Abstract
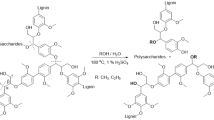
We report the effect of an organic solvent on the selective cleavage of individual lignin model compounds and lignin C–O linkages during the fast thermal pyrolysis of lignin. During this process, it was possible to lower the aliphatic hydroxyl contents of lignin, along with increasing the amount of single and double bonded aliphatics. It was found that the addition of solvent during fast pyrolysis of lignin lowered the molecular weight distribution of the obtained bio-oil (~ 49–52% decrease) and at the same time inhibited the formation of a high amount of char. A detailed study of the cleavage of complex model compounds using Ethanol-Assisted Fast Pyrolysis (EAFP) revealed that the aliphatic hydroxyl groups and etheric linkages are very reactive during this process. By the use of deuterated lignin model compounds and solvent, it was then possible to elucidate the mechanism for cleavage of lignin in the EAFP process that involves the formation of a transition state between solvent and oxygen bonds of lignin. This transition state involves the cleavage of etheric bonds by the in situ transfer of hydrogen from ethanol to this linkage.











Similar content being viewed by others
References
Dabral, S., Engel, J., Mottweiler, J., Spoehrle, S.S.M., Lahive, C.W., Bolm, C.: Mechanistic studies of base-catalysed lignin depolymerisation in dimethyl carbonate. Green Chem. 20(1), 170–182 (2018)
Lancefield, C.S., Panovic, I., Deuss, P.J., Barta, K., Westwood, N.J.: Pre-treatment of lignocellulosic feedstocks using biorenewable alcohols: towards complete biomass valorisation. Green Chem. 19(1), 202–214 (2017)
Wang, M., Li, L.H., Lu, J.M., Li, H.J., Zhang, X.C., Liu, H.F., Luo, N.C., Wang, F.: Acid promoted C–C bond oxidative cleavage of β-O-4 and β-1 lignin models to esters over a copper catalyst. Green Chem. 19(3), 702–706 (2017)
Koelewijn, S.-F., Van Den Bosch, S., Renders, T., Schutyser, W., Lagrain, B., Smet, M., Thomas, J., Dehaen, W., Van Puyvelde, P., Witters, H., Sels, B.F.: Sustainable bisphenols from renewable softwood lignin feedstock for polycarbonates and cyanate ester resins. Green Chem. 19(11), 2561–2570 (2017)
Paysepar, H., Rao, K.T.V., Yuan, Z., Nazari, L., Shui, H., Xu, C.: Zeolite catalysts screening for production of phenolic bio-oils with high contents of monomeric aromatics/phenolics from hydrolysis lignin via catalytic fast pyrolysis. Fuel Process. Technol. 178, 362–370 (2018)
Wu, Y., Wu, S., Zhang, H., Xiao, R.: Cellulose-lignin interactions during catalytic pyrolysis with different zeolite catalysts. Fuel Process. Technol. 179, 436–442 (2018)
Lui, M.Y., Lokare, K.S., Hemming, E., Stanley, J.N.G., Perosa, A., Selva, M., Masters, A.F., Maschmeyer, T.: Microwave-assisted methylation of dihydroxybenzene derivatives with dimethyl carbonate. RSC Adv. 6(63), 58443–58451 (2016)
Santos, J.H.P.M., Martins, M., Silvestre, A.J.D., Coutinho, J.A.P., Ventura, S.P.M.: Fractionation of phenolic compounds from lignin depolymerisation using polymeric aqueous biphasic systems with ionic surfactants as electrolytes. Green Chem. 18(20), 5569–5579 (2016)
Díaz-Urrutia, C., Chen, W.-C., Crites, C.-O., Daccache, J., Korobkov, I., Baker, R.T.: Towards lignin valorisation: comparing homogeneous catalysts for the aerobic oxidation and depolymerisation of organosolv lignin. RSC Adv. 5(86), 70502–70511 (2015)
Yang, H., Norinaga, K., Li, J., Zhu, W., Wang, H.: Effects of HZSM-5 on volatile products obtained from the fast pyrolysis of lignin and model compounds. Fuel Process. Technol. 181, 207–214 (2018)
Agarwal, A., Rana, M., Park, J.-H.: Advancement in technologies for the depolymerization of lignin. Fuel Process. Technol. 181, 115–132 (2018)
Li, H., Fang, Z., Smith, R.L., Yang, S.: Efficient valorization of biomass to biofuels with bifunctional solid catalytic materials. Prog. Energy Combust. Sci. 55, 98–194 (2016)
Schneider, L., Dong, Y., Haverinen, J., Jaakkola, M., Lassi, U.: Efficiency of acetic acid and formic acid as a catalyst in catalytical and mechanocatalytical pretreatment of barley straw. Biomass Bioenergy 91, 134–142 (2016)
Deng, W., Zhang, H., Wu, X., Li, R., Zhang, Q., Wang, Y.: Oxidative conversion of lignin and lignin model compounds catalyzed by CeO2 -supported Pd nanoparticles. Green Chem. 17(11), 5009–5018 (2015)
Byambajav, E., Paysepar, H., Nazari, L., Xu, C.: Co-pyrolysis of lignin and low rank coal for the production of aromatic oils. Fuel Process. Technol. 181, 1–7 (2018)
Ma, Z., Wang, J., Zhou, H., Zhang, Y., Yang, Y., Liu, X., Ye, J., Chen, D., Wang, S.: Relationship of thermal degradation behavior and chemical structure of lignin isolated from palm kernel shell under different process severities. Fuel Process. Technol. 181, 142–156 (2018)
Cui, K., Yang, L., Ma, Z., Yan, F., Wu, K., Sang, Y., Chen, H., Li, Y.: Selective conversion of guaiacol to substituted alkylphenols in supercritical ethanol over MoO3. Appl. Catal. B Environ. 219, 592–602 (2017)
Sturgeon, M.R., O’Brien, M.H., Ciesielski, P.N., Katahira, R., Kruger, J.S., Chmely, S.C., Hamlin, J., Lawrence, K., Hunsinger, G.B., Foust, T.D., Baldwin, R.M., Biddy, M.J., Beckham, G.T.: Lignin depolymerisation by nickel supported layered-double hydroxide catalysts. Green Chem. 16(2), 824–835 (2014)
Zhang, J., Lu, G., Cai, C.: Self-hydrogen transfer hydrogenolysis of β-O-4 linkages in lignin catalyzed by MIL-100(Fe) supported Pd-Ni BMNPs. Green Chem. 2, 4538–4543 (2017)
da Costa Sousa, L., Jin, M., Chundawat, S.P.S., Bokade, V., Tang, X., Azarpira, A., Lu, F., Avci, U., Humpula, J., Uppugundla, N., Gunawan, C., Pattathil, S., Cheh, A.M., Kothari, N., Kumar, R., Ralph, J., Hahn, M.G., Wyman, C.E., Singh, S., Simmons, B.A., Dale, B.E., Balan, V.: Next-generation ammonia pretreatment enhances cellulosic biofuel production. Energy Environ. Sci. 9(4), 1215–1223 (2016)
Chen, W., McClelland, D.J., Azarpira, A., Ralph, J., Luo, Z., Huber, G.W.: Low temperature hydrogenation of pyrolytic lignin over Ru/TiO 2: 2D HSQC and 13 C NMR study of reactants and products. Green Chem. 18(1), 271–281 (2016)
Behling, R., Valange, S., Chatel, G.: Heterogeneous catalytic oxidation for lignin valorization into valuable chemicals: what results? What limitations? What trends? Green Chem. 18(7), 1839–1854 (2016)
Dayton, D.C., Hlebak, J., Carpenter, J.R., Wang, K., Mante, O.D., Peters, J.E.: Biomass hydropyrolysis in a fluidized bed reactor. Energy Fuels 30(6), 4879–4887 (2016)
Custodis, V.B.F., Hemberger, P., Ma, Z., Van Bokhoven, J.A.: Mechanism of fast pyrolysis of lignin: studying model compounds. J. Phys. Chem. B 118(29), 8524–8531 (2014)
Wang, S., Dai, G., Yang, H., Luo, Z.: Lignocellulosic biomass pyrolysis mechanism: a state-of-the-art review. Prog. Energy Combust. Sci. 62, 33–86 (2017)
He, T., Zhang, Y., Zhu, Y., Wen, W., Pan, Y., Wu, J., Wu, J.: Pyrolysis mechanism study of lignin model compounds by synchrotron vacuum ultraviolet photoionization mass spectrometry. Energy Fuels 30(3), 2204–2208 (2016)
Jarvis, M.W., Daily, J.W., Carstensen, H.H., Dean, A.M., Sharma, S., Dayton, D.C., Robichaud, D.J., Nimlos, M.R.: Direct detection of products from the pyrolysis of 2-phenethyl phenyl ether. J. Phys. Chem. A 115(4), 428–438 (2011)
Mukkamala, S., Wheeler, M.C., Van Heiningen, A.R.P., Desisto, W.J.: Formate-assisted fast pyrolysis of lignin. Energy Fuels 26(2), 1380–1384 (2012)
Zhou, S., Brown, R.C., Bai, X.: The use of calcium hydroxide pretreatment to overcome agglomeration of technical lignin during fast pyrolysis. Green Chem. 17(10), 4748–4759 (2015)
Tarves, P.C., Mullen, C.A., Strahan, G.D., Boateng, A.A.: Depolymerization of lignin via co-pyrolysis with 1,4-butanediol in a microwave reactor. ACS Sustain. Chem. Eng. 5(1), 988–994 (2017)
Fortin, M., Mohadjer Beromi, M., Lai, A., Tarves, P.C., Mullen, C.A., Boateng, A.A., West, N.M.: Structural analysis of pyrolytic lignins isolated from switchgrass fast-pyrolysis oil. Energy Fuels 29(12), 8017–8026 (2015)
Kan, T., Strezov, V., Evans, T.J.: Lignocellulosic biomass pyrolysis: a review of product properties and effects of pyrolysis parameters. Renew. Sustain. Energy Rev. 57, 1126–1140 (2016)
Beste, A., Buchanan, A.C.: Kinetic analysis of the phenyl-shift reaction in??-O-4 lignin model compounds: a computational study. J. Org. Chem. 76(7), 2195–2203 (2011)
Beste, A., Buchanan, A.C.: Computational study of bond dissociation enthalpies for lignin model compounds. substituent effects in phenethyl phenyl ethers. J. Org. Chem. 74(7), 2837–2841 (2009)
Song, Q., Wang, F., Xu, J.: Hydrogenolysis of lignosulfonate into phenols over heterogeneous nickel catalysts. Chem. Commun. 48(56), 7019 (2012)
Mottweiler, J., Rinesch, T., Besson, C., Buendia, J., Bolm, C.: Iron-catalysed oxidative cleavage of lignin and β-O-4 lignin model compounds with peroxides in DMSO. Green Chem. 17(11), 5001–5008 (2015)
Kleine, T., Buendia, J., Bolm, C.: Mechanochemical degradation of lignin and wood by solvent-free grinding in a reactive medium. Green Chem. 15(1), 160–166 (2013)
Acknowledgements
“Australian Institute for Bioengineering and Nanotechnology (AIBN) at The University of Queensland, Australia” has financially supported this research. The authors also acknowledge the facilities, the scientific and technical assistance of “Australian National Fabrication Facility” (ANFF) at The University of Queensland. The first author gratefully appreciates the University of Queensland international (UQI) scholarship and candidature travel award (CTA).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rashidi, M., Beltramini, J.N. & Martin, D. The selective cleavage of lignin aliphatic C–O linkages by solvent-assisted fast pyrolysis (SAFP). J Incl Phenom Macrocycl Chem 94, 297–307 (2019). https://doi.org/10.1007/s10847-019-00905-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-019-00905-x