Abstract
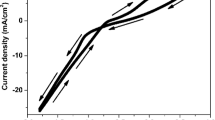
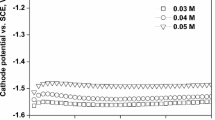
Nanocrystalline nickel (Ni) films were obtained by electrodeposition from chloride aqueous solution with different pH values. The influence of electrolyte pH on the morphological, structural, and magnetic properties is studied, using scanning electron microscopy (SEM), X-ray diffraction (XRD), and alternating gradient force magnetometry (AGFM) techniques. SEM studies revealed a granular and compact structure of the surface of the electrodeposited Ni layers, and the variation of film roughness with bath pH is established. XRD analysis gives evidence of strongly textured Ni films along the (111) direction, with the face-centered cubic (fcc) structure for all baths pH. Magnetic properties such as coercivity, remanence, saturation magnetization, and squareness showed strong dependence on the baths pH and crystallite size. High coercivity was attributed to the presence of the small crystallite size of deposits. The properties of the deposit are greatly influenced by the solution pH.




Similar content being viewed by others
References
C.D. Mee, E.D. Daniel (eds.), Magnetic Recording Technology (IEEE. Press, New York, 1997)
Y. Nakamura, in Magnetic Storage Systems Beyond 2000, Ed. by G. C. Hadjipanayis, NATO Sciences Series Kluwer, Vol. 41, (Dordrecht, 2001) p. 75
K. Ouchi, N. Honda, IEEE. Trans. Magn. 36, 16 (2000)
P. Ciureanu, S. Middelhoek, Thin Film Resistive Sensors (IOP Publishing, London, 1992)
K. O`Grady, H. Laidler, J. Magn. Magn. Mater. 200, 616 (1999)
U. Erb, A.M. El-Sherik, G. Palumbo, K.T. Aust, “Synthesis, structure and properties of electrodeposited nanocrystalline materials”. NanoStructured. Mater. 2, 383 (1993)
K. Appelt, Z. Dominiczak, A. Nowacki, M. Pasz-Kiewicz, Electrochim. Acta 10, 617 (1965)
J. Deng, X. Zhang, E. Min. Appl. Catal. 37, 339 (1988)
T. Homma, K. Noda, T. Watanabe, T. Osaka, Japn. J. Appl. Phys. 30, 1979 (1991)
C. Scheck, P. Evans, G. Zangari, R. Schad, Appl. Phys. Lett. 8, 2853 (2003)
U.S. Mohanty, B.C. Tripathy, P. Singh, S.C. Das, J. Electroanal.Chem. 526, 63 (2002)
Southampton Electrochemistry Group, in Instrumental Methods in Electrochemistry, ed. by T.J. Kemp, et al. (Ellis Horwood Ltd, Chichester, UK, 1985). Chapter 9
B.C. Banerjee, A. Goswani, J. Electrochem. Soc. 106, 20 (1959)
J. Amblard, M. Froment, G. Maurin, N. Spyrellis, E. Trevisan-Souteyr, Electrochim. Acta 28, 909 (1983)
E. Gómez, R. Pollina, E. Vallé, J. Electroanal. Chem. 397, 111 (1995)
C.S. Lin, P.C. Hsu, L. Chang, C.H. Chen, J. Appl. Electrochim. 31, 925 (2001)
A.G. Munoz, D.R. Salinas, J.B. Bessone, Thin Solid Films 429, 119 (2003)
G.R.P. Malpass, M. Kalaji, E.C. Venancio, A.J. Motheo, Electrochim. Acta 49, 4933 (2004)
D. Grujicic, B. Pesic, Electrochim. Acta 51, 2678 (2005)
A. Azizi, A. Sahari, G. Schmerber, A. Dinia, Int. J. Nanosci. 7, 345 (2008)
J.P. Hoare, J. Electrochem. Soc. 133, 2491 (1986)
H.P. Klug, L.E. Alexander, X-ray Diffraction Procedures for Polycrystalline and Amorphous Materials, 2nd edn. (Wiley, New York, 1974), p. 757
T. Watanabe, Nano Plating, Elsevier Science (2004) pp. 4, 29, 344
M. Motoyama, Y. Fukunaka, T. Sakka, Y.H. Ogata, J. Electrochem. Soc. 153, C502 (2006)
N.D. Nikolic, K.I. Popov, Lj.J. Pavlovic, M.G. Pavlovic, Surf. Coat. Technol. 201, 560 (2006)
P.L. Cavallotti, E. Galbiati, Tu. Chen, in Electroplating Engineering and Waste Recycle, ed. by D.D. Snyder, U. Landau, R. Sard (ECS Pub, Pennington, NJ, 1983), p. 130
B. D. Cullity, Elements of X-rays Diffraction, 2nd edn. (Addison-Wesley, London, 1978), p. 102
M. Pourbaix, Atlas of Electrochemical Equliibria in Aqueous Solution (Pergamon Press, Oxford, 1966)
G.B. Han, R.W. Gao, S.S. Yan, H.Q. Liu, S. Fu, W.C. Feng, W. Li, X.M. Li, J. Magn. Magn. Mater. 281, 6 (2004)
M.K. Griffiths, J.E.L. Bishop, J.M. Tucker, H.A. Davies, J. Magn. Magn. Mater. 183, 49 (1999)
G. Herzer, IEEE. Trans. Magn. 26, 1397 (1990)
T. Schrefl, J. Fidler, H. Kronmüller, Phys. Rev. B. 49, 6100 (1994)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boubatra, M., Azizi, A., Schmerber, G. et al. Morphology, structure, and magnetic properties of electrodeposited Ni films obtained from different pH solutions. J Mater Sci: Mater Electron 22, 1804–1809 (2011). https://doi.org/10.1007/s10854-011-0366-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-011-0366-1