Abstract
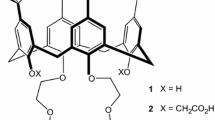
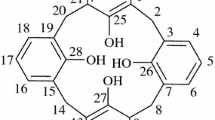
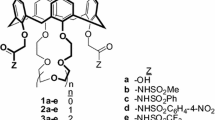
A regioisomer of a previously reported p-tert-butylcalix[4]arene-1,3-thiacrown-5 di(carboxylic acid) is prepared in which the thiacrown ring attachment sites are moved from distal phenolic oxygens of a cone p-tert-butylcalix[4]arene scaffold to proximal phenolic oxygens. The influence of this structural variation in the di-ionizable calixcrown ligand on competitive solvent extractions of alkali metal cations and of alkaline earth metal cations and single species solvent extractions of Hg2+ and Pb2+ from aqueous solutions into chloroform is evaluated.








Similar content being viewed by others
References
Asfari, Z., Wagner, S., Vicens, J.: Calixcrowns and related molecules. J. Incl. Phenom. Mol. Recognit. Chem. 19, 137–148 (1994)
Casnati, A., Ungaro, R., Asfari, Z., Vicens, J.: Crown ethers derived from calix[4]arenes. In: Asfari, A., Böhmer, V., Harrowfield, J., Vicens, J. (eds.) Calixarenes 2001, pp. 365–384. Kluwer, Dordrecht (2001)
Gutsche, C.D.: Calixarenes. An Introduction, 2nd edn, pp. 104–107. Royal Society of Chemistry, Cambridge (2008)
Silorinne, K., Nissinen, M.: J. Incl. Phenom. Macrocycl. Chem. 61, 11–27 (2008)
Sliwa, W., Kozlowski, C.: Calixarenes and Resorcinarenes. Synthesis, Properties and Applications, pp. 165–174. Wiley-VCH Verlag, Weinheim (2009)
Kim, J.S., Vicens, J.: Progress of calixcrowns chemistry. J. Incl. Phenom. Macrocycl. Chem. 63, 189–193 (2009)
Alfieri, C., Dradi, E., Pochini, A., Ungaro, R., Andreeti, B.D.: Synthesis and x-ray crystal and molecular structure of a novel macrobicyclic ligand: crowned p-tert-butylcalix[4]arene. J. Chem. Soc., Chem. Commun. 1075–1077 (1983)
Arduini, A., Pochini, A., Ungaro, R.: p-tert-Butylcalix[4]arene tetracarboxylic acid. A water soluble calixarene in a cone conformation. J. Chem. Soc., Chem. Commun. 981–982 (1984)
Ogata, M., Fujimoto, K., Shinkai, S.: Molecular design of calix[4]arene-based extractants which show high Ca2+ selectivity. J. Am. Chem. Soc. 116, 4505–4506 (1995)
Talanov, V.S., Talanova, G.G., Gorbunova, M.G., Bartsch, R.A.: Novel cesium selective, 1, 3-alternate calix[4]arene-bis(crown-6-ethers) with proton-ionizable groups for enhanced extraction efficiency. J. Chem. Soc., Perkin Trans. 2, 209–215 (2002)
Tu, C., Surowiec, K., Bartsch, R.A.: Novel calix[4]arene-thiacrown ether for selective and efficient extraction of Ba(II), Pb(II) and Hg(II). J. Incl. Phenom. Macrocycl. Chem. 58, 361–366 (2007)
Mass, G.E., Bradshaw, J.S., Izatt, R.M., Christensen, J.J.: Synthesis of a new series of macrocyclic polyether–diester ligands. J. Org. Chem. 42, 3937–3941 (1977)
Ouchi, M., Inoue, Y., Kanzaki, T., Hakushi, T.: Molecular design of crown ethers. 1. Effect of methylene chain length: 15- to 17-crown-5 and 18- to 22-crown-6. J. Org. Chem. 49, 1408–1412 (1984)
Yamamoto, H., Sakaki, T., Shinkai, S.: Regioselective synthesis of 1,2- and 1,3-bridged calix[4]crowns. What are the factors controlling regioselectivity? Chem. Lett. 469–472 (1994)
Acknowledgment
We thank the Division of Chemical Sciences, Geosciences and Biosciences of the Office of Basic Energy Sciences of the U.S. Department of Energy (Grant DE-FG02-90ER14416) for support of this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, Y., Cao, X., Surowiec, K. et al. Calix[4]arene-thiacrown-5 di(carboxylic acid) regioisomers as metal ion extractants. J Incl Phenom Macrocycl Chem 66, 163–169 (2010). https://doi.org/10.1007/s10847-009-9675-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-009-9675-x