Abstract
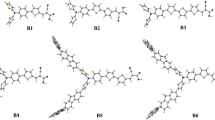
To screen efficient sensitizers for dye-sensitized solar cells (DSSCs), two series of porphyrin sensitizers have been reengineered based on one of the best sensitizers YD2-o-C8 by introducing different heterocycles into acceptor part to form stronger acceptors. The electronic structures and optical properties of these sensitizers have been investigated using density functional theory and its time-dependent density functional theory version. The computational results suggest that the stronger acceptor can result in a narrower HOMO–LUMO energy gap, an obvious red-shift and stronger absorption in long-wavelength region compared with YD2-o-C8. Meanwhile, the analyses of electron density difference plots suggest that all designed sensitizers possess longer electron transfer distance, larger fraction of electron exchange, and smaller overlap between the zones of density depletion and increment than these of YD2-o-C8, indicating enhanced electron transfer ability from donor to acceptor groups. Moreover, the designed dyes exhibit good performance in terms of the electron injection ability, the excited state lifetime, and the strength of the interaction between dye and the TiO2 surface. As a whole, all the designed dyes, especially P4 and P6 may act as excellent sensitizers for high-efficiency DSSCs.




Similar content being viewed by others
References
O’regan B, Grfitzeli M (1991) A low-cost, high-efficiency solar cell based on dye-sensitized. Nature 353(6346):737–740
Yu G, Gao J, Hummelen JC, Wudl F, Heeger AJ (1995) Polymer photovoltiac cells: enhanced efficiencies via a network of internal donor–acceptor heterojunctions. Science 270(5243):1789
Lee MM, Teuscher J, Miyasaka T, Murakami TN, Snaith HJ (2012) Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites. Science 338(6107):643–647
Jung J, Pang X, Feng C, Lin Z (2013) Semiconducting conjugated polymer-inorganic tetrapod nanocomposites. Langmuir 29(25):8086–8092
Pang X, Zhao L, Feng C, Lin Z (2011) Novel amphiphilic multiarm, starlike coil-rod diblock copolymers via a combination of click chemistry with living polymerization. Macromolecules 44(18):7176–7183
Yella A, Lee H-W, Tsao HN, Yi C, Chandiran AK, Nazeeruddin MK, Diau EW-G, Yeh C-Y, Zakeeruddin SM, Grätzel M (2011) Porphyrin-sensitized solar cells with cobalt(II/III)-based redox electrolyte exceed 12 % efficiency. Science 334(6056):629–634
Mathew S, Yella A, Gao P, Humphry-Baker R, Curchod BF, Ashari-Astani N, Tavernelli I, Rothlisberger U, Nazeeruddin MK, Grätzel M (2014) Dye-sensitized solar cells with 13 % efficiency achieved through the molecular engineering of porphyrin sensitizers. Nat Chem 6(3):242–247
Li L-L, Diau EW-G (2013) Porphyrin-sensitized solar cells. Chem Soc Rev 42(1):291–304
Wang C-L, Chang Y-C, Lan C-M, Lo C-F, Diau EW-G, Lin C-Y (2011) Enhanced light harvesting with π-conjugated cyclic aromatic hydrocarbons for porphyrin-sensitized solar cells. Energy Environ Sci 4(5):1788–1795
Luo J, Xu M, Li R, Huang K-W, Jiang C, Qi Q, Zeng W, Zhang J, Chi C, Wang P (2013) N-annulated perylene as an efficient electron donor for porphyrin-based dyes: enhanced light-harvesting ability and high-efficiency Co(II/III)-based dye-sensitized solar cells. J Am Chem Soc 136(1):265–272
Wang S-W, Wu K-L, Ghadiri E, Lobello MG, Ho S-T, Chi Y, Moser J-E, De Angelis F, Grätzel M, Nazeeruddin MK (2013) Engineering of thiocyanate-free Ru(II) sensitizers for high efficiency dye-sensitized solar cells. Chem Sci 4(6):2423–2433
Cao K, Lu J, Cui J, Shen Y, Chen W, Alemu G, Wang Z, Yuan H, Xu J, Wang M (2014) Highly efficient light harvesting ruthenium sensitizers for dye-sensitized solar cells featuring triphenylamine donor antennas. J Mater Chem A 2(14):4945–4953
Wang P, Klein C, Humphry-Baker R, Zakeeruddin SM, Grätzel M (2005) A high molar extinction coefficient sensitizer for stable dye-sensitized solar cells. J Am Chem Soc 127(3):808–809
El-Shafei A, Hussain M, Islam A, Han L (2014) Structure–property relationship of hetero-aromatic-electron-donor antennas of polypyridyl Ru(II) complexes for high efficiency dye-sensitized solar cells. Prog Photovoltaics Res Appl 22(9):958–969
Reynal A, Palomares E (2011) Ruthenium polypyridyl sensitisers in dye solar cells based on mesoporous TiO2. Eur J Inorg Chem 29:4509–4526
Reynal A, Forneli A, Palomares E (2010) Dye structure–charge transfer process relationship in efficient ruthenium-dye based dye sensitized solar cells. Energy Environ Sci 3(6):805–812
Chiba Y, Islam A, Watanabe Y, Komiya R, Koide N, Han L (2006) Dye-sensitized solar cells with conversion efficiency of 11.1 %. Jpn J Appl Phys 45(7L):L638
Wang Q, Campbell WM, Bonfantani EE, Jolley KW, Officer DL, Walsh PJ, Gordon K, Humphry-Baker R, Nazeeruddin MK, Grätzel M (2005) Efficient light harvesting by using green Zn-porphyrin-sensitized nanocrystalline TiO2 films. J Phys Chem B 109(32):15397–15409
Campbell WM, Jolley KW, Wagner P, Wagner K, Walsh PJ, Gordon KC, Schmidt-Mende L, Nazeeruddin MK, Wang Q, Grätzel M (2007) Highly efficient porphyrin sensitizers for dye-sensitized solar cells. J Phys Chem C 111(32):11760–11762
Bessho T, Zakeeruddin SM, Yeh CY, Diau EWG, Grätzel M (2010) Highly efficient mesoscopic dye-sensitized solar cells based on donor–acceptor-substituted porphyrins. Angew Chem Int Ed 49(37):6646–6649
Yella A, Mai CL, Zakeeruddin SM, Chang SN, Hsieh CH, Yeh CY, Grätzel M (2014) Molecular engineering of push-pull porphyrin dyes for highly efficient dye-sensitized solar cells: the role of benzene spacers. Angew Chem 126(11):3017–3021
Yasin A, Jose R, Yusoff MM (2015) Predicting larger absorption cross-section in porphyrin dyes using DFT calculations. J Porphyrins Phthalocyanines 19(12):1270–1278
Biroli AO, Tessore F, Vece V, Di Carlo G, Mussini P, Trifiletti V, De Marco L, Giannuzzi R, Manca M, Pizzotti M (2015) Highly improved performance of Zn II tetraarylporphyrinates in DSSCs by the presence of octyloxy chains in the aryl rings. J Mater Chem A 3(6):2954–2959
Magnano G, Marinotto D, Cipolla M, Trifiletti V, Listorti A, Mussini P, Di Carlo G, Tessore F, Manca M, Biroli AO (2016) Influence of alkoxy chain envelopes on the interfacial photoinduced processes in tetraarylporphyrin-sensitized solar cells. Phys Chem Chem Phys 18(14):9577–9585
Jacquemin D, Wathelet V, Perpete EA, Adamo C (2009) Extensive TD-DFT benchmark: singlet-excited states of organic molecules. J Chem Theory Comput 5(9):2420–2435
Sánchez-de-Armas R, San-Miguel MA, Oviedo J, Sanz JF (2012) Molecular modification of coumarin dyes for more efficient dye sensitized solar cells. J Chem Phys 136(19):194702
Martsinovich N, Troisi A (2011) High-throughput computational screening of chromophores for dye-sensitized solar cells. J Phys Chem C 115(23):11781–11792
Labat F, Le Bahers T, Ciofini I, Adamo C (2012) First-principles modeling of dye-sensitized solar cells: challenges and perspectives. Acc Chem Res 45(8):1268–1277
McNamara WR, Snoeberger Iii RC, Li G, Schleicher JM, Cady CW, Poyatos M, Schmuttenmaer CA, Crabtree RH, Brudvig GW, Batista VS (2008) Acetylacetonate anchors for robust functionalization of TiO2 nanoparticles with Mn(II)–terpyridine complexes. J Am Chem Soc 130(43):14329–14338
Gu X, Zhou L, Li Y, Sun Q, Jena P (2012) Design of new metal-free dyes for dye-sensitized solar cells: a first-principles study. Phys Lett A 376(38):2595–2599
Frisch M, Trucks G, Schlegel HB, Scuseria G, Robb M, Cheeseman J, Scalmani G, Barone V, Mennucci B, Ge Petersson (2009) Gaussian 09. Gaussian, Inc., Wallingford
Hay PJ, Wadt WR (1985) Ab initio effective core potentials for molecular calculations. Potentials for K to Au including the outermost core orbitals. J Chem Phys 82(1):299–310
Zhao Y, Truhlar DG (2008) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theoret Chem Acc 120(1–3):215–241
Barone V, Cossi M (1998) Quantum calculation of molecular energies and energy gradients in solution by a conductor solvent model. J Phys Chem A 102(11):1995–2001
Karthikeyan S, Lee JY (2013) Zinc-porphyrin based dyes for dye-sensitized solar cells. J Phys Chem A 117(42):10973–10979
Balanay MP, Kim DH (2008) DFT/TD-DFT molecular design of porphyrin analogues for use in dye-sensitized solar cells. Phys Chem Chem Phys 10(33):5121–5127
Lu T, Chen F (2012) Multiwfn: a multifunctional wavefunction analyzer. J Comput Chem 33(5):580–592
Ciofini I, Le Bahers T, Adamo C, Odobel F, Jacquemin D (2012) Through-space charge transfer in rod-like molecules: lessons from theory. J Phys Chem C 116(22):11946–11955
De Angelis F (2010) Direct vs. indirect injection mechanisms in perylene dye-sensitized solar cells: A DFT/TDDFT investigation. Chem Phys Lett 493(4):323–327
Delley B (2000) From molecules to solids with the DMol3 approach. J Chem Phys 113(18):7756–7764
Zeng W, Liu T, Wang Z, Tsukimoto S, Saito M, Ikuhara Y (2010) Oxygen adsorption on anatase TiO2 (101) and (001) surfaces from first principles. Mater Trans 51(1):171–175
Liu D-S, Ding W-L, Zhu K-L, Geng Z-Y, Wang D-M, Zhao X-L (2014) The master factors influencing the efficiency of D–A–π–A configurated organic sensitizers in dye-sensitized solar cell via theoretically characterization: design and verification. Dyes Pigm 105:192–201
Ding W-L, Wang D-M, Geng Z-Y, Zhao X-L, Xu W-B (2013) Density functional theory characterization and verification of high-performance indoline dyes with D–A–π–A architecture for dye-sensitized solar cells. Dyes Pigm 98(1):125–135
Fitri A, Benjelloun AT, Benzakour M, Mcharfi M, Hamidi M, Bouachrine M (2014) Theoretical investigation of new thiazolothiazole-based D–π–A organic dyes for efficient dye-sensitized solar cell. Spectrochim Acta Part A Mol Biomol Spectrosc 124:646–654
Yu P, Zhang F, Li M, He R (2015) Influence of position of auxiliary acceptor in D–A–π–A photosensitizes on photovoltaic performances of dye-sensitized solar cells. J Mater Sci 50(22):7333–7342. doi:10.1007/s10853-015-9290-8
Guo M, He R, Dai Y, Shen W, Li M, Zhu C, Lin SH (2012) Electron-deficient pyrimidine adopted in porphyrin sensitizers: a theoretical interpretation of π-spacers leading to highly efficient photo-to-electric conversion performances in dye-sensitized solar cells. J Phys Chem C 116(16):9166–9179
Yang L-N, Sun Z-Z, Chen S-L, Li Z-S (2013) The effects of various anchoring groups on optical and electronic properties of dyes in dye-sensitized solar cells. Dyes Pigm 99(1):29–35
Chen S-L, Yang L-N, Li Z-S (2013) How to design more efficient organic dyes for dye-sensitized solar cells? Adding more sp 2-hybridized nitrogen in the triphenylamine donor. J Power Sources 223:86–93
Li M, Kou L, Diao L, Zhang Q, Li Z, Wu Q, Lu W, Pan D, Wei Z (2015) Theoretical study of WS-9-based organic sensitizers for unusual vis/NIR absorption and highly efficient dye-sensitized solar cells. J Phys Chem C 119(18):9782–9790
Acknowledgements
We acknowledge the generous financial support from Natural Science Foundation of China (21173169, 20803059), and Chongqing Municipal Natural Science Foundation (cstc2013jcyjA90015).
Author information
Authors and Affiliations
Corresponding author
Additional information
Weixia Hu and Pei Yu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, W., Yu, P., Zhang, Z. et al. Theoretical study of YD2-o-C8-based derivatives as promising sensitizers for dye-sensitized solar cells. J Mater Sci 52, 1235–1245 (2017). https://doi.org/10.1007/s10853-016-0364-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-016-0364-z