Abstract
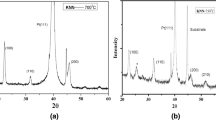
New nanosmooth potassium hexatitanate films have been prepared on crystalline Si (111) and ITO glass substrates by sol–gel method using Ti(n-OC4H9)4 and CH3COOK as precursors. Atomic force microscopy (AFM) topographic images were analyzed to select the optimal preparation conditions for the films. It is shown that the films consist of flat particles with the ratio of diameter to height around 11. The root mean square (RMS) roughness of the films based on the measurement of an area of 2,000 nm × 2,000 nm in AFM images is 6.4 nm. The crystal growth process of potassium hexatitanate film was characterized by XRD, Raman spectra, and TEM. The results showed that the crystal growth of potassium hexatitanate nanofilm is a confined growth mechanism. Electrochemical measurements demonstrated that the photocurrent of potassium hexatitanate film electrode is more stable than that of TiO2 film electrode.








Similar content being viewed by others
References
Urban JJ, Yun WS, Gu Q, Park H (2002) J Am Chem Soc 124:1186
Zhu HY, Lan Y, Gao XP, Ringer SP, Zheng ZF, Song DY, Zhao JC (2005) J Am Chem Soc 127:6730
Ma R, Fukuda K, Sasaki T, Osada M, Bando Y (2005) J Phys Chem B 109:6210
Du GH, Chen Q, Han PD, Yu Y, Peng LM (2003) Phys Rev 67:035323
Corcoran DJ, Tunstall DP, Irvine JT (2000) Solid State Ionics 136/137:297
Bao N, Lu X, Ji X, Feng X, Xie J (2002) Fluid Phase Equilib 193(1–2):229
Choy JH, Lee HC, Jung H, Huang SJ (2001) J Mater Chem 11:2232
He M, Feng X, Lu X, Ji X, Liu CH, Bao N, Xie J (2004) J Mater Sci 39:1
Ogawa M, Kuroda K (1995) Chem Rev 95:399
Yahya RB, Hayashi H, Nagase T, Ebina T, Onodera Y, Saitoh N (2001) Chem Mater 13:842
Yang Z, Bao N, Liu CH, Feng X, Xie J, Ji X, Lu X (2002) Chem J Chinese U 23:1371
Airdoldi C, Nunes LM, Farias RF (2000) Mater Res Bull 35:2081
Li JH, Ning XG, Ye HQ, Pan J, Fukunaga H (1997) J Mater Sci 32:543
Feng X, LV J, Lu X, Bao N, Ceng D (1999) Acta Mater Comp Sinica 16:1
Ogura S, Kohno M, Sato K, Inoue Y (1997) Appl Surf Sci 121/122:521
Lee HK, Shim JP, Shim MJ, Kim SW, Lee JS (1996) Mater Chem Phys 45:243
Barnes MC, Kumar S, Green L, Hwang NM, Andrea GR (2005) Surf Coat Technol 190(2–3):321
Sandell A, Anderson MP, Alfredsson Y (2002) J Appl Phys 92(6):3381
Hench LL, West JK (1990) Chem Rev 90(1):33
Sammelselg V, Rosental A, Tarre A (1998) Appl Surf Sci 134(1–4):78
Bao N, Feng X, Lu X, Shen L, Yanagisawa K (2004) AIChE J 50:1568
Yu L, He M, Liu C, Lu X, Feng X (2005) Mater Chem Phys 93:342
Liu C, Lu X, Yu G, Feng X, Zhang Q, Xu Z (2005) Mater Chem Phys 94:401
Bamberger CE, Begun GM, Macdougall CS (1990) Appl Spectrosc 44:30
Du GH, Chen Q, Han PD, Yu Y, Peng LM (2003) Phys Rev 67:035323
Byrne JA, Davidson A, Dunlop PSM, Eggins BR (2002) J Photochem Photobiol A: Chem 148:365
Gerischer H, Heller A (1991) J Phys Chem 95:5261
Leng WH, Zhang Z, Zhang JQ, Cao CN (2005) J Phys Chem B 109(31):5008
Acknowledgement
This work was supported by the National Natural Science Foundation of China (Grant Nos. 20246002 and 20236010), National High Technology Research and Development Program of China (No. 2003CB615700), the Key Science Foundation of Jiangsu Province, China (BK 2004215) and the Key Laboratory of Material-oriented Chemical Engineering of Ministry of Education, China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qian, Q.H., Zhou, X.F., Hu, Y.Y. et al. Preparation of smooth potassium hexatitanate nanofilms by sol–gel method. J Mater Sci 42, 8222–8229 (2007). https://doi.org/10.1007/s10853-007-1695-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-007-1695-6