Abstract
Purpose
To assess the feasibility and safety of same-day discharge after S-ICD implantation by implementing a specific analgesia protocol and phone follow-up.
Methods
Consecutive patients presenting for outpatient S-ICD implantation were enrolled between 1/1/2018 and 4/30/2019. An analgesia protocol included pre-operative acetaminophen and oxycodone, intraoperative local bupivacaine, and limited use of oxycodone-acetaminophen at discharge. The primary outcome was successful same-day discharge. Numerical Pain Rating Scale (NPRS) on postoperative day (POD) 1, 3, 14, and 30 and any unplanned health care visits during the 1-month follow-up period were assessed.
Results
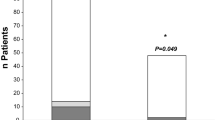
Out of 53 potentially eligible S-ICD patients, 49 patients (92.5%) were enrolled and successfully discharged on the same day. Mean age of these 49 patients was 47 ± 14 years. There were no acute procedural complications. Severe pain (NPRS ≥ 8) on POD 0, 1, and 3 was present in 14.3%, 14.3%, and 8.2% of patients, respectively. The total in-hospital stay was 534 ± 80 min. Four unplanned visits (8%) due to cardiac or device-related issues occurred during 1-month follow-up, including 2 patients with heart failure exacerbation, one patient with an incisional infection, and one patient with inappropriate shocks.
Conclusions
With the appropriate institutional protocol including specific analgesics and phone follow-up, same-day discharge after outpatient S-ICD implantation is feasible and appears safe for most patients.. Device-related pain can be severe in the first 3 days post-implantation and can be successfully treated with limited supply of narcotic medications.



Similar content being viewed by others
References
Marijon E, Albenque JP, Boveda S, Jacob S, Schmutz M, Bortone A, et al. Feasibility and safety of same-day home discharge after radiofrequency catheter ablation. Am J Cardiol. 2009;104(2):254–8. https://doi.org/10.1016/j.amjcard.2009.03.024.
Peplow J, Randall E, Campbell-Cole C, Kamdar R, Petzer E, Dhillon P, et al. Day-case device implantation-a prospective single-center experience including patient satisfaction data. Pacing Clin Electrophysiol. 2018;41(5):546–52. https://doi.org/10.1111/pace.13324.
Weiss R, Knight BP, Gold MR, Leon AR, Herre JM, Hood M, et al. Safety and efficacy of a totally subcutaneous implantable-cardioverter defibrillator. Circulation. 2013;128(9):944–53. https://doi.org/10.1161/CIRCULATIONAHA.113.003042.
Friedman DJ, Parzynski CS, Varosy PD, Prutkin JM, Patton KK, Mithani A, et al. Trends and in-hospital outcomes associated with adoption of the subcutaneous implantable cardioverter defibrillator in the United States. JAMA Cardiol. 2016;1(8):900–11. https://doi.org/10.1001/jamacardio.2016.2782.
Gold MR, Aasbo JD, El-Chami MF, Niebauer M, Herre J, Prutkin JM, et al. Subcutaneous implantable cardioverter-defibrillator post-approval study: clinical characteristics and perioperative results. Heart Rhythm. 2017;14(10):1456–63. https://doi.org/10.1016/j.hrthm.2017.05.016.
Burke MC, Gold MR, Knight BP, Barr CS, Theuns D, Boersma LVA, et al. Safety and efficacy of the totally subcutaneous implantable defibrillator: 2-year results from a pooled analysis of the IDE study and EFFORTLESS registry. J Am Coll Cardiol. 2015;65(16):1605–15. https://doi.org/10.1016/j.jacc.2015.02.047.
Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2018;72(14):e91–e220. https://doi.org/10.1016/j.jacc.2017.10.054.
Francia P, Ziacchi M, De Filippo P, Viani S, D'Onofrio A, Russo V, et al. Subcutaneous implantable cardioverter defibrillator eligibility according to a novel automated screening tool and agreement with the standard manual electrocardiographic morphology tool. J Interv Card Electrophysiol. 2018;52(1):61–7. https://doi.org/10.1007/s10840-018-0326-2.
Dripps RD, Lamont A, Eckenhoff JE. The role of anesthesia in surgical mortality. JAMA. 1961;178:261–6. https://doi.org/10.1001/jama.1961.03040420001001.
Essandoh MK, Otey AJ, Abdel-Rasoul M, Stein EJ, Turner KR, Joseph NC, et al. Monitored anesthesia care for subcutaneous cardioverter-defibrillator implantation: a single-center experience. J Cardiothorac Vasc Anesth. 2016;30(5):1228–33. https://doi.org/10.1053/j.jvca.2016.06.007.
Essandoh MK, Mark GE, Aasbo JD, Joyner CA, Sharma S, Decena BF, et al. Anesthesia for subcutaneous implantable cardioverter-defibrillator implantation: perspectives from the clinical experience of a U.S. panel of physicians. Pacing Clin Electrophysiol. 2018. https://doi.org/10.1111/pace.13364.
Boonstra AM, Stewart RE, Koke AJ, Oosterwijk RF, Swaan JL, Schreurs KM, et al. Cut-off points for mild, moderate, and severe pain on the numeric rating scale for pain in patients with chronic musculoskeletal pain: variability and influence of sex and catastrophizing. Front Psychol. 2016;7:1466. https://doi.org/10.3389/fpsyg.2016.01466.
Aldrete JA, Kroulik D. A postanesthetic recovery score. Anesth Analg. 1970;49(6):924–34.
Droghetti A, Basso Ricci E, Scimia P, Harizai F, Marini M. Ultrasound-guided serratus anterior plane block combined with the two-incision technique for subcutaneous ICD implantation. Pacing Clin Electrophysiol. 2018;41(5):517–23. https://doi.org/10.1111/pace.13318.
Miller MA, Garg J, Salter B, Brouwer TF, Mittnacht AJ, Montgomery ML, et al. Feasibility of subcutaneous implantable cardioverter-defibrillator implantation with opioid sparing truncal plane blocks and deep sedation. J Cardiovasc Electrophysiol. 2019;30(1):141–8. https://doi.org/10.1111/jce.13750.
Manzoor S, Taneja R, Sood N, Puri A, Kadayaprath G. Comparative study to assess the quality of analgesia of bupivacaine and bupivacaine with dexmedetomidine in ultrasound-guided pectoral nerve block type I and II in breast surgeries. J Anaesthesiol Clin Pharmacol. 2018;34(2):227–31. https://doi.org/10.4103/joacp.JOACP_366_16.
Noviasky J, Pierce DP, Whalen K, Guharoy R, Hildreth K. Bupivacaine liposomal versus bupivacaine: comparative review. Hosp Pharm. 2014;49(6):539–43. https://doi.org/10.1310/hpj4906-539.
Edlund MJ, Martin BC, Russo JE, DeVries A, Braden JB, Sullivan MD. The role of opioid prescription in incident opioid abuse and dependence among individuals with chronic noncancer pain: the role of opioid prescription. Clin J Pain. 2014;30(7):557–64. https://doi.org/10.1097/AJP.0000000000000021.
Wilkoff BL, Fauchier L, Stiles MK, Morillo CA, Al-Khatib SM, Almendral J, et al. 2015 HRS/EHRA/APHRS/SOLAECE expert consensus statement on optimal implantable cardioverter-defibrillator programming and testing. Heart Rhythm. 2016;13(2):e50–86. https://doi.org/10.1016/j.hrthm.2015.11.018.
Knops RE, Brouwer TF, Barr CS, Theuns DA, Boersma L, Weiss R, et al. The learning curve associated with the introduction of the subcutaneous implantable defibrillator. Europace. 2016;18(7):1010–5. https://doi.org/10.1093/europace/euv299.
Funding
Boston Scientific Corporation (Natick, MA, USA) funded the study. The funding source did not have any involvement in the study design, in the collection, analysis and interpretation of data, in the writing of the report, or in the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
Conceptualization: Toshimasa Okabe, Emile G. Daoud, and Michael K Essandoh
Methodology: Toshimasa Okabe, Emile G. Daoud, and Michael K Essandoh
Data collection: Toshimasa Okabe, Adrianne Miller, Tanner Koppert, Rafael Cavalcanti, Diego Alcivar-Franco, Jemina Osei, and Omar Kahaly
Formal analysis and investigation: Toshimasa Okabe, Emile G. Daoud, Michael K Essandoh, and Muhammad R. Afzal
Writing - original draft preparation: Toshimasa Okabe, Emile G. Daoud.
Writing - review and editing: Muhammad R. Afzal, Jaret Tyler, Mahmoud Houmsse, Steven J. Kalbfleisch, Raul WeissJohn D. Hummel, Ralph S. Augostini, Sujatha Bhandary, Galina Dimitrova, Kasey Fiorini Hamdy Elsayed-Awad, Antolin Flores, MD#, Leonid Gorelik, Manoj H. Iyer, Samiya Saklayen, Erica Stein, Katja Turner, William Perez
Funding acquisition: Emile G. Daoud
Supervision: Emile G. Daoud, Michael K Essandoh, Michael J. Andritsos
Corresponding author
Ethics declarations
Conflict of interest
Drs Weiss and Essandoh have received consulting fees and speaking fees from Boston Scientific. The remaining authors have no conflicts to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Okabe, T., Miller, A., Koppert, T. et al. Feasibility and safety of same day subcutaneous defibrillator implantation and send home (DASH) strategy. J Interv Card Electrophysiol 57, 311–318 (2020). https://doi.org/10.1007/s10840-019-00673-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-019-00673-1