Abstract
Background
Detailed mapping studies of accessory pathway (AP) conduction have not been previously performed using ultra-high resolution mapping systems. We sought to evaluate the clinical utility of ultra-high resolution mapping systems and the novel “Lumipoint” algorithm in AP ablation.
Methods
This study included 17 patients who underwent AP mapping using minielectrode basket catheters and Rhythmia systems. Ablation was performed with 4-mm irrigated-tip catheters.
Results
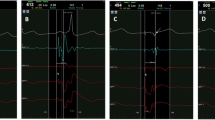
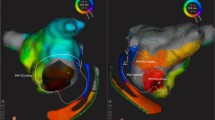
Antegrade and retrograde AP conduction was observed in 6 and 16 patients. Atrial activation map was obtained during orthodromic tachycardia and ventricular pacing in 13 (76.5%) and 14 (82.3%) patients, and the earliest activation area was identical. Ventricular activation maps were created during atrial pacing in 3 patients. All maps showed focal activation patterns on global activation histograms, and the valley on the histogram highlighted the earliest activation area. “Complex activation” features further highlighted limited areas with continuous electrical activity during the time period in the majority. APs were located at the mitral and tricuspid annuli in 15 and 2 patients, and all were successfully eliminated with 3.4 ± 0.6 s applications. No patients had recurrences during a median follow-up of 15 [10.5–22.5] months. At successful ablation sites, the local atrial and ventricular electrogram amplitudes and ratio tended to be greater, and fusion or continuous electrical activity between the atrial and ventricular components was more frequently observed on the minielectrode than ablation catheter (17/17 vs. 12/17, p = 0.005).
Conclusions
Ultra-high resolution activation mapping and a novel algorithm facilitated the AP localization. The local electrogram characteristics differed between the minielectrode and ablation catheters.






Similar content being viewed by others
References
Haïssaguerre M, Dartigues JF, Warin JF, Le Metayer P, Montserrat P, Salamon R. Electrogram patterns predictive of successful catheter ablation of accessory pathways. Value of unipolar recording mode. Circulation. 1991;84:188–202.
Calkins H, Kim YN, Schmaltz S, Sousa J, el-Atassi R, Leon A, Kadish A, Langberg JJ, Morady F. Electrogram criteria for identification of appropriate target sites for radiofrequency catheter ablation of accessory atrioventricular connections. Circulation. 1992;85:565–573.
Grimm W, Miller J, Josephson ME. Successful and unsuccessful sites of radiofrequency catheter ablation of accessory atrioventricular connections. Am Heart J. 1994;128:77–87.
Knight BP, Ebinger M, Oral H, Kim MH, Sticherling C, Pelosi F, et al. Diagnostic value of tachycardia features and pacing maneuvers during paroxysmal supraventricular tachycardia. J Am Coll Cardiol. 2000;36:574–82.
Mantziari L, Butcher C, Kontogeorgis A, Panikker S, Roy K, Markides V, et al. Utility of a novel rapid high-resolution mapping system in the catheter ablation of arrhythmias: an initial human experience of mapping the atria and the left ventricle. JACC Clin Electrophysiol. 2015;1:411–20.
Josephson ME, Anter E. Substrate mapping for ventricular tachycardia: assumptions and misconceptions. JACC Clin Electrophysiol. 2015;1:341–52.
Laţcu DG, Bun SS, Viera F, Delassi T, El Jamili M, Al Amoura A, et al. Selection of critical isthmus in scar-related atrial tachycardia using a new automated ultrahigh resolution mapping system. Circ Arrhythm Electrophysiol. 2017;10:e004510.
Chua K, Upadhyay GA, Lee E, Aziz Z, Beaser AD, Ozcan C, et al. High-resolution mapping of the triangle of Koch: spatial heterogeneity of fast pathway atrionodal connections. Heart Rhythm. 2018;15:421–9.
Takigawa M, Martin CA, Derval N, Denis A, Vlachos K, Kitamura T, et al. Insights from atrial surface activation throughout atrial tachycardia cycle length: a new mapping tool. Heart Rhythm. 2019;16:1652–60.
Martin CA, Takigawa M, Martin R, Maury P, Meyer C, Wong T, et al. Use of novel electrogram “Lumipoint” algorithm to detect critical isthmus and abnormal potentials for ablation in ventricular tachycardia. JACC Clin Electrophysiol. 2019;5:470–9.
Solimene F, Schillaci V, Shopova G, Maddaluno F, Malacrida M, Stabile G. High-resolution mapping and ablation of recurrent left lateral accessory pathway conduction. J Arrhythm. 2017;33:328–9.
Yanagisawa S, Inden Y, Fujii A, Sakamoto Y, Ichiyanagi H, Shibata R, et al. Identification of precise accessory pathway using ultra-high-resolution three-dimensional mapping system: utility and feasibility in preliminary experience. J Interv Card Electrophysiol. 2019;55:241–2.
Acknowledgments
We would like to thank Mr. John Martin for his help in the preparation of the manuscript.
Funding
This work was supported in part by the Grants (19 K08487, 19 K08576) for Cardiovascular Diseases from the Ministry of Health, Labour and Welfare, Japan (to S.M. and H.T.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Miyazaki belongs to the endowed departments of Medtronic, Boston, Abbott, and Japan Lifeline.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Miyazaki, S., Ishikawa, E., Mukai, M. et al. Ultra-high resolution mapping and ablation of accessory pathway conduction. J Interv Card Electrophysiol 62, 309–318 (2021). https://doi.org/10.1007/s10840-020-00900-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-020-00900-0