Abstract
The artificial light sources are useful tools for sampling night active insects, however, they also possess potential environmental risks in their habitats. To test their applicability and evaluate environmental risk for caddisflies (Trichoptera), the attractivity of different portable light traps working with LED, UV and mixed-white light sources was studied and compared with attractivity of traditionally used mercury-vapour lamp (in Jermy-type light trap), which is tool of sampling and street-light. Analysing 1135 caught individuals of 19 species light sources emitting different wavelength spectra showed different attractivity and selectivity on caddisflies both on species and family levels. Attractivity of mercury-vapour lamp was generally lower than the other tested light sources. We found that the most attractive wavelength range for caddisflies is between 360 and 407 nm. One of the tested LED and mixed-white lamps together could cover this spectrum and a high and wide spectral peak of mixed-white light source between 375 and 391 nm resulted additional catches considering both species and number of individuals. Lamps emitting between 360 and 407 nm may be both a useful tool for sampling caddisflies and dangerous source of light pollution along lowland water courses where the sampled species are common and widespread.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Traps using different light sources are traditionally used in both basic and applied entomological surveys since they can attract wide range of insects with positive phototaxis (Williams 1939; Blanton et al. 1955; Hartstack et al. Jr 1968; Szentkirályi 2002) e.g. species of Lepidoptera (Baker and Sadovy 1978; Maelzer et al. 1996; Jonason et al. 2014; Tikoca et al. 2016), Coleoptera (Kádár and Szél 1995; Járfás and Tóth 1977) and Trichoptera (Waringer 1989, 1991; Schmera 2003; Stanić-Koštroman et al. 2012; Larsson et al. 2020). The order of Trichoptera is the most species rich taxa of primarily aquatic insects (Adler and Foottit 2017; Morse 2017) and they play essential role in food webs both of freshwater and neighbouring terrestrial ecosystems providing food source for many predatory taxa (e.g., fish, crayfish, birds, bats) (Jackson and Resh 1989; Morse 2017). Considering these features, they are commonly used indicators of water quality, which are sensitive especially for high sediment and nutrient concentrations (Resh and Unzicker 1975; Lenat 1993; Resh 1993; Barbour et al. 1999; Dohet 2002; Jehamalar et al. 2010).
Depending on targeted insect taxa, aim of the given investigation and characteristics (e.g., climatic conditions, habitat structure) of the sampling site, we can choose among plenty of trap types and light sources with different operating- and colour temperature and spectral distribution. There are quite a lot of data on the effects of weather (temperature, wind strength, humidity, air pressure, etc.), moon phases and other environmental factors on the efficiency of light traps (Kiss 2002; Nowinszky 2003; Nowinszky et al. 2014), while spectral distribution is one of the most important parameters affecting the attractivity and selectivity of light sources (Nowinszky 2003). Light sources emitting relatively large amount of UV light generally perform better in catching of night-active insects than others (Blomberg et al. 1976; Ashfaq et al. 2005; Cowan and Gries 2009), since beyond the visibility and the spectral distribution of the lamp, at least a minimal amounts of UV radiation are a crucial factor of the attractiveness (Barghini 2008, Barghini and Medeiros 2012).
The most widely used mercury-vapour lamp is an appropriate tool for attracting species of Lepidoptera (van Langevelde et al. 2011; Bates et al. 2013; Brehm 2017; Infusino et al. 2017), Coleoptera (Kádár and Szél 1995; Band et al. 2019) and Trichoptera (Ward et al. 1996; Kiss 2002; Nowinszky 2003; Blahnik and Holzenthal 2004; Calor and Mariano 2012) and even Hemiptera, Hymenoptera, Diptera and Neuroptera (Ramamurthy et al. 2010; van Grunsven et al. 2014). Mercury-vapour lamps attract more species and individuals of moths than the UV light alone probably since they have high peaks both in the UV and in the visible spectrum (Jonason et al. 2014; Tikoca et al. 2016; Brehm 2017). In case of Lepidoptera blacklight (BL) tube (Belton and Kempster 1963; Mészáros 1966; Bhandari et al. 2017) and LED lights with wide spectra including UV radiation (Brehm 2017; Infusino et al. 2017) can be also effective. For attracting mosquitos, the incandescent lights, for agricultural pests the green lights, and for some aquatic insects the cyalume lightsticks are the most commonly used light sources (Epsky et al. 2008).
The high attractivity of the mercury-vapour lamp for caddisflies (Mikkola 1972; Blomberg 1976; Walker and Galbreath 1979) and the role of its UV spectra were also proven (e.g., Chernyshev 1961; Calor and Mariano 2012; Pohe et al. 2017), thus this trap type and black light lamps became the most widely used standard light source types in caddisfly sampling (Blahnik and Holzenthal 2004; Calor and Mariano 2012). According to Mikkola (1972) the eyes of Trichoptera are similar to Lepidoptera and sensitive for 350–600 nm wavelength, with a maximum around 550 nm, but the effect of spectral distribution of light sources on caddisflies is poorly known.
Although it is one of the most effective tools for sampling many taxa mercury-vapour lamps have been phased out in Europe since April 2005 as per EU Regulation 245/2009, which favours more cost-effective and energy-efficient LED technology (van Grunsven et al. 2014; Donatello et al. 2019; Haddock et al. 2019). Furthermore, the manufacturing and trade of mercury lamps have been gradually prohibited worldwide since 2020 by Minamata Convention Agreement (www.mercuryconvention.org), thus it should be replaced also in entomological studies.
Here we compared the efficiency and selectivity of different light sources to find the most appropriate method and tool for the caddisfly sampling and establish the most attractive wavelength range for them using mercury vapour lamp as kind of “traditional control method”. Beyond that we provided recommendations on use of different artificial lights considering conservation of caddisfly fauna. Finally large amount of distribution data of caddisfly species was also collected in a mainly unknown underinvestigated region of the Carpathian Lowland (Transcarpathia, W Ukraine).
Materials and methods
Sampling site
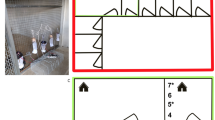
The samplings were carried out in the Velyka Dobron’ Game Reserve, in the eastern part of the Bereg Plain (West Ukraine) (Fig. 1). The Bereg Plain belongs to the Upper Tisza Region on the northeastern part of the Pannonian Lowland. This area has cool, humid continental climate strongly affected by the nearby Carpathians (Simon 1953).
Map of the sampling sites with location of the Jermy-type light trap with mercury-vapour lamp (A; empty triangle) and portable light traps (B; empty circle) with different light sources in the surroundings of Velyka Dobron’. black: rivers, channels, and other water bodies; dark grey: forests; middle grey: semi-natural grasslands; light grey: arable lands
The study area was the margin of the former Szernye Marsh thus it is still water-logged and wet despite the drainage (Szanyi et al. 2015). The Szernye Canal constructed during the drainage of the marsh and the Latorca River flow through the study area. On the floodplain, these watercourses provide a permanent water supply with a maximum in spring. Beyond that, the reserve is surrounded by many artificial canals containing water periodically, and ephemeral ponds and puddles.
The light traps were placed on the banks of the Szernye Canal, near to the Latorca River in order that samplings represents both artificial and natural waterflows of the region.
Sampling method
Between 11th August and 9th September 2018 14 samplings were made with six portable light traps and one Jermy-type light-trap. The Jermy-type light trap worked with a mercury-vapour bulb (80 W Hg), while the portable light traps contained different light sources: one mixed-white light tube, 2 types of LED and 3 types of UV light tubes with different spectral distribution. The spectral distribution of the tested light sources were measured with a Red Tide spectrophotometer working with a linear silicon CCD (Charge Coupled Device) detector. It gives the wavelengths in nanometres and describes the intensity with counts (Fig. 2).
The mercury-vapour lamp (MV) had many peak wavelengths in a wide range (around 366, 405, 408, 436, 492, 547, 578, 594, 621, 697 nm-s). The mixed-white light source (W) had high peaks both in the visible (around 405, 436, 546 nm-s) and in the UV spectrum (around 370 nm). The UV1 and UV2 light sources were similar, and they showed only minor peaks in the visible compared to the UV spectrum. Their main peaks were similar with those of W light source (around 370 nm), but narrower. The peaks of the UV1 were higher in the UV range, while the peaks of the UV2 were higher in the visible spectrum. UV3 covered almost the whole UV-A spectrum, but only with low intensity without high peaks. The two tested LED light sources had similar peaks in the blue spectrum range (around 395 nm), although the peak of the L2 light source was slightly wider (Fig. 2).
Location of the mercury-vapour lamps are limited by available grid power, or they can work with a generator. The small portable light traps can be easily located at different habitats, since they work with 12 V batteries. These portable light traps were placed about 100 m distance from each other along the banks of the Szernye Canal. To eliminate the effect of sampling sites they were relocated site by site in each sampling time. Portable light traps were smaller versions of the Robinson-type light trap (Upton and Mantle 2010). The light tubes were mounted vertically with the top of a large funnel and have three radiating baffles around it. The funnel fitted on a plastic bucket, and had a transparent cover mounted above it to keep the rain out. The structure of the Jermy-type light trap is described in Szentkirályi (2002).
The collected insects were stored in 70 V/V% ethanol till identification carried out in laboratory.
Data analyses
The caught caddisflies were identified based on the keys of Malicky (2004). The nomenclature of Nógrádi and Uherkovich (2002) was followed. In case of Hydropsyche spp. males were identified at species level, while females were identified only at genera level since the extreme similarity of the female genitalia of different species.
The fauna was characterised with checklist and relative frequencies of the caught species and families. To characterise efficiency and selectivity of the tested light sources the total and mean number of species and individuals (species/ and individuals/sample) were calculated, and the number of unique species (species attracted only by a given light source) also was provided by light sources. The mean number of caught individuals (individual/samples) was also calculated both for caddisfly families and species. To evaluate how different light sources sample diversity Shannon-Wiener and Hill’s diversity indices were calculated and compared by different methods. For calculation of indices an online diversity calculator developed by Young T. M. was used (alyoung.com/labs/biodiversity_calculator.html).
Light sources were compared using the above variables. For comparisons Kruskal-Wallis test was used since data didn’t fulfil the assumptions of parametric tests. If the test showed significant differences the pairwise comparison was made with Mann-Whitney U-test using Statistica 7 program.
Results
Totally 1135 individuals of 19 caddisfly species belonging to six families were caught by the tested light traps. The most species rich family were the Hydropsychidae with 6 species. Leptoceridae family showed similarly large species richness, however the abundances of species were especially low. The most abundant family was also the Hydropsychidae followed by Limnephilidae. Based on male catches the most abundant Hydropsychidae species was Hydropsyche modesta followed Hydropsyche contubernalis and Hydropsyche bulgaromanorum. Among Limnephilidae Glyphotaelius pellucidus was dominant. Only one or two species belonging to Psychomyiidae, Ecnomidae and Phryganeidae families was caught only with low abundances (Table 1).
Traps caught both sexes with near 1:1 ratio, since the mean percentage of males was 45.5 (± 18.3) % (Table 1). Species showed different sex ratios. Ecnomus tenellus and Hydropsyche spp. showed 1:4 and 1:3, contrary Agrypnia varia and Limnephilus flavicornis showed 3:1 and 3:2 ratios. Ratios of other species were average (Table 1).
Although mixed-white light (W) attracted almost all the species registered during the study (17/19, except Setodes punctatus and Oecetis furva) and the number of caught species varied between 9 and 17, but the efficiency provided by mean number of caught species per sample did not differ (KW: H(6;98) = 4.8261, p = 0.5663). Considering species number, the less efficient light sources were the UV1 and the mercury-vapour lamps (Table 2). Using diversity indices different pattern was found. Based on mean value of Shannon-Wiener and Hill’s diversity indexes of samples there were no significant differences between the tested sampling methods (KW: H(6;98) = 2.8543–4.489, p = 0.6108–0.8911), but mercury-vapour lamp showed the largest values followed by mixed-white light source. In this point of view LED and UV lamps were less effective (Table 3).
The number of individuals attracted by tested light sources differed between a smaller range (127–183) and the mean number of caught individuals also did not show significant differences (KW: H(6;98) = 2.6317, p = 0.8534) (Table 2).
During the study only three species showed light source specific presence (Table 2). Two especially rare Hydropsyche species (H. ornatula and H. pellucidula) were attracted only with the mixed-white light source (W), while the also rare O. furva was attracted only with L1 light. Beyond that, specimens of Mystacides azureus were caught only with mixed-white (W) and UV1 traps while individuals of S. punctatus were caught only with mercury-vapour (MV) and UV2 light sources. Other species were attracted with at least three types of tested light sources.
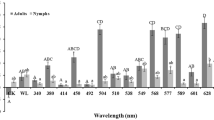
Considering the number of caught individuals the selectivity of light sources could be detected both on family and species level. The mercury-vapour light source attracted significantly more individuals of Leptoceridae than other light sources except mixed-white light bulb, but the difference was conspicuous also in this case. Contrary, in case of Limnephilidae, Phryganeidae and Psychomyiidae families mercury-vapour light source attracted less individuals than the other light sources. In case of Limnephilidae the differences were significant in all comparisons. In case of Phryganeidae mercury-vapour lamp attracted significantly less individuals than mixed-white and UV (UV1-3) lights, while the differences were not significant in case of L1 and L2 lights. Considering Psychomyiidae UV3 light source attracted the most individuals, and the differences were significant in case of UV1, UV2 and mercury-vapour lights. In case of Ecnomidae and Hydropsychidae there were no remarkable differences between the selectivity of the tested light sources (Fig. 3).
The attraction of the caddisfly families to different light sources based on the number of caught individuals (individuals/sample). Small letters indicate significant differences between light sources based on Mann-Whitney U-test, P < 0.05. MV: mercury-vapour lamp; W: mixed-white light tube; L1, L2: LED lights; UV1, UV2, UV3: UV light tubes
Among 19 caught species, 10 were caught with more than 10 individuals. Considering mean number of caught individuals three of them were differently attracted by different light sources. UV1 light attracted the most T. minor individuals. Traps with UV1, UV2 and mixed-white lights caught significantly more individuals than traps with mercury-vapour bulb that did not catch any individuals. Individuals of G. pellucidus attracted by all tested light sources except mercury-vapour bulb, that attracted significantly less individuals than others. Contrary, in case of Oecetis notata mercury-vapour light bulb attracted significantly more individuals than other light sources from which L1, UV1 and UV3 bulbs did not attract any individuals (Table 4). In case of the other seven species obvious specificity could not be detected.
Discussion
In the present investigation the mercury-vapour lamp (MV) seems to be less effective than the other studied light sources. It attracted the least number of individuals and less species than the most UV, LED and mixed-white light sources. Additionally, 9 species: Cheumatopsyche lepida, Hydropsyche ornatula, Psychomyia pusilla, Grammotaulius nigropunctatus, Mystacides azureus, Hydropsyche pellucidula, Lype phaeopa, Trichostegia minor and Oecetis furva were collected only with alternative light sources and the last 4 of them have not been caught formerly, during the 3-year samplings with mercury-vapour lamp. (Szanyi and Szanyi 2018, 2019). Researchers generally use mercury-vapour lamps and black light lamps for collecting caddisflies and also moths (Blahnik and Holzenthal 2004; Calor and Mariano 2012) since the attraction of caddisflies to near-ultraviolet radiation has been known for a long time (Chernyshev 1961; Mikkola 1972; Blomberg 1976; Walker and Galbreath 1979; Pohe et al. 2017). Although between them mercury-vapour lamps are seen more efficient (Mikkola 1972; Blomberg 1976; Walker and Galbreath 1979), but shorter wavelength light sources generally attract higher number of species and higher abundance (van Langevelde et al. 2011; Barghini and Medeiros 2012; Kadlec et al. 2016; Brehm 2017). The tested alternative light sources emitted in this shorter wavelength spectra which can explain their higher efficiency and lower selectivity for different caddisfly species. Regarding diversity indexes the samples taken with mercury-vapour lamp showed the highest diversity, that can explain the wide use of this type of light source. The differences might be caused by the attractivity of the other light sources for Limnephilidae species that appeared in their samples with high abundances decreasing the value of diversity indices despite of the higher species diversity of samples.
Considering number of caught individuals belong to Ecnomidae and Hydropsychidae families there were no differences between attractivity of different light sources. In case of Leptoceridae mercury-vapour lamp was the most efficient, while in case of Limnephilidae and Phryganeidae it was the less effective one. Individuals of Psychomyiidae were highly attracted by UV3 light while UV1, UV2 and mercury-vapour lamps were totally inefficient. Chernyshev (1961) reported different attractivity of light sources. In their experiment the black light blue lamp was highly attractive for specimens of Hydroptilidae, Limnephilidae and Psychomyiidae families and in contrast to our results mercury-vapour lamp showed higher attractivity for family of Hydropsychidae. Mikkola (1972) also observed, that the yellow radiation was highly attractive for species of Leptoceridae family. In our investigation, only two light sources (mercury-vapour and mixed-white lights) emitted in the range of yellow radiation and the individuals of Leptoceridae family attracted with higher number exclusively to these light sources. The importance of the green-yellow light was also proved for moths e.g. Pandemis dumetanai Tr. and Pandemis heparana Den. et Schiff. (Járfás 1977; Nowinszky 2013). Laboratory measurements showed two major peaks in the spectral sensitivity of moth species, one UV range between 360 and 365 nm and another in the green-yellow range between 515 and 575 nm (Agee 1973; McFarlane and Eaton 1973; Teel et al. 1976; Pappas and Eaton 1977; Eguchi et al. 1982).
The UV1, UV2 and L1 lights together caught all species (10) attracted with mercury-vapour lamp. The wavelength range of these light sources is between 360 and 407 nm (Fig. 1), thus in case of mercury-vapour lamp these wavelength range can be seen attractive to caddisflies while light above 407 nm is inefficient. The mixed-white light source (W) attracted almost all collected species (17 from 19). The spectral distribution of this light source extends from 360 to 577 nm. Above the wavelength of 391 nm, its spectral peaks are smaller, narrower, and similar to those that the mercury-vapour lamp showed (Fig. 1). It suggests that the high efficiency of the W light source due to its peaks below 391 nm. Differences between UV1, UV2 and L1 lights, which together perform as mercury-vapour lamps, and mixed-white light could be found between 375 and 391 nm. In this range mixed-white light have wider and higher spectral peak, that might result in its higher attractivity. Summarily the 360–391 nm wavelength range attracted most of the collected caddisfly species. It supports result of Mikkola (1972), who found that the eyes of caddisflies are sensitive in the spectrum of 350–600 nm.
The only two species did not attract with mixed-white light tube were O. furva and S. punctatus. First of them was attracted only with L1 light tube with wavelength range of 388–407 nm. Despite of the wavelength range of mixed-white tube covers the spectral distribution of UV2 light, S. punctatus was attracted only with UV2 and mercury-vapour lamps. The wavelength preference of S. punctatus can’t be evaluated since the especially low number of caught individuals (N = 4).
The wavelength range of the L1 and mixed-white (W) light sources together cover the whole spectrum that attracted all the collected caddisfly species. This spectrum and light sources provide the most appropriate wavelength range and tool for caddisfly sampling and parallelly cause largest risk of light pollution for caddisflies when they are used in public lighting.
Since the habitat loss and fragmentation and water pollution endanger caddisflies and other aquatic insects, we should reveal all other threatening factors and should provide solution for them. Caddisfly species trapped in the study area were all common and widespread in small watercourses of the Carpathian lowlands. Along these wetlands use of the tested light sources and any light sources emit between 360 and 407 nm should avoid and lamps work in different wavelength range are suggested. On the other hand, we can collect valuable data for monitoring and conservation planning with use the most effective light sources.
Data availability
Not applicable.
Code availability
During the analysis SynTax 2000, and IndVal 2.0 were used, no computer code was created.
References
Adler PH, Foottit RG (2017) Introduction. In: Foottit RG, Adler PH (eds) Insect Biodiversity: Science and Society, 2nd edn. John Wiley & Sons, Hoboken, New Jersey, pp 1–7
Agee HR (1973) Spectral sensitivity of the compound eyes of field-collected adult bollworms and tobacco budworms. Ann Entomol Soc Am 66:613–615. https://doi.org/10.1093/aesa/66.3.613
Ashfaq M, Khan RA, Khan MA, Rasheed F, Hafeez S (2005) Insect Orientation to various colour lights in the agricultural biomes of Faisalabad. Pakistan Entomol 27(1):49–52
Baker PR, Sadovy Y (1978) The distance and nature of the light-trap response of moths. Nature 276:818–821. https://doi.org/10.1038/276818a0
Band SS, Vaishampayan S, Shrikant P, Navya M (2019) Comparative efficiency of ultra violet black lamp and mercury vapour lamp as a light source in light trap against major insect pest of Kharif crops. J Entomol Zool Stud 7(1):532–537
Barbour MT, Gerritsen J, Snyder BD, Stribling JB (1999) Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates and Fish. U.S. Environmental Protection Agency, Office of Water, Washington DC
Barghini A (2008) Influência da Illuminãçao Artificial sobre a Vida Silvestre: técnicas para minimizar os impactos, com especial enfoque sobre os insectos. Dissertation., Universidade de São Paolo (in Portuguese)
Barghini A, Medeiros BAS (2012) UV radiation as an attractor for insects. LEUKOS - Journal of Illuminating Engineering Society of North America 9(1):47–56. https://doi.org/10.1582/LEUKOS.2012.09.01.003
Bates AJ, Sadler JP, Everett G, Grundy D, Lowe N, Davis G, Baker D, Bridge M, Clifton J, Freestone R, Gardner D, Gibson C, Hemming R, Howarth S, Orridge S, Shaw M, Tams T, Young H (2013) Assessing the value of the Garden Moth Scheme citizen science dataset: How does light trap type affect catch? Entomol Exp Appl 146(3):386–397. https://doi.org/10.1111/eea.12038
Belton P, Kempster RH (1963) Some factors affecting the catches of Lepidoptera in light traps. Can Entomol 95(2):832–837. https://doi.org/10.4039/Ent95832-8
Bhandari G, Jha SK, Giri YP, Manandhar HK, Jha PK, Devkota N, Thapa P, Thapa RB (2017) Performance evaluation of locally developed black light trap for maize insects monitoring in Chitwan, Nepal. J Maize Res Dev 3(1):98–107. https://doi.org/10.3126/jmrd.v3i1.18926
Blahnik RJ, Holzenthal RW (2004) Collection and curation of Trichoptera, with an emphasis on pinned material. Nectopsyche Neotropical Trichoptera Newsletter 1:8–25
Blanton FS, Galindo P, Peyton EL (1955) Report of a three-year light trap survey for biting Diptera in Panama. Mosq News 15:90–93
Blomberg O, Itamies J, Kuusela K (1976) Insect catches in a blended and a black light-trap in northern Finland. Oikos 27:57–63
Brehm G (2017) A new LED lamp for the collection of nocturnal Lepidoptera and a spectral comparison of light-trapping lamps. Nota Lepidopterologica 40(1):87–108. https://doi.org/10.3897/nl.40.11887
Calor A, Mariano R (2012) UV light pan traps for collecting aquatic insects. Entomo Brasilis 5(2):164–166. https://doi.org/10.12741/ebrasilis.v5i2.187
Chernyshev VB (1961) Comparison between the flight responses of insects to light from a mercury quartz lamp and to clear ultra-violet radiation from the same lamp. Entomol Rev 40:308–309
Cowan T, Gries G (2009) Ultraviolet and violet light: attractive orientation cues for the Indian meal moth, Plodia interpunctella. Entomol Exp Appl 131(2):148–158. https://doi.org/10.1111/j.1570-7458.2009.00838.x
Dohet A (2002) Are caddisflies an ideal group for the biological assessment of water quality in streams? Nova Supplementa Entomologica 15:507–520
Donatello S, Rodríguez R, Quintero MGC, Van Tichelen JRCOW, Van P, Hoof V TGV (2019) Revision of the EU Green Public Procurement Criteria for Road Lighting and Traffic Signals. Publications Office of the European Union, Luxembourg
Eguchi E, Watanabe K, Hariyama T, Yamamoto K (1982) A comparison of electrophysiologically determined spectral responses in 35 species of Lepidoptera. J Insect Physiol 28(8):675–682. https://doi.org/10.1016/0022-1910(82)90145-7
Epsky ND, Morrill WL, Mankin RW (2008) Traps for Capturing Insects. In: Capinera JL (ed) Encyclopedia of Entomology. Springer, Dordrecht, pp 3887–3901
Haddock JK, Threlfall GC, Law B, Hochuli DF (2019) Responses of insectivorous bats and nocturnal insects to local changes in street light technology. Austral Ecol 44:1052–1064. https://doi.org/10.1111/aec.12772
Hartstack AW Jr, Hollingsworth JP, Lindquist DA (1968) A technique for measuring trapping efficiency of electric insect traps. J Econ Entomol 61:546–552. https://doi.org/10.1093/jee/61.2.546
Infusino M, Brehm G, Di Marco C, Scalercio S (2017) Assessing the efficiency of UV LEDs as light sources for sampling the diversity of macro-moths (Lepidoptera). Eur J Entomol 114(1):25–33. https://doi.org/10.14411/eje.2017.004
Jackson JK, Resh VH (1989) Activities and Ecological Role of Adult Aquatic Insects in the Riparian Zone of Streams. USDA Forest Service General Technical Report PSW-110 342–344
Járfás J (1977) Results of light trapping of harmful tortricid moths. Kertészeti Egyetem Közleményei 41:123–126 (in Hungarian)
Járfás J, Tóth J (1977) Forecast and protection of the damaging Melolontha species in vineyard. Szőlőtermesztési Agrokémiai Tájékoztató 3(1):2–7 (in Hungarian)
Jehamalar EE, Golda DB, Kiruba S, Das SSM (2010) Trichopterans as a bioindicators of a stream ecosystem. J Basic Appl Biology 4(3):86–90
Jonason D, Franzén M, Ranius T (2014) Surveying moths using light traps: effects of weather and time of year. PLoS ONE 9:e92453. https://doi.org/10.1371/journal.pone.0092453
Kádár F, Szél G (1995) Data on ground beetles captured by light traps in Hungary (Coleoptera, Carabidae). Folia Entomologica Hungarica LVI:37–43
Kadlec T, Pikner M, Piknerova G (2016) Sex-biased response in activity to light sources with different spectral composition in geometrid moths with flightless females (Lepidoptera: Geometridae). Bull Entomol Res 106(5):581–590. https://doi.org/10.1017/S0007485316000298
Kiss O (2002) Diversity of Trichoptera. Cinóber Gyorsnyomda, Eger
Larsson M, Göthberg A, Milberg P (2020) Night, light and flight: light attraction in Trichoptera. Insect Conserv Divers 13(3):296–302. https://doi.org/10.1111/icad.12379
Lenat DR (1993) A biotic index for the southeastern United States: derivation and list of tolerance values, with criteria for assigning water-quality ratings. Journal of the North American Benthological Society 12:279–290. https://doi.org/1467463
Maelzer D, Zalucki MP, Laughlin R (1996) Analysis and interpretation of long-term light trap data for Helicoverpa punctigera (Lepidoptera; Noctuidae) in Australia: population changes and forecasting pest pressure. Bull Entomol Res 86:547–557. https://doi.org/10.1017/S0007485300039341
Malicky H (2004) Atlas of European Trichoptera (Second Edition). Springer, Neatherlands
McFarlane JH, Eaton JL (1973) Comparison of electroretinogram and electromyogram responses to radiant energy stimulation in the moth, Trichoplusia ni. J Insect Physiol 19(4):811–822. https://doi.org/10.1016/0022-1910(73)90153-4
Mészáros Z (1966) A comparison of the Microlepidoptera materials collected by normal and UV light traps. Folia Entomologica Hungarica 19(3):109–131
Mikkola K (1972) Behavioural and electrophysiological responses of night-flying insects, especially Lepidoptera, to near-ultraviolet and visible light. Ann Zool Fenn 9:225–254
Morse JC (2017) Biodiversity of aquatic insects. In: Foottit RG, Adler PH (eds) Insect Biodiversity: Science and Society, 2nd edn. John Wiley & Sons, Hoboken, New Jersey, pp 205–227
Nógrádi S, Uherkovich Á (2002) Magyarország tegzesei (Trichoptera). Dunántúli Dolgozatok Természettudományi Sorozat, Pécs (in Hungarian)
Nowinszky L (2003) The handbook of light trapping. Savaria University Press, Szombathely
Nowinszky L (2013) The dependence of normal and black light type trapping results upon the wingspan of moth species. Appl Ecol Environ Res 11(4):593–610. https://doi.org/10.15666/aeer/1104_593610
Nowinszky L, Kiss O, Puskas J (2014) Swarming patterns of light trapped individuals of caddisfly species (Trichoptera) in Central Europe. Cent Eur J Biology 9:417–430. https://doi.org/10.2478/s11535-013-0272-z
Pappas LG, Eaton JL (1977) The internal ocellus of Manduca sexta and spectral sensitivity. J Insect Physiol 23(1–2):1355–1358. https://doi.org/10.1016/0022-1910(77)90157-3
Pohe S, Winterbourn M, Harding J (2017) Comparison of fluorescent lights with differing spectral properties on catches of adult aquatic and terrestrial insects. New Z Entomol 41(1):1–11. https://doi.org/10.1080/00779962.2017.1398704
Ramamurthy VV, Akhtar MS, Patankar NV, Menon P, Kumar R, Singh SK, Ayri S, Parveen S, Mittal V (2010) Efficiency of different light sources in light traps in monitoring insect diversity. Munis Entomol Zool 5:109–114
Resh VH (1993) Recent trends in the use of Trichoptera in water quality monitoring. In: Otto C (ed) Proceedings of the 7th International Symposium on Trichoptera. Backhuys Publishers, Leiden, pp 285–291
Resh VH, Unzicker JD (1975) Water quality monitoring and aquatic organisms: the importance of species identification. J Water Pollution Control Federation 47:9–19
Schmera D (2003) Assessing stream dwelling caddisfly assemblages (Insecta: Trichoptera) collected by light traps in Hungary. Biodivers Conserv 12(6):1175–1191. https://doi.org/10.1023/A:1023033904518
Simon T (1953) Az Északi-Alföld erdői. Akadémiai Kiadó, Budapest (in Hungarian)
Stanić-Koštroman S, Kučinić M, Kolobara A, Škobić D, Knezović L, Durbešić P (2012) Light-trapped caddisflies (Insecta: Trichoptera) as indicators of the ecological integrity of the Lištica river, Bosnia and Herzegovina. Entomol Croatica 16(1–4):21–36
Szanyi K, Szanyi S (2019) Further data on the caddisfly (Trichoptera) fauna of the Velyka Dobron’ Game Reserve (Ukraine). e-Acta Naturalia Pannonica 19:51–58. https://doi.org/10.24369/eANP.2019.19.51
Szanyi K, Sz S (2018) New data on the caddisfly (Trichoptera) fauna of Transcarpathia. Hidrológiai Közlöny 98(K):66–70
Szanyi Sz, Szőcs L, Varga Z (2015) The zoogeographical and ecological characteristics of Macroheterocera fauna of the Bockerek forest reserve. Erdészettudományi Közlemények 5(1):119–128. https://doi.org/10.17164/EK.2015.008
Szentkirályi F (2002) Fifty-year long insect survey in Hungary: T. Jermy’s contributions to light-trapping. Acta Zool Academiae Scientiarum Hung 48(1):85–105
Teel PD, VanCleave HW, Hollingsworth JP, Harstack AW (1976) Spectral sensitivity of the Hickory Shuckworm to electromagnetic radiation. J Econ Entomol 69(1):57–58. https://doi.org/10.1093/jee/69.1.57
Tikoca S, Hodge S, Tuiwawa M, Brodie G, Pene S, Clayton J (2016) An appraisal of sampling methods and effort for investigating moth assemblages in a Fijian forest. Austral Entomol 55:455–462. https://doi.org/10.1111/aen.12209
Upton MS, Mantle EL (2010) Methods for Collecting, Preserving and Studying Insects and Other Terrestrial Arthropods. Australian Entomological Society. Miscellaneous Publication: No. 3. Fifth edition. Paragon Printers Australasia, Canberra
van Grunsven R, Donners M, Boekee K, Tichelaar I, van Geffen K, Groenendijk D, Berendse F, Veenendaal E (2014) Spectral composition of light sources and insect phototaxis, with an evaluation of existing spectral response models. J Insect Conserv 18(2):225–231. https://doi.org/10.1007/s10841-014-9633-9
van Langevelde F, Ettema JA, Donners M, Wallis DeVries MF, Groenendijk D (2011) Effect of spectral composition of artificial light on the attraction of moths. Biol Conserv 144(9):2274–2281. https://doi.org/10.1016/j.biocon.2011.06.004
Walker A, Galbreath R (1979) Collecting insects at lights: a test of four types of lamp. New Z Entomol 7(1):83–85. https://doi.org/10.1080/00779962.1979.9722336
Ward JB, Henderson IM, Patrick BH, Norrie PH (1996) Seasonality, sex ratios and arrival pattern of some New Zealand caddis (Trichoptera) to light-traps. Aquat Insects 18(3):157–174. https://doi.org/10.1080/01650429609361618
Waringer JA (1989) The abundance and temporal distribution of caddisflies (Insecta: Trichoptera) caught by light traps on the Austrian Danube from 1986 to 1987. Freshw Biol 21:387–399. https://doi.org/10.1111/J.1365-2427.1989.TB01371.X
Waringer JA (1991) Phenology and the influence of meteorological parameters on the catching success of light-trapping for Trichoptera. Freshw Biol 25:307–319. https://doi.org/10.1111/j.1365-2427.1991.tb00493.x
Williams CB (1939) An analysis of four years captures of insects in a light trap. Part 1. General survey; sex proportion; phenology; and time of flight. Trans Royal Entomol Soc Lond 89:79–130. https://doi.org/10.1111/j.1365-2311.1939.tb00738.x
Acknowledgements
The work of Kálmán Szanyi was supported by International Visegrad Fund (Scholarship number: 52010905) 2020.
Funding
The study was supported by International Visegrad Fund: Visegrad Scholarship Program (2020) (Serial Number: 52010905).
Open access funding provided by University of Debrecen.
Author information
Authors and Affiliations
Contributions
SK, NA and SS designed the experiment and analysed the data, all authors contributed to writing of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethics approval
Compliance with Ethical Standards
Informed consent
Informed consent was obtained from all individual participants included in the study.
Research involving human participants and/or animals
This article does not contain any studies with human participants performed by any of the authors. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Szanyi, K., Nagy, A., Varga, Z. et al. Attractivity of various artificial light sources to caddisfly (Trichoptera) species and its importance in their sampling and conservation. J Insect Conserv 26, 839–849 (2022). https://doi.org/10.1007/s10841-022-00427-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-022-00427-x