Abstract
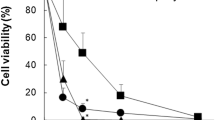
Methyl-β-cyclodextrin (M-β-CyD) is widely used as a raft disrupting agent through extraction of cholesterol from lipid rafts which are highly expressed in cell membranes of tumor cells, but it does not have tumor cell-selective action. Meanwhile, the widespread use of folic acid (FA) as a tumor-targeting ligand has been known, because folate receptor (FR) overexpresses in various kinds of epithelial tumor cells. In the present study, in order to obtain more tumor cell-selectivity and antitumor activity of M-β-CyD, we designed folate-appended M-β-CyD (FA-M-β-CyD), and evaluated its physicochemical properties and antitumor activity. The 1H-NMR study demonstrated that FA-M-β-CyD having average degree of substitution of FA (DSF) of 1.0 was prepared. In addition, FA-M-β-CyD (DSF 1.0) was found to be amorphous in a solid state and surface-active. Importantly, FA-M-β-CyD (DSF 1.0) had potent cytotoxicity, compared to M-β-CyD in KB cells, but not in A549 cells. These results suggest that FA-M-β-CyD (DSF 1.0) has the potential as a novel antitumor agent.






Similar content being viewed by others
References
O’Brien, M.E., Wigler, N., Inbar, M., Rosso, R., Grischke, E., Santoro, A., Catane, R., Kieback, D.G., Tomczak, P., Ackland, S.P., Orlandi, F., Mellars, L., Alland, L., Tendler, C.: Reduced cardiotoxicity and comparable efficacy in a phase III trial of pegylated liposomal doxorubicin HCl (CAELYX/Doxil) versus conventional doxorubicin for first-line treatment of metastatic breast cancer. Ann. Oncol. 15, 440–449 (2004)
Harata, M., Soda, Y., Tani, K., Ooi, J., Takizawa, T., Chen, M., Bai, Y., Izawa, K., Kobayashi, S., Tomonari, A., Nagamura, F., Takahashi, S., Uchimaru, K., Iseki, T., Tsuji, T., Takahashi, T.A., Sugita, K., Nakazawa, S., Tojo, A., Maruyama, K., Asano, S.: CD19-targeting liposomes containing imatinib efficiently kill Philadelphia chromosome-positive acute lymphoblastic leukemia cells. Blood 104, 1442–1449 (2004)
Roche, A.C., Fajac, I., Grosse, S., Frison, N., Rondanino, C., Mayer, R., Monsigny, M.: Glycofection: facilitated gene transfer by cationic glycopolymers. Cell. Mol. Life Sci. 60, 288–297 (2003)
Chen, H., Ahn, R., Van den Bossche, J., Thompson, D.H., O’Halloran, T.V.: Folate-mediated intracellular drug delivery increases the anticancer efficacy of nanoparticulate formulation of arsenic trioxide. Mol. Cancer. Ther. 8, 1955–1963 (2009)
Lu, Y., Low, P.S.: Folate-mediated delivery of macromolecular anticancer therapeutic agents. Adv. Drug. Deliv. Rev. 54, 675–693 (2002)
Hatakeyama, H., Akita, H., Maruyama, K., Suhara, T., Harashima, H.: Factors governing the in vivo tissue uptake of transferrin-coupled polyethylene glycol liposomes in vivo. Int. J. Pharm. 281, 25–33 (2004)
Miyajima, Y., Nakamura, H., Kuwata, Y., Lee, J.D., Masunaga, S., Ono, K., Maruyama, K.: Transferrin-loaded nido-carborane liposomes: tumor-targeting boron delivery system for neutron capture therapy. Bioconjug. Chem. 17, 1314–1320 (2006)
Kim, I.Y., Kang, Y.S., Lee, D.S., Park, H.J., Choi, E.K., Oh, Y.K., Son, H.J., Kim, J.S.: Antitumor activity of EGFR targeted pH-sensitive immunoliposomes encapsulating gemcitabine in A549 xenograft nude mice. J. Control. Release 140, 55–60 (2009)
Schiffelers, R.M., Koning, G.A., ten Hagen, T.L., Fens, M.H., Schraa, A.J., Janssen, A.P., Kok, R.J., Molema, G., Storm, G.: Anti-tumor efficacy of tumor vasculature-targeted liposomal doxorubicin. J. Control. Release 91, 115–122 (2003)
Leamon, C.P., Low, P.S.: Folate-mediated targeting: from diagnostics to drug and gene delivery. Drug Discov. Today 6, 44–51 (2001)
Parker, N., Turk, M.J., Westrick, E., Lewis, J.D., Low, P.S., Leamon, C.P.: Folate receptor expression in carcinomas and normal tissues determined by a quantitative radioligand binding assay. Anal. Biochem. 338, 284–293 (2005)
Szente, L., Szejtli, J.: Highly soluble cyclodextrin derivatives: chemistry, properties, and trends in development. Adv. Drug Deliv. Rev. 36, 17–28 (1999)
Uekama, K., Otagiri, M.: Cyclodextrins in drug carrier systems. Crit. Rev. Ther. Drug Carrier Syst. 3, 1–40 (1987)
Motoyama, K., Arima, H., Toyodome, H., Irie, T., Hirayama, F., Uekama, K.: Effect of 2, 6-di-O-methyl-α-cyclodextrin on hemolysis and morphological change in rabbit’s red blood cells. Eur. J. Pharm. Sci. 29, 111–119 (2006)
Motoyama, K., Toyodome, H., Onodera, R., Irie, T., Hirayama, F., Uekama, K., Arima, H.: Involvement of lipid rafts of rabbit red blood cells in morphological changes induced by methylated β-cyclodextrins. Biol. Pharm. Bull. 32, 700–705 (2009)
Ohtani, Y., Irie, T., Uekama, K., Fukunaga, K., Pitha, J.: Differential effects of α-, β- and γ-cyclodextrins on human erythrocytes. Eur. J. Biochem. 186, 17–22 (1989)
Galbiati, F., Razani, B., Lisanti, M.P.: Emerging themes in lipid rafts and caveolae. Cell 106, 403–411 (2001)
Simons, K., Ehehalt, R.: Cholesterol, lipid rafts, and disease. J. Clin. Invest. 110, 597–603 (2002)
Anderson, R.G., Jacobson, K.: A role for lipid shells in targeting proteins to caveolae, rafts, and other lipid domains. Science 296, 1821–1825 (2002)
Motoyama, K., Kameyama, K., Onodera, R., Araki, N., Hirayama, F., Uekama, K., Arima, H.: Involvement of PI3K-Akt-Bad pathway in apoptosis induced by 2, 6-di-O-methyl-β-cyclodextrin, not 2, 6-di-O-methyl-α-cyclodextrin, through cholesterol depletion from lipid rafts on plasma membranes in cells. Eur. J. Pharm. Sci. 38, 249–261 (2009)
Grosse, P.Y., Bressolle, F., Pinguet, F.: Antiproliferative effect of methyl-β-cyclodextrin in vitro and in human tumour xenografted athymic nude mice. Br. J. Cancer 78, 1165–1169 (1998)
Arima, H., Chihara, Y., Arizono, M., Yamashita, S., Wada, K., Hirayama, F., Uekama, K.: Enhancement of gene transfer activity mediated by mannosylated dendrimer/α-cyclodextrin conjugate (generation 3, G3). J. Control. Release 116, 64–74 (2006)
Wada, K., Arima, H., Tsutsumi, T., Chihara, Y., Hattori, K., Hirayama, F., Uekama, K.: Improvement of gene delivery mediated by mannosylated dendrimer/α-cyclodextrin conjugates. J. Control. Release 104, 397–413 (2005)
Hamasaki, K., Kogure, K., Ohwada, K.: A biological method for the quantitative measurement of tetrodotoxin (TTX): tissue culture bioassay in combination with a water-soluble tetrazolium salt. Toxicon. 34, 490–495 (1996)
Ono, N., Arima, H., Hirayama, F., Uekama, K.: A moderate interaction of maltosyl-α-cyclodextrin with Caco-2 cells in comparison with the parent cyclodextrin. Biol. Pharm. Bull. 24, 395–402 (2001)
Takahashi, K., Hattori, K., Toda, F.: Monotosylated α- and β-cyclodextrins prepared in an alkaline aqueous solution. Tetrahedron Lett. 25, 3331–3334 (1984)
Murakami, T., Harata, K., Morimoto, S.: Synthesis and chiral recognition property of 3-acetylamino-3-deoxy-β-cyclodextrin. Chem. Lett. 17, 553–556 (1988)
Acknowledgments
This work was partially supported by a Grant-in-Aid from Young Scientists (B) from the Ministry of Education, Science and Culture of Japan (22790040).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Onodera, R., Motoyama, K. & Arima, H. Design and evaluation of folate-appended methyl-β-cyclodextrin as a new antitumor agent. J Incl Phenom Macrocycl Chem 70, 321–326 (2011). https://doi.org/10.1007/s10847-010-9843-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-010-9843-z