Abstract
Purpose
The impact of sperm DNA damage on intracytoplasmic sperm injection (ICSI) outcomes remains controversial. The purpose of the study was to evaluate the prognostic value of several types of sperm nuclear damage on ICSI clinical pregnancy.
Methods
Our retrospective study included a total of 132 couples who consulted for male or mixed-factor infertility that benefited from ICSI cycles from January 2006 to December 2015. All infertile males presented at least one conventional semen parameter alteration. Sperm nuclear damage was assessed using the Motile Sperm Organelle Morphological Examination for sperm head relative vacuolar area (RVA), aniline blue staining for chromatin condensation, terminal deoxynucleotidyl transferase dUTP nick-end labeling for DNA fragmentation, and fluorescence in situ hybridization for aneuploidy.
Results
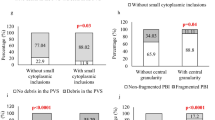
Infertile males who achieved pregnancy after ICSI had fewer chromatin condensation defects than did males who did not achieve any pregnancy (15.8 ± 12.0% vs. 11.4 ± 7.9%, respectively, P = 0.0242), which remained significant in multivariate regression analysis (RR = 0.40 [0.18 to 0.86], P = 0.02). RVA, DNA fragmentation, and aneuploidy were not predictive factors of ICSI outcomes. The pregnancy rate was significantly decreased by number of progressive motile spermatozoa with normal morphology after migration (P = 0.04). In female partners, 17β estradiol of less than 2000 pg/mL on the day of ovulation induction significantly reduced the occurrence of clinical pregnancy (P = 0.04).
Conclusion
Sperm chromatin condensation defects were more frequently observed in couples with ICSI failure and should be considered a negative predictive factor for the occurrence of clinical pregnancy.


Similar content being viewed by others
References
Rubino P, Vigano P, Luddi A, Piomboni P. The ICSI procedure from past to future: a systematic review of the more controversial aspects. Hum Reprod Update. 2016;22:194–227.
Agarwal A, Said TM. Role of sperm chromatin abnormalities and DNA damage in male infertility. Hum Reprod Update. 2003;9:331–45.
Bakos HW, Thompson JG, Feil D, Lane M. Sperm DNA damage is associated with assisted reproductive technology pregnancy. Int J Androl. 2008;31:518–26.
Pinborg A, Gaarslev C, Hougaard CO, Andersen AN, Andersen PK, Andersen PK, et al. Influence of female bodyweight on IVF outcome: a longitudinal multicentre cohort study of 487 infertile couples. Reprod BioMed Online. 2011;23:490–9.
Andersen AN, Witjes H, Gordon K, Mannaerts B. Predictive factors of ovarian response and clinical outcome after IVF/ICSI following a rFSH/GnRH antagonist protocol with or without oral contraceptive pre-treatment. Hum Reprod. 2011;26:3413–23.
Meseguer M, Santiso R, Garrido N, Garcia-Herrero S, Remohi J, Fernandez JL. Effect of sperm DNA fragmentation on pregnancy outcome depends on oocyte quality. Fertil Steril. 2011;95:124–8.
Vural B, Sofuoglu K, Caliskan E, Delikara N, Aksoy E, Uslu H, et al. Predictors of intracytoplasmic sperm injection (ICSI) outcome in couples with and without male factor infertility. Clin Exp Obstet Gynecol. 2005;32:158–62.
Kaarouch I, Bouamoud N, Louanjli N, Madkour A, Copin H, Benkhalifa M, et al. Impact of sperm genome decay on day-3 embryo chromosomal abnormalities from advanced-maternal-age patients. Mol Reprod Dev. 2015;82:809–19.
Erenpreiss J, Spano M, Erenpreisa J, Bungum M, Giwercman A. Sperm chromatin structure and male fertility: biological and clinical aspects. Asian J Androl. 2006;8:11–29.
Spano M, Seli E, Bizzaro D, Manicardi GC, Sakkas D. The significance of sperm nuclear DNA strand breaks on reproductive outcome. Curr Opin Obstet Gynecol. 2005;17:255–60.
Seli E, Sakkas D. Spermatozoal nuclear determinants of reproductive outcome: implications for ART. Hum Reprod Update. 2005;11:337–49.
O'Brien J, Zini A. Sperm DNA integrity and male infertility. Urology. 2005;65:16–22.
Gandini L, Lombardo F, Paoli D, Caruso F, Eleuteri P, Leter G, et al. Full-term pregnancies achieved with ICSI despite high levels of sperm chromatin damage. Hum Reprod. 2004;19:1409–17.
Morris ID, Ilott S, Dixon L, Brison DR. The spectrum of DNA damage in human sperm assessed by single cell gel electrophoresis (comet assay) and its relationship to fertilization and embryo development. Hum Reprod. 2002;17:990–8.
Henkel R, Kierspel E, Hajimohammad M, Stalf T, Hoogendijk C, Mehnert C, et al. DNA fragmentation of spermatozoa and assisted reproduction technology. Reprod BioMed Online. 2003;7:477–84.
Hammadeh ME, al-Hasani S, Stieber M, Rosenbaum P, Kupker D, Diedrich K, et al. The effect of chromatin condensation (aniline blue staining) and morphology (strict criteria) of human spermatozoa on fertilization, cleavage and pregnancy rates in an intracytoplasmic sperm injection programme. Hum Reprod. 1996;11:2468–71.
Saleh RA, Agarwal A, Nada EA, El-Tonsy MH, Sharma RK, Meyer A, et al. Negative effects of increased sperm DNA damage in relation to seminal oxidative stress in men with idiopathic and male factor infertility. Fertil Steril. 2003;79(Suppl 3):1597–605.
Lewis SE, Aitken RJ. DNA damage to spermatozoa has impacts on fertilization and pregnancy. Cell Tissue Res. 2005;322:33–41.
Sun JG, Jurisicova A, Casper RF. Detection of deoxyribonucleic acid fragmentation in human sperm: correlation with fertilization in vitro. Biol Reprod. 1997;56:602–7.
Lopes S, Sun JG, Jurisicova A, Meriano J, Casper RF. Sperm deoxyribonucleic acid fragmentation is increased in poor-quality semen samples and correlates with failed fertilization in intracytoplasmic sperm injection. Fertil Steril. 1998;69:528–32.
Benchaib M, Braun V, Lornage J, Hadj S, Salle B, Lejeune H, et al. Sperm DNA fragmentation decreases the pregnancy rate in an assisted reproductive technique. Hum Reprod. 2003;18:1023–8.
Huang CC, Lin DP, Tsao HM, Cheng TC, Liu CH, Lee MS. Sperm DNA fragmentation negatively correlates with velocity and fertilization rates but might not affect pregnancy rates. Fertil Steril. 2005;84:130–40.
Avendaño C, Franchi A, Duran H, Oehninger S. DNA fragmentation of normal spermatozoa negatively impacts embryo quality and intracytoplasmic sperm injection outcome. Fertil Steril. 2010;94(2):549–57.
Colaco S, Sakkas D. Paternal factors contributing to embryo quality. J Assist Reprod Genet. 2018 Sep 11;35:1953–68. https://doi.org/10.1007/s10815-018-1304-4.25.
Berkovitz A, Eltes F, Ellenbogen A, Peer S, Feldberg D, Bartoov B. Does the presence of nuclear vacuoles in human sperm selected for ICSI affect pregnancy outcome? Hum Reprod. 2006;21:1787–90.
Vanderzwalmen P, Hiemer A, Rubner P, Bach M, Neyer A, Stecher A, et al. Blastocyst development after sperm selection at high magnification is associated with size and number of nuclear vacuoles. Reprod BioMed Online. 2008;17:617–27.
Goswami G, Sharma M, Jugga D, Gouri DM. Can intracytoplasmic morphologically selected spermatozoa injection be used as first choice of treatment for severe male factor infertility patients? J Hum Reprod Sci. 2018;11:40–4.
Gaspard O, Vanderzwalmen P, Wirleitner B, Ravet S, Wenders F, Eichel V, et al. Impact of high magnification sperm selection on neonatal outcomes: a retrospective study. J Assist Reprod Genet. 2018;35:1113–21.
Kacem O, Sifer C, Barraud-Lange V, Ducot B, De Ziegler D, Poirot C, et al. Sperm nuclear vacuoles, as assessed by motile sperm organellar morphological examination, are mostly of acrosomal origin. Reprod BioMed Online. 2010;20:132–7.
Franco JG Jr, Baruffi RL, Mauri AL, Petersen CG, Oliveira JB, Vagnini L, et al. Significance of large nuclear vacuoles in human spermatozoa: implications for ICSI. Reprod BioMed Online. 2008;17:42–5.
Perdrix A, Travers A, Chelli MH, Escalier D, Do Rego JL, Milazzo JP, et al. Assessment of acrosome and nuclear abnormalities in human spermatozoa with large vacuoles. Hum Reprod. 2011;26:47–58.
Perdrix A, Rives N. Motile sperm organelle morphology examination (MSOME) and sperm head vacuoles: state of the art in 2013. Hum Reprod Update. 2013;19:527–41.
Collins JA, Barnhart KT, Schlegel PN. Do sperm DNA integrity tests predict pregnancy with in vitro fertilization? Fertil Steril. 2008;89:823–31.
Bungum M, Humaidan P, Spano M, Jepson K, Bungum L, Giwercman A. The predictive value of sperm chromatin structure assay (SCSA) parameters for the outcome of intrauterine insemination, IVF and ICSI. Hum Reprod. 2004;19(6):1401–8.
Larson-Cook KL, Brannian JD, Hansen KA, Kasperson KM, Aamold ET, Evenson DP. Relationship between the outcomes of assisted reproductive techniques and sperm DNA fragmentation as measured by the sperm chromatin structure assay. Fertil Steril. 2003;80:895–902.
Sakkas D, Alvarez JG. Sperm DNA fragmentation: mechanisms of origin, impact on reproductive outcome, and analysis. Fertil Steril. 2010;93:1027–36.
Zhao J, Zhang Q, Wang Y, Li Y. Whether sperm deoxyribonucleic acid fragmentation has an effect on pregnancy and miscarriage after in vitro fertilization/intracytoplasmic sperm injection: a systematic review and meta-analysis. Fertil Steril. 2014;102:998–1005.e8.
Borini A, Tarozzi N, Bizzaro D, Bonu MA, Fava L, Flamigni C, et al. Sperm DNA fragmentation: paternal effect on early post-implantation embryo development in ART. Hum Reprod. 2006;21:2876–81.
Irez T, Sahmay S, Ocal P, Goymen A, Senol H, Kaleli S, et al. Investigation of the association between the outcomes of sperm chromatin condensation and decondensation tests, and assisted reproduction techniques. Andrologia. 2015;47:438–47.
Li Z, Wang L, Cai J, Huang H. Correlation of sperm DNA damage with IVF and ICSI outcomes: a systematic review and meta-analysis. J Assist Reprod Genet. 2006;23:367–76.
Zini A, Sigman M. Are tests of sperm DNA damage clinically useful? Pros and cons. J Androl. 2009;30:219–29.
Zini A. Are sperm chromatin and DNA defects relevant in the clinic? Syst Biol Reprod Med. 2011;57:78–85.
Practice Committee of the American Society for Reproductive Medicine. The clinical utility of sperm DNA integrity testing: a guideline. Fertil Steril. 2013;99:673–7.
Agarwal A, Allamaneni SS. Sperm DNA damage assessment: a test whose time has come. Fertil Steril. 2005;84:850–3.
World Health Organization. WHO laboratory manual for the examination and processing of human semen. Geneva: WHO Press; 2010.
Auger J, Eustache F, Andersen AG, Irvine DS, Jorgensen N, Skakkebaek NE, et al. Sperm morphological defects related to environment, lifestyle and medical history of 1001 male partners of pregnant women from four European cities. Hum Reprod. 2001;16:2710–7.
Perdrix A, Saidi R, Menard JF, Gruel E, Milazzo JP, Macé B, et al. Relationship between conventional sperm parameters and motile sperm organelle morphology examination (MSOME). Int J Androl. 2012;35:491–8.
Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of Embryology. The Istanbul consensus workshop on embryo assessment: proceedings of an expert meeting. Hum Reprod. 2011;26:1270–83.
Hammadeh ME, Stieber M, Haidl G, Schmidt W. Association between sperm cell chromatin condensation, morphology based on strict criteria, and fertilization, cleavage and pregnancy rates in an IVF program. Andrologia. 1998;30(1):29–35.
Avendano C, Franchi A, Taylor S, Morshedi M, Bocca S, Oehninger S. Fragmentation of DNA in morphologically normal human spermatozoa. Fertil Steril. 2009;91:1077–84.
Cohen-Bacrie P, Belloc S, Menezo YJ, Clement P, Hamidi J, Benkhalifa M. Correlation between DNA damage and sperm parameters: a prospective study of 1,633 patients. Fertil Steril. 2009;91:1801–5.
Belloc S, Benkhalifa M, Cohen-Bacrie M, Dalleac A, Chahine H, Amar E, et al. Which isolated sperm abnormality is most related to sperm DNA damage in men presenting for infertility evaluation. J Assist Reprod Genet. 2014;31:527–32.
Rives N, Saint Clair A, Mazurier S, Sibert L, Simeon N, Joly G, et al. Relationship between clinical phenotype, semen parameters and aneuploidy frequency in sperm nuclei of 50 infertile males. Hum Genet. 1999;105:266–72.
Sadeghi MR, Hodjat M, Lakpour N, Arefi S, Amirjannati N, Modarresi T, et al. Effects of sperm chromatin integrity on fertilization rate and embryo quality following intracytoplasmic sperm injection. Avicenna J Med Biotechnol. 2009;1:173–80.
Filatov MV, Semenova EV, Vorobeva OA, Leont'eva OA, Drobchenko EA. Relationship between abnormal sperm chromatin packing and IVF results. Mol Hum Reprod. 1999;5:825–30.
Hofmann N, Hilscher B. Use of aniline blue to assess chromatin condensation in morphologically normal spermatozoa in normal and infertile men. Hum Reprod. 1991;6:979–82.
Evenson DP, Jost LK, Marshall D, Zinaman MJ, Clegg E, Purvis K, et al. Utility of the sperm chromatin structure assay as a diagnostic and prognostic tool in the human fertility clinic. Hum Reprod. 1999;14:1039–49.
Depa-Martynow M, Kempisty B, Lianeri M, Jagodzinski PP, Jedrzejczak P. Association between fertilin beta, protamines 1 and 2 and spermatid-specific linker histone H1-like protein mRNA levels, fertilization ability of human spermatozoa, and quality of preimplantation embryos. Folia Histochem Cytobiol. 2007;45(Suppl 1):S79–85.
Singh NP, Muller CH, Berger RE. Effects of age on DNA double-strand breaks and apoptosis in human sperm. Fertil Steril. 2003;80:1420–30.
Schmid TE, Eskenazi B, Baumgartner A, Marchetti F, Young S, Weldon R, et al. The effects of male age on sperm DNA damage in healthy non-smokers. Hum Reprod. 2007;22:180–7.
Aitken RJ, De Iuliis GN, McLachlan RI. Biological and clinical significance of DNA damage in the male germ line. Int J Androl. 2009;32:46–56.
Yatsenko AN, Turek PJ. Reproductive genetics and the aging male. J Assist Reprod Genet. 2018;35:933–41.
Belloc S, Hazout A, Zini A, Merviel P, Cabry R, Chahine H, et al. How to overcome male infertility after 40: influence of paternal age on fertility. Maturitas. 2014;78:22–9.
Kaarouch I, Bouamoud N, Madkour A, Louanjli N, Saadani B, Assou S, et al. Paternal age: negative impact on sperm genome decays and IVF outcomes after 40 years. Mol Reprod Dev. 2018;85:271–80.
Belloc S, Benkhalifa M, Junca AM, Dumont M, Bacrie PC, Ménézo Y. Paternal age and sperm DNA decay: discrepancy between chromomycin and aniline blue staining. Reprod BioMed Online. 2009;19:264–9.
Moskovtsev SI, Willis J, Mullen JB. Age-related decline in sperm deoxyribonucleic acid integrity in patients evaluated for male infertility. Fertil Steril. 2006;85:496–9.
Bungum M, Humaidan P, Axmon A, Spano M, Bungum L, Erenpreiss J, et al. Sperm DNA integrity assessment in prediction of assisted reproduction technology outcome. Hum Reprod. 2007;22:174–9.
Bungum M, Bungum L, Lynch KF, Wedlund L, Humaidan P, Giwercman A. Spermatozoa DNA damage measured by sperm chromatin structure assay (SCSA) and birth characteristics in children conceived by IVF and ICSI. Int J Androl. 2012;35:485–90.
Foroozanfard F, Moraveji SA, Taghavi SA, Karimi F. Association between serum estradiol level on the day of hCG administration and IVF-ICSI outcome. J Obstet Gynaecol India. 2016;66:170–3.
Yuan X, Saravelos SH, Wang Q, Xu Y, Li TC, Zhou C. Endometrial thickness as a predictor of pregnancy outcomes in 10787 fresh IVF-ICSI cycles. Reprod BioMed Online. 2016;33:197–205.
Cooper TG, Noonan E, von Eckardstein S, Auger J, Baker HW, Behre HM. World Health Organization reference values for human semen characteristics. Hum Reprod Update. 2010;16:231–45.
Acknowledgments
This work was supported by the Normandy Rouen University Hospital.
Funding
No specific funding was sought for the study.
Author information
Authors and Affiliations
Contributions
Cynthia Bichara contributed in data collection, analysis, and interpretation, statistical analysis, and writing of the manuscript; Benoit Berby and Aurélie Rives contributed in collection of biological and clinical data; F. Jumeau contributed in data analysis and revision of the manuscript; Véronique Sétif contributed in execution of experiments; M. Letailleur contributed in female patient recruitment, clinical examination, and ART procedure; Louis Sibert contributed in male patient recruitment and clinical examination; Nathalie Rives contributed in design and supervision of the study, funding support, patient recruitment, clinical data, and writing of the manuscript. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bichara, C., Berby, B., Rives, A. et al. Sperm chromatin condensation defects, but neither DNA fragmentation nor aneuploidy, are an independent predictor of clinical pregnancy after intracytoplasmic sperm injection. J Assist Reprod Genet 36, 1387–1399 (2019). https://doi.org/10.1007/s10815-019-01471-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-019-01471-4