Abstract
Purpose
To evaluate the influence of the endometrial receptivity array (ERA) test on the implantation rate (IR) and pregnancy rate (PR) in patients with previous failed euploid embryo transfers (Euploid-ET) or oocyte donation embryo transfers (Donor-ET).
Methods
Single-center retrospective study of patients with ≥ 1 previous failed Euploi-ET (n = 24) or ≥ 2 failed Donor-ET (n = 32) who underwent an ERA test and a post-ERA Euploid-ET/Donor-ET between 2012 and 2018. Controls were patients with ≥ 1 previously failed Euploid-ET (n = 119) or ≥ 2 failed Donor-ET (n = 158) who underwent Euploid-ET/Donor-ET during the same period without performing an ERA test. Only blastocyst stage embryos were included. IR/PR was compared between the post-ERA ET and the last ET in the control group.
Results
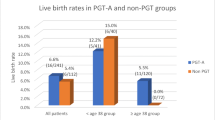
There was no statistically significant difference regarding IR [55.6% (34.6–76.5%) vs. 65.0% (56.9–73.1%)] nor PR (58.3% vs.70.6%, p = 0.238) in the Euploid-ET ERA vs. Euploid-ET control groups. In the Donor-ET arm, both IR [26.8% (12.3–41.4%) vs. 57.2% (50.1–64.3%)] and PR (34.4% vs. 65.2%, p = 0.001) were significantly lower in the ERA group. Multivariate analysis confirmed that performing an ERA test did not influence the PR in the Euploid-ET arm and was associated with a diminished PR in the Donor-ET arm. In the ERA group, 41.1% patients were non-receptive (NR). No significant difference was found regarding IR/PR in NR vs. receptive patients in both Euploid-ET/Donor-ET arms.
Conclusions
In our sample, the performance of an ERA test did not improve pregnancy outcomes. Future prospective studies in larger samples are needed to confirm the role of the ERA test in Euploid-ET/Donor-ET.

Similar content being viewed by others
References
Fox C, Morin S, Jeong JW, Scott RT, Lessey BA. Local and systemic factors and implantation: What is the evidence? Fertil Steril. 2016;105(4):873–84.
Craciunas L, Gallos I, Chu J, Bourne T, Quenby S, Brosens JJ, et al. Conventional and modern markers of endometrial receptivity: a systematic review and meta-analysis. Hum Reprod Update. 2019;25(2):202–23.
Díaz-Gimeno P, Horcajadas JA, Martinez-Conejero JA, Esteban FJ, Alama P, Pellicer A, et al. A genomic diagnostic tool for human endometrial receptivity based on the transcriptomic signature. Fertil Steril. 2011;95(1):50–60.
Mahajan N. Endometrial receptivity array: Clinical application. J Hum Reprod Sci. 2015;8(3):121–9.
Bassil R, Casper R, Samara N, Hsieh TB, Barzilay E, Orvieto R, et al. Does the endometrial receptivity array really provide personalized embryo transfer? J Assist Reprod Genet. 2018;35(7):1301–5.
Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, Gómez E, Fernández-Sánchez M, Carranza F, et al. The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil Steril. 2013;100(3):818–24.
Hashimoto T, Koizumi M, Doshida M, Toya M, Sagara E, Oka N, et al. Efficacy of the endometrial receptivity array for repeated implantation failure in Japan: A retrospective, two-centers study. Reprod Med Biol. 2017;16(3):290–6.
Kort JD, McCoy RC, Demko Z, Lathi RB. Are blastocyst aneuploidy rates different between fertile and infertile populations? J Assist Reprod Genet. 2018;35(3):403–8.
Rubio C, Rodrigo L, Garcia-Pascual C, Peinado V, Campos-Galindo I, Garcia-Herrero S, Simón C Clinical application of embryo aneuploidy testing by NGS. Biol Reprod. 2019.
Franasiak JM, Forman EJ, Hong KH, Werner MD, Upham KM, Treff NR, et al. The nature of aneuploidy with increasing age of the female partner: A review of 15,169 consecutive trophectoderm biopsies evaluated with comprehensive chromosomal screening. Fertil Steril. 2014;101:656–663.e1.
Shapiro BS, Daneshmand ST, Desai J, Garner FC, Aguirre M, Hudson C. The risk of embryo-endometrium asynchrony increases with maternal age after ovarian stimulation and IVF. Reprod Biomed Online. 2016;33(1):50–5.
Coughlan C, Ledger W, Wang Q, Liu F, Demirol A, Gurgan T, et al. Recurrent implantation failure: definition and management. Reprod Biomed Online. 2014;28(1):14–38.
Martínez F, Latre L, Clua E, Rodriguez I, Coroleu B. Replacing GnRH agonists with GnRH antagonists in oocyte recipient cycle did not adversely affect the pregnancy rates. Eur J Obstet Gynecol Reprod Biol. 2011;159:355–8.
Clua E, Tur R, Coroleu B, Boada M, Barri PN, Veiga A. Analysis of factors associated with multiple pregnancy in an oocyte donation programme. Reprod Biomed Online. 2010;21(5):694–9.
Martínez F, Kava-Braverman A, Clúa E, Rodríguez I, Gaggiotti Marre S, Coroleu B, et al. Reproductive outcomes in recipients are not associated with oocyte donor body mass index up to 28 kg/m2: a cohort study of 2722 cycles. Reprod Biomed Online. 2017;35(6):739–46.
Simon AL, Kiehl M, Fischer E, Proctor JG, Bush MR, Givens C, et al. Pregnancy outcomes from more than 1,800 in vitro fertilization cycles with the use of 24-chromosome single-nucleotide polymorphism–based preimplantation genetic testing for aneuploidy. Fertil Steril. 2018;110(1):113–21.
Polanski LT, Baumgarten MN, Quenby S, Brosens J, Campbell BK, Raine-Fenning NJ. What exactly do we mean by “recurrent implantation failure”? A systematic review and opinion. Reprod Biomed Online. 2014;28(4):409–23.
Martinez F, Rodriguez I, Devesa M, Buxaderas R, Gómez MJ, Coroleu B. Should progesterone on the human chorionic gonadotropin day still be measured? Fertil Steril. 2015;105:86–92.
Coll L, Parriego M, Boada M, Devesa M, Arroyo G, Rodríguez I, et al. Transition from blastomere to trophectoderm biopsy: Comparing two preimplantation genetic testing for aneuploidies strategies. Zygote. 2018;26:191–8.
Permanente del Grupo de Interés de Embriología C, Hurtado de Mendoza M, Arroyo G, Figueroa M, Busquets A, De los Santos M, et al. Cuadernos de embriología clínica II. Criterios ASEBIR de valoración morfológica de oocitos, embriones tempranos y blastocistos humanos. 3rd ed. Madrid: Góbalo; 2015.
Cobo A, De Los Santos MJ, Castellò D, Gámiz P, Campos P, Remohí J. Outcomes of vitrified early cleavage-stage and blastocyst-stage embryos in a cryopreservation program: Evaluation of 3,150 warming cycles. Fertil Steril. 2012;98(5):1138–1146.e1.
Coroleu B, Carreras O, Veiga A, Martell A, Martinez F, Belil I, et al. Embryo transfer under ultrasound guidance improves pregnancy rates after in-vitro fertilization. Hum Reprod. 2000;15:616–20.
Frantz S, Parinaud J, Kret M, Rocher-Escriva G, Papaxanthos-Roche A, Creux H, et al. Decrease in pregnancy rate after endometrial scratch in women undergoing a first or second in vitro fertilization. A multicenter randomized controlled trial. Hum Reprod. 2019;34(1):92–9.
Tan J, Kan A, Hitkari J, Taylor B, Tallon N, Warraich G, et al. The role of the endometrial receptivity array (ERA) in patients who have failed euploid embryo transfers. J Assist Reprod Genet. 2018;35(4):683–92.
Reignier A, Lammers J, Barriere P, Freour T. Can time-lapse parameters predict embryo ploidy? A systematic review. Reprod Biomed Online. 2018;36(4):380–7.
Zhang J, Tao W, Liu H, Yu G, Li M, Ma S, et al. Morphokinetic parameters from a time-lapse monitoring system cannot accurately predict the ploidy of embryos. J Assist Reprod Genet. 2017;34(9):1173–8.
Nastri C, Lensen S, Gibreel A, Raine-Fenning N, Ferriani R, Bhattacharya S, et al. Endometrial injury in women undergoing assisted reproductive techniques. Cochrane Database Syst Rev. 2015;(3):CD009517.
Acknowledgments
This work was performed under the auspices of the Cátedra de Investigació en Obstetrícia I Ginecologia of the Department of Obstetrics, Gynaecology and Reproduction, Dexeus University Hospital, Universitat Autònoma de Barcelona.
Competing interests
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Contributions
ARN designed the study, performed the literature search, wrote the manuscript, and constructed the tables and figures. MD, FM, NP, and BC contributed to the design of the research study, edited the manuscript, and provided critical review of the manuscript. SGM and IR contributed to data collection and statistical analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The study was performed in accordance with the Helsinki Declaration and with approval of the Institutional Review Board. Because the study involved completely de-identified data extraction from electronic medical records, patient consent was not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Neves, A.R., Devesa, M., Martínez, F. et al. What is the clinical impact of the endometrial receptivity array in PGT-A and oocyte donation cycles?. J Assist Reprod Genet 36, 1901–1908 (2019). https://doi.org/10.1007/s10815-019-01535-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-019-01535-5