, M. Sh. Zoromba, Doped copolymer of polyanthranilic acid and o-aminophenol (AA-co-OAP): synthesis, spectral characterization and the use of the doped copolymer as precursor of α-Fe2O3 nanoparticles. J. Mol. Struct. 1106, 479–484 (2016)
R. Srivastava, R. Pandey, P. Venkataramani, Barium Manganate Oxidation in Organic-Synthesis. 1. Oxidation of Aromatic Primary Amines (Council Scientific Industrial Research Publ & Info Directorate, New Delhi, 1981), pp. 995–996
D. Borole, U. Kapadi, P. Kumbhar, D. Hundiwale, Influence of inorganic and organic supporting electrolytes on the electrochemical synthesis of polyaniline, poly(o-toluidine) and their copolymer thin films. Mater. Lett. 56, 685–691 (2002)
M. Bassyouni, S.A. Gutub, U. Javaid, M. Awais, S. Rehman, S.M.-S. Abdel Hamid, M.H. Abdel-Aziz, A. Abouel-Kasem, H. Shafeek, Assessment and analysis of wind power resource using Weibull parameters. Energy Explor. Exploit. 33, 105–122 (2015)
N.M. Hosny, M. Sh., G. Zoromba, Samir, S. Alghool, Synthesis, structural and optical properties of Eskolaite nanoparticles derived from Cr doped polyanthranilic acid (CrPANA). J. Mol. Struct. 1122, 117–122 (2016)
M.K. Ram, N. Sarkar, H. Ding, C. Nicolini, Synthesis of controlled copolymerisation of aniline and ortho-anisidine: a physical insight in its Langmuir–Schaefer films. Synth. Met. 123 (2001) 197–206
L.H. Mascaro, A.N. Berton, L. Micaroni, Electrochemical synthesis of polyaniline/poly-o-aminophenol copolymers in chloride medium. Int. J. Electrochem. (2011). https://doi.org/10.4061/2011/292581
R. Mazeikiene, A. Malinauskas, Electrochemical copolymerization of aniline with m-phenylenediamine. Synth. Met. 92, 259–263 (1998)
A. Malinauskas, M. Bron, R. Holze, Electrochemical and Raman spectroscopic studies of electrosynthesized copolymers and bilayer structures of polyaniline and poly(o-phenylenediamine). Synth. Met. 92, 127–137 (1998)
M.Sh.. Zoromba, N.A. El-Ghamaz, A.Z. El-Sonbati, A.A. El-Bindary, M.A. Diab, O. El-Shahat, Conducting polymers .VII. Effect of doping with iodine on the dielectrical and electrical conduction properties of polyaniline. Synth. React. Inorg. Met. Org. Nano-Met. Chem. 46, 1179–1188 (2016)
M.S. Zoromba, Novel and economic acid-base indicator based on (p-toluidine) oligomer: synthesis; characterization and solvatochromism applications. Spectrochim. Acta Part A 187, 61–67 (2017)
M.S. Zoromba, M. Abdel-Aziz, Ecofriendly method to synthesize poly(ο-aminophenol) based on solid state polymerization and fabrication of nanostructured semiconductor thin film. Polymer 120, 20–29 (2017)
S. Mu, Y. Zhang, J. Zhai, The electrochemical copolymerization of aniline with 2, 4-diaminophenol and the electric properties of the resulting copolymer. Electrochim. Acta 54, 3923–3929 (2009)
T. Nogami, T. Hishida, M. Yamada, H. Mikawa, Y. Shirota, Formations and reactions of o-benzoquinone mono-and di-imines. I. Bull. Chem. Soc. Jpn. 48, 3709–3714 (1975)
M. Fetizon, V. Balogh, M. Golfier, Oxidations with silver carbonate/celite. V. Oxidations of phenols and related compounds. J. Org. Chem. 36, 1339–1341 (1971)
F. Hua, E. Ruckenstein, Highly soluble conducting poly(ethylene oxide) grafted at two sites of poly(o-aminobenzyl alcohol). J. Polym. Sci. Part A 42, 4756–4764 (2004)
S. Kunimura, T. Ohsaka, N. Oyama, Preparation of thin polymeric films on electrode surfaces by electropolymerization of o-aminophenol. Macromolecules 21, 894–900 (1988)
C. Barbero, J.J. Silber, L. Sereno, Formation of a novel electroactive film by electropolymerization of ortho-aminophenol: study of its chemical structure and formation mechanism. Electropolymerization of analogous compounds. J. Electroanal. Chem. Interfacial Electrochem. 263, 333–352 (1989)
A. Zhang, C. Cui, Y. Chen, J. Lee, Synthesis and electrochromic properties of poly-o-aminophenol. J. Electroanal. Chem. 373, 115–121 (1994)
R. Tucceri, C. Barbero, J. Silber, L. Sereno, D. Posadas, Spectroelectrochemical study of poly-o-aminophenol. Electrochim. Acta 42, 919–927 (1997)
R. Torresi, S.C. de Torresi, C. Gabrielli, M. Keddam, H. Takenouti, Quartz crystal microbalance characterization of electrochemical doping of polyaniline films. Synth. Met. 61, 291–296 (1993)
M.S. Zoromba, A. Al-Hossainy, M. Abdel-Aziz, Conductive thin films based on poly(aniline-co-o-anthranilic acid)/magnetite nanocomposite for photovoltaic applications. Synth. Met. 231, 34–43 (2017)
J. Zhang, D. Shan, S. Mu, Chemical synthesis and electric properties of the conducting copolymer of aniline and o-aminophenol. J. Polym. Sci. Part A. 45, 5573–5582 (2007)
F. Hua, E. Ruckenstein, Copolymers of aniline and 3-aminophenol derivatives with oligo (oxyethylene) side chains as novel water-soluble conducting polymers. Macromolecules 37, 6104–6112 (2004)
J. Zhang, D. Shan, S. Mu, Electrochemical copolymerization of aniline with m-aminophenol and novel electrical properties of the copolymer in the wide pH range. Electrochim. Acta 51, 4262–4270 (2006)
U. Javaid, Z.M. Khan, M.B. Khan, M. Bassyouni, S.M.-S. Abdel-Hamid, M.H. Abdel-Aziz, S.W. ul Hasan, Fabrication and thermo-mechanical characterization of glass fiber/vinyl ester wind turbine rotor blade. Compos. Part B 91, 257–266 (2016)
J. Lambert, H. Shurvell, D. Lightner, R. Cooks, Organic Structural Spectroscopy (Prentice-Hall, Inc., New Jersey, 1998)
M. Liu, Y. Zhang, M. Wang, C. Deng, Q. Xie, S. Yao, Adsorption of bovine serum albumin and fibrinogen on hydrophilicity-controllable surfaces of polypyrrole doped with dodecyl benzene sulfonate—a combined piezoelectric quartz crystal impedance and electrochemical impedance study. Polymer 47, 3372–3381 (2006)
S. Mu, Poly(aniline-co-o-aminophenol) nanostructured network: electrochemical controllable synthesis and electrocatalysis. Electrochim. Acta 51, 3434–3440 (2006)
S. Mu, Electrochemical copolymerization of aniline and o-aminophenol. Synth. Met. 143, 259–268 (2004)
H. Xia, Q. Wang, Ultrasonic irradiation: a novel approach to prepare conductive polyaniline/nanocrystalline titanium oxide composites. Chem. Mater. 14, 2158–2165 (2002)
L. Ren, G. Zhang, J. Wang, L. Kang, Z. Lei, Z. Liu, Z. Liu, Z. Hao, Z.h. Liu, Adsorption–template preparation of polyanilines with different morphologies and their capacitance. Electrochim. Acta. 145, 99–108 (2014)
S. Banerjee, J.P. Saikia, A. Kumar, B. Konwar, Antioxidant activity and haemolysis prevention efficiency of polyaniline nanofibers. Nanotechnology 21, 045101 (2009)
J. Pouget, M. Jozefowicz, A.e..a. Epstein, X. Tang, A. MacDiarmid, X-ray structure of polyaniline. Macromolecules 24, 779–789 (1991)
J.P. Pouget, M.E. Jozefowicz, A.J. Epstein, X. Tang, A.G. MacDiarmidL, X-ray structure of polyaniline. Macromolecules 24(3), 779–789 (1991)
C. Saravanan, S. Palaniappan, F. Chandezon, Synthesis of nanoporous conducting polyaniline using ternary surfactant. Mater. Lett. 62, 882–885 (2008)
G. Thenmozhi, P. Arockiasamy, R.J. Santhi, Isomers of poly aminophenol: chemical synthesis, characterization, and its corrosion protection aspect on mild steel in 1 M HCl. Int. J. Electrochem. (2014). https://doi.org/10.1155/2014/961617
A.F. Al-Hossainy, Synthesis, spectral, thermal, optical dispersion and dielectric properties of nanocrystalline dimer complex (PEPyr–diCd) thin films as novel organic semiconductor. Bull. Mater. Sci. 39, 209–222 (2016)
V.D. Mote, Y. Purushotham, B.N. Dole, Williamson-Hall analysis in estimation of lattice strain in nanometer-sized ZnO particles. J. Theor. Appl. Phys. 6, 6 (2012)
A.M. El Sayed, S. Taha, M. Shaban, G. Said, Tuning the structural, electrical and optical properties of tin oxide thin films via cobalt doping and annealing. Superlattices Microstruct. 95, 1e13 (2016)
C.A. Amarnath, J. Kim, K. Kim, J. Choi, D. Sohn, Nanoflakes to nanorods and nanospheres transition of selenious acid doped polyaniline. Polymer 49, 432–437 (2008)
J. Albuquerque, L.C. Mattoso, D. Balogh, R. Faria, J. Masters, A. MacDiarmid, A simple method to estimate the oxidation state of polyanilines. Synth. Met. 113, 19–22 (2000)
H.-Q. Xie, Y.-M. Ma, J.-S. Guo, Secondary doping phenomena of two conductive polyaniline composites. Synth. Met. 123, 47–52 (2001)
G. Lucovsky, On the photoionization of deep impurity centers in semiconductors. Solid State Commun. 3, 299–302 (1965)
A. Badr, A. El-Amin, A. Al-Hossainy, Elucidation of charge transport and optical parameters in the newly 1CR-dppm organic crystalline semiconductors. J. Phys. Chem. C 112, 14188–14195 (2008)
M.D. Giulio, G. Micocci, R. Rella, P. Siciliano, A. Tepore, Optical absorption of tellurium suboxide thin films. Phys. Status Solidi (a). (1993). https://doi.org/10.1002/pssa.2211360236
A.F. Al-Hossainy, A. Ibrahim, Structural, optical dispersion and dielectric properties of novel chromium nickel organic crystalline semiconductors. Mater. Sci. Semicond. Process. 38, 13–23 (2015)
S. Karadeniz, 60 Co γ-ray irradiation effects on dielectric characteristics of tin oxide films of different thicknesses on n-type Si (111) substrates. Nucl. Instrum. Methods Phys. Res. Sect. B 260, 571–578 (2007)
B. Barış, H.G. Özdemir, N. Tuğluoğlu, S. Karadeniz, ÖF. Yüksel, Z. Kişnişci, Optical dispersion and dielectric properties of rubrene organic semiconductor thin film. J. Mater. Sci.Mater. Electron. 25, 3586–3593 (2014)
M. Dongol, M. El-Nahass, A. El-Denglawey, A. Elhady, A. Abuelwafa, Optical properties of nano 5, 10, 15, 20-tetraphenyl-21H, 23H-prophyrin nickel (II) Thin Films. Curr. Appl. Phys. 12, 1178–1184 (2012)
A.F. Al-Hossainy, A. Ibrahim, The effects of annealing temperature on the structural properties and optical constants of a novel DPEA-MR-Zn organic crystalline semiconductor nanostructure thin films. Opt. Mater. 73, 138–153 (2017)
M. Leclerc, G. D’Aprano, G. Zotti, Structure-property relationships in polyaniline derivatives. Synth. Met. 55, 1527–1532 (1993)
F. Zuo, M. Angelopoulos, A.G. MacDiarmid, A.J. Epstein, Transport studies of protonated emeraldine polymer: a granular polymeric metal system. Phys. Rev. B 36, 3475 (1987)
M.A. Chougule, S.G. Pawar, P.R. Godse, R.N. Mulik, S. Sen, V.B. Patil, Synthesis and characterization of polypyrrole (PPy) thin films. Soft Nanosci. Lett. 1, 6 (2011)
A. Kobayashi, H. Ishikawa, K. Amano, M. Satoh, E. Hasegawa, Electrical conductivity of annealed polyaniline. J. Appl. Phys. 74, 296–299 (1993)
M. Gosh, A. Barman, A. Meikap, S. De, S. Chatterjee, Hopping transport in HCl doped conducting polyaniline. Phys. Lett. A 260, 138–148 (1999)
Z. Zhao, D. Morel, C. Ferekides, Electrical and optical properties of tin-doped CdO films deposited by atmospheric metalorganic chemical vapor deposition. Thin Solid Films. 413, 203–211 (2002)
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Slimane, A.B., Al-Hossainy, A.F. & Zoromba, M.S. Synthesis and optoelectronic properties of conductive nanostructured poly(aniline-co-o-aminophenol) thin film. J Mater Sci: Mater Electron 29, 8431–8445 (2018). https://doi.org/10.1007/s10854-018-8856-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-018-8856-z