Abstract
Purpose
The study aims to test the hypothesis that platelet-rich plasma (PRP) stimulates cellular processes involved in endometrial regeneration relevant to clinical management of poor endometrial growth or intrauterine scarring.
Methods
Human endometrial stromal fibroblasts (eSF), endometrial mesenchymal stem cells (eMSC), bone marrow-derived mesenchymal stem cells (BM-MSC), and Ishikawa endometrial adenocarcinoma cells (IC) were cultured with/without 5% activated (a) PRP, non-activated (na) PRP, aPPP (platelet-poor-plasma), and naPPP. Treatment effects were evaluated with cell proliferation (WST-1), wound healing, and chemotaxis Transwell migration assays. Mesenchymal-to-epithelial transition (MET) was evaluated by cytokeratin and vimentin expression. Differential gene expression of various markers was analyzed by multiplex Q-PCR.
Results
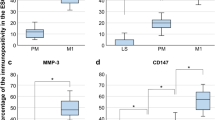
Activated PRP enhanced migration of all cell types, compared to naPRP, aPPP, naPPP, and vehicle controls, in a time-dependent manner (p < 0.05). The WST-1 assay showed increased stromal and mesenchymal cell proliferation by aPRP vs. naPRP, aPPP, and naPPP (p < 0.05), while IC proliferation was enhanced by aPRP and aPPP (p < 0.05). There was no evidence of MET. Expressions of MMP1, MMP3, MMP7, and MMP26 were increased by aPRP (p < 0.05) in eMSC and eSF. Transcripts for inflammation markers/chemokines were upregulated by aPRP vs. aPPP (p < 0.05) in eMSC and eSF. No difference in estrogen or progesterone receptor mRNAs was observed.
Conclusions
This is the first study evaluating the effect of PRP on different human endometrial cells involved in tissue regeneration. These data provide an initial ex vivo proof of principle for autologous PRP to promote endometrial regeneration in clinical situations with compromised endometrial growth and scarring.







Similar content being viewed by others
References
Mascarenhas MN, Flaxman SR, Boerma T, Vanderpoel S, Stevens GA. National, regional, and global trends in infertility prevalence since 1990: a systematic analysis of 277 health surveys. PLoS Med. 2012;9(12):e1001356. https://doi.org/10.1371/journal.pmed.1001356.
Horcajadas JA, Diaz-Gimeno P, Pellicer A, Simon C. Uterine receptivity and the ramifications of ovarian stimulation on endometrial function. Semin Reprod Med. 2007;25(6):454–60. https://doi.org/10.1055/s-2007-991043.
Labarta E, Martinez-Conejero JA, Alama P, Horcajadas JA, Pellicer A, Simon C, et al. Endometrial receptivity is affected in women with high circulating progesterone levels at the end of the follicular phase: a functional genomics analysis. Hum Reprod. 2011;26(7):1813–25. https://doi.org/10.1093/humrep/der126.
Moreno I, Codoner FM, Vilella F, Valbuena D, Martinez-Blanch JF, Jimenez-Almazan J, et al. Evidence that the endometrial microbiota has an effect on implantation success or failure. Am J Obstet Gynecol. 2016;215(6):684–703. https://doi.org/10.1016/j.ajog.2016.09.075.
Kumbak B, Erden HF, Tosun S, Akbas H, Ulug U, Bahceci M. Outcome of assisted reproduction treatment in patients with endometrial thickness less than 7 mm. Reprod BioMed Online. 2009;18(1):79–84.
Abdalla HI, Brooks AA, Johnson MR, Kirkland A, Thomas A, Studd JW. Endometrial thickness: a predictor of implantation in ovum recipients? Hum Reprod. 1994;9(2):363–5.
Dessolle L, Darai E, Cornet D, Rouzier R, Coutant C, Mandelbaum J, et al. Determinants of pregnancy rate in the donor oocyte model: a multivariate analysis of 450 frozen-thawed embryo transfers. Hum Reprod. 2009;24(12):3082–9. https://doi.org/10.1093/humrep/dep303.
Kasius A, Smit JG, Torrance HL, Eijkemans MJ, Mol BW, Opmeer BC, et al. Endometrial thickness and pregnancy rates after IVF: a systematic review and meta-analysis. Hum Reprod Update. 2014;20(4):530–41. https://doi.org/10.1093/humupd/dmu011.
Lebovitz O, Orvieto R. Treating patients with “thin” endometrium—an ongoing challenge. Gynecol Endocrinol. 2014;30(6):409–14. https://doi.org/10.3109/09513590.2014.906571.
Schenker JG. Etiology of and therapeutic approach to synechia uteri. Eur J Obstet Gynecol Reprod Biol. 1996;65(1):109–13.
Fernandez H, Al-Najjar F, Chauveaud-Lambling A, Frydman R, Gervaise A. Fertility after treatment of Asherman's syndrome stage 3 and 4. J Minim Invasive Gynecol. 2006;13(5):398–402. https://doi.org/10.1016/j.jmig.2006.04.013.
Nagori CB, Panchal SY, Patel H. Endometrial regeneration using autologous adult stem cells followed by conception by in vitro fertilization in a patient of severe Asherman’s syndrome. J Hum Reprod Sci. 2011;4(1):43–8. https://doi.org/10.4103/0974-1208.82360.
Amer MI, Abd-El-Maeboud KH, Abdelfatah I, Salama FA, Abdallah AS. Human amnion as a temporary biologic barrier after hysteroscopic lysis of severe intrauterine adhesions: pilot study. J Minim Invasive Gynecol. 2010;17(5):605–11. https://doi.org/10.1016/j.jmig.2010.03.019.
Santamaria X, Cabanillas S, Cervello I, Arbona C, Raga F, Ferro J, et al. Autologous cell therapy with CD133+ bone marrow-derived stem cells for refractory Asherman’s syndrome and endometrial atrophy: a pilot cohort study. Hum Reprod. 2016;31(5):1087–96. https://doi.org/10.1093/humrep/dew042.
Chen MJ, Yang JH, Peng FH, Chen SU, Ho HN, Yang YS. Extended estrogen administration for women with thin endometrium in frozen-thawed in-vitro fertilization programs. J Assist Reprod Genet. 2006;23(7–8):337–42. https://doi.org/10.1007/s10815-006-9053-1.
Weckstein LN, Jacobson A, Galen D, Hampton K, Hammel J. Low-dose aspirin for oocyte donation recipients with a thin endometrium: prospective, randomized study. Fertil Steril. 1997;68(5):927–30.
Sher G, Fisch JD. Effect of vaginal sildenafil on the outcome of in vitro fertilization (IVF) after multiple IVF failures attributed to poor endometrial development. Fertil Steril. 2002;78(5):1073–6.
Gleicher N, Kim A, Michaeli T, Lee HJ, Shohat-Tal A, Lazzaroni E, et al. A pilot cohort study of granulocyte colony-stimulating factor in the treatment of unresponsive thin endometrium resistant to standard therapies. Hum Reprod. 2013;28(1):172–7. https://doi.org/10.1093/humrep/des370.
Chang Y, Li J, Chen Y, Wei L, Yang X, Shi Y, et al. Autologous platelet-rich plasma promotes endometrial growth and improves pregnancy outcome during in vitro fertilization. Int J Clin Exp Med. 2015;8(1):1286–90.
Zadehmodarres S, Salehpour S, Saharkhiz N, Nazari L. Treatment of thin endometrium with autologous platelet-rich plasma: a pilot study. JBRA Assist Reprod. 2017;21(1):54–6. https://doi.org/10.5935/1518-0557.20170013.
Dragoo JL, Wasterlain AS, Braun HJ, Nead KT. Platelet-rich plasma as a treatment for patellar tendinopathy: a double-blind, randomized controlled trial. Am J Sports Med. 2014;42(3):610–8. https://doi.org/10.1177/0363546513518416.
Jo CH, Shin JS, Lee YG, Shin WH, Kim H, Lee SY, et al. Platelet-rich plasma for arthroscopic repair of large to massive rotator cuff tears: a randomized, single-blind, parallel-group trial. Am J Sports Med. 2013;41(10):2240–8. https://doi.org/10.1177/0363546513497925.
Cervelli V, Gentile P, Scioli MG, Grimaldi M, Casciani CU, Spagnoli LG, et al. Application of platelet-rich plasma in plastic surgery: clinical and in vitro evaluation. Tissue Eng Part C Methods. 2009;15(4):625–34. https://doi.org/10.1089/ten.TEC.2008.0518.
Picard F, Hersant B, Bosc R, Meningaud JP. The growing evidence for the use of platelet-rich plasma on diabetic chronic wounds: a review and a proposal for a new standard care. Wound Repair Regen. 2015;23(5):638–43. https://doi.org/10.1111/wrr.12317.
Langer HF, Stellos K, Steingen C, Froihofer A, Schonberger T, Kramer B, et al. Platelet derived bFGF mediates vascular integrative mechanisms of mesenchymal stem cells in vitro. J Mol Cell Cardiol. 2009;47(2):315–25. https://doi.org/10.1016/j.yjmcc.2009.03.011.
Stellos K, Seizer P, Bigalke B, Daub K, Geisler T, Gawaz M. Platelet aggregates-induced human CD34+ progenitor cell proliferation and differentiation to macrophages and foam cells is mediated by stromal cell derived factor 1 in vitro. Semin Thromb Hemost. 2010;36(2):139–45. https://doi.org/10.1055/s-0030-1251497.
Gargett CE, Chan RW, Schwab KE. Hormone and growth factor signaling in endometrial renewal: role of stem/progenitor cells. Mol Cell Endocrinol. 2008;288(1–2):22–9. https://doi.org/10.1016/j.mce.2008.02.026.
Matsumoto H, Nasu K, Nishida M, Ito H, Bing S, Miyakawa I. Regulation of proliferation, motility, and contractility of human endometrial stromal cells by platelet-derived growth factor. J Clin Endocrinol Metab. 2005;90(6):3560–7. https://doi.org/10.1210/jc.2004-1918.
Gargett CE, Masuda H. Adult stem cells in the endometrium. Mol Hum Reprod. 2010;16(11):818–34. https://doi.org/10.1093/molehr/gaq061.
Kaitu'u-Lino TJ, Ye L, Salamonsen LA, Girling JE, Gargett CE. Identification of label-retaining perivascular cells in a mouse model of endometrial decidualization, breakdown, and repair. Biol Reprod. 2012;86(6):184. https://doi.org/10.1095/biolreprod.112.099309.
Du H, Taylor HS. Contribution of bone marrow-derived stem cells to endometrium and endometriosis. Stem Cells. 2007;25(8):2082–6. https://doi.org/10.1634/stemcells.2006-0828.
Aghajanova L, Horcajadas JA, Esteban FJ, Giudice LC. The bone marrow-derived human mesenchymal stem cell: potential progenitor of the endometrial stromal fibroblast. Biol Reprod. 2010;82(6):1076–87. https://doi.org/10.1095/biolreprod.109.082867.
Ikoma T, Kyo S, Maida Y, Ozaki S, Takakura M, Nakao S, et al. Bone marrow-derived cells from male donors can compose endometrial glands in female transplant recipients. Am J Obstet Gynecol. 2009;201(6):608 e1–8. https://doi.org/10.1016/j.ajog.2009.07.026.
Kakudo N, Minakata T, Mitsui T, Kushida S, Notodihardjo FZ, Kusumoto K. Proliferation-promoting effect of platelet-rich plasma on human adipose-derived stem cells and human dermal fibroblasts. Plast Reconstr Surg. 2008;122(5):1352–60. https://doi.org/10.1097/PRS.0b013e3181882046.
Aghajanova L, Hamilton A, Kwintkiewicz J, Vo KC, Giudice LC. Steroidogenic enzyme and key decidualization marker dysregulation in endometrial stromal cells from women with versus without endometriosis. Biol Reprod. 2009;80(1):105–14.
Aghajanova L, Horcajadas JA, Weeks JL, Esteban FJ, Nezhat CN, Conti M, et al. The protein kinase a pathway-regulated transcriptome of endometrial stromal fibroblasts reveals compromised differentiation and persistent proliferative potential in endometriosis. Endocrinology. 2010;151(3):1341–55.
Chen JC, Hoffman JR, Arora R, Perrone LA, Gonzalez-Gomez CJ, Vo KC, et al. Cryopreservation and recovery of human endometrial epithelial cells with high viability, purity, and functional fidelity. Fertil Steril. 2016;105(2):501–10.e1. https://doi.org/10.1016/j.fertnstert.2015.10.011.
Irwin JC, Kirk D, King RJ, Quigley MM, Gwatkin RB. Hormonal regulation of human endometrial stromal cells in culture: an in vitro model for decidualization. Fertil Steril. 1989;52(5):761–8.
Spitzer TL, Rojas A, Zelenko Z, Aghajanova L, Erikson DW, Barragan F, et al. Perivascular human endometrial mesenchymal stem cells express pathways relevant to self-renewal, lineage specification, and functional phenotype. Biol Reprod. 2012;86(2):58. https://doi.org/10.1095/biolreprod.111.095885.
Barragan F, Irwin JC, Balayan S, Erikson DW, Chen JC, Houshdaran S, et al. Human endometrial fibroblasts derived from mesenchymal progenitors inherit progesterone resistance and acquire an inflammatory phenotype in the endometrial niche in endometriosis. Biol Reprod. 2016;94(5):118. https://doi.org/10.1095/biolreprod.115.136010.
Gurung S, Werkmeister JA, Gargett CE. Inhibition of transforming growth factor-beta receptor signaling promotes culture expansion of undifferentiated human endometrial mesenchymal stem/stromal cells. Sci Rep. 2015;5:15042. https://doi.org/10.1038/srep15042.
Aghajanova L, Stavreus-Evers A, Lindeberg M, Landgren BM, Sparre LS, Hovatta O. Thyroid-stimulating hormone receptor and thyroid hormone receptors are involved in human endometrial physiology. Fertil Steril. 2011;95(1):230–7. 7 e1-2
Cho HS, Song IH, Park SY, Sung MC, Ahn MW, Song KE. Individual variation in growth factor concentrations in platelet-rich plasma and its influence on human mesenchymal stem cells. Korean J Lab Med. 2011;31(3):212–8. https://doi.org/10.3343/kjlm.2011.31.3.212.
Kim DH, Je YJ, Kim CD, Lee YH, Seo YJ, Lee JH, et al. Can platelet-rich plasma be used for skin rejuvenation? Evaluation of effects of platelet-rich plasma on human dermal fibroblast. Ann Dermatol. 2011;23(4):424–31. https://doi.org/10.5021/ad.2011.23.4.424.
Kushida S, Kakudo N, Suzuki K, Kusumoto K. Effects of platelet-rich plasma on proliferation and myofibroblastic differentiation in human dermal fibroblasts. Ann Plast Surg. 2013;71(2):219–24. https://doi.org/10.1097/SAP.0b013e31823cd7a4.
Hulkower KI, Herber RL. Cell migration and invasion assays as tools for drug discovery. Pharmaceutics. 2011;3(1):107–24. https://doi.org/10.3390/pharmaceutics3010107.
Liao Y, He X, Qiu H, Che Q, Wang F, Lu W, et al. Suppression of the epithelial-mesenchymal transition by SHARP1 is linked to the NOTCH1 signaling pathway in metastasis of endometrial cancer. BMC Cancer. 2014;14:487. https://doi.org/10.1186/1471-2407-14-487.
Chen JC, Erikson DW, Piltonen TT, Meyer MR, Barragan F, McIntire RH, et al. Coculturing human endometrial epithelial cells and stromal fibroblasts alters cell-specific gene expression and cytokine production. Fertil Steril. 2013;100(4):1132–43. https://doi.org/10.1016/j.fertnstert.2013.06.007.
Tamaresis JS, Irwin JC, Goldfien GA, Rabban JT, Burney RO, Nezhat C, et al. Molecular classification of endometriosis and disease stage using high-dimensional genomic data. Endocrinology. 2014;155(12):4986–99. https://doi.org/10.1210/en.2014-1490.
Patterson AL, Zhang L, Arango NA, Teixeira J, Pru JK. Mesenchymal-to-epithelial transition contributes to endometrial regeneration following natural and artificial decidualization. Stem Cells Dev. 2013;22(6):964–50. https://doi.org/10.1089/scd.2012.0435.
Cousins FL, Murray A, Esnal A, Gibson DA, Critchley HO, Saunders PT. Evidence from a mouse model that epithelial cell migration and mesenchymal-epithelial transition contribute to rapid restoration of uterine tissue integrity during menstruation. PLoS One. 2014;9(1):e86378. https://doi.org/10.1371/journal.pone.0086378.
Deane JA, Gualano RC, Gargett CE. Regenerating endometrium from stem/progenitor cells: is it abnormal in endometriosis, Asherman’s syndrome and infertility? Curr Opin Obstet Gynecol. 2013;25(3):193–200. https://doi.org/10.1097/GCO.0b013e32836024e7.
Gurtner GC, Werner S, Barrandon Y, Longaker MT. Wound repair and regeneration. Nature. 2008;453(7193):314–21. https://doi.org/10.1038/nature07039.
van den Dolder J, Mooren R, Vloon AP, Stoelinga PJ, Jansen JA. Platelet-rich plasma: quantification of growth factor levels and the effect on growth and differentiation of rat bone marrow cells. Tissue Eng. 2006;12(11):3067–73. https://doi.org/10.1089/ten.2006.12.3067.
Lee JK, Lee S, Han SA, Seong SC, Lee MC. The effect of platelet-rich plasma on the differentiation of synovium-derived mesenchymal stem cells. J Orthop Res. 2014;32(10):1317–25. https://doi.org/10.1002/jor.22668.
Creeper F, Ivanovski S. Effect of autologous and allogenic platelet-rich plasma on human gingival fibroblast function. Oral Dis. 2012;18(5):494–500. https://doi.org/10.1111/j.1601-0825.2011.01897.x.
Dvorakova M, Nenutil R, Bouchal P. Transgelins, cytoskeletal proteins implicated in different aspects of cancer development. Expert Rev Proteomics. 2014;11(2):149–65. https://doi.org/10.1586/14789450.2014.860358.
Dos Santos Hidalgo G, Meola J, Rosa ESJC, Paro de Paz CC, Ferriani RA. TAGLN expression is deregulated in endometriosis and may be involved in cell invasion, migration, and differentiation. Fertil Steril. 2011;96(3):700–3. https://doi.org/10.1016/j.fertnstert.2011.06.052.
Zhang Q, Duan J, Liu X, Guo SW. Platelets drive smooth muscle metaplasia and fibrogenesis in endometriosis through epithelial-mesenchymal transition and fibroblast-to-myofibroblast transdifferentiation. Mol Cell Endocrinol. 2016;428:1–16. https://doi.org/10.1016/j.mce.2016.03.015.
Liu X, Shen M, Qi Q, Zhang H, Guo SW. Corroborating evidence for platelet-induced epithelial-mesenchymal transition and fibroblast-to-myofibroblast transdifferentiation in the development of adenomyosis. Hum Reprod. 2016;31(4):734–49. https://doi.org/10.1093/humrep/dew018.
Zhu B, Chen Y, Shen X, Liu X, Guo SW. Anti-platelet therapy holds promises in treating adenomyosis: experimental evidence. Reprod Biol Endocrinol. 2016;14(1):66. https://doi.org/10.1186/s12958-016-0198-1.
Zhang Q, Liu X, Guo SW. Progressive development of endometriosis and its hindrance by anti-platelet treatment in mice with induced endometriosis. Reprod BioMed Online. 2017;34(2):124–36. https://doi.org/10.1016/j.rbmo.2016.11.006.
Herndon CN, Aghajanova L, Balayan S, Erikson D, Barragan F, Goldfien G, et al. Global transcriptome abnormalities of the eutopic endometrium from women with adenomyosis. Reprod Sci. 2016;23(10):1289–303. https://doi.org/10.1177/1933719116650758.
Wang CC, Lee CH, Peng YJ, Salter DM, Lee HS. Platelet-rich plasma attenuates 30-kDa fibronectin fragment-induced chemokine and matrix metalloproteinase expression by meniscocytes and articular chondrocytes. Am J Sports Med. 2015;43(10):2481–9. https://doi.org/10.1177/0363546515597489.
Marini MG, Perrini C, Esposti P, Corradetti B, Bizzaro D, Riccaboni P, et al. Effects of platelet-rich plasma in a model of bovine endometrial inflammation in vitro. Reprod Biol Endocrinol. 2016;14(1):58. https://doi.org/10.1186/s12958-016-0195-4.
Reghini MF, Ramires Neto C, Segabinazzi LG, Castro Chaves MM, Dell’Aqua Cde P, Bussiere MC, et al. Inflammatory response in chronic degenerative endometritis mares treated with platelet-rich plasma. Theriogenology. 2016;86(2):516–22. https://doi.org/10.1016/j.theriogenology.2016.01.029.
Broughton G II, Janis JE, Attinger CE. The basic science of wound healing. Plast Reconstr Surg. 2006;117(7 Suppl):12S–34S. https://doi.org/10.1097/01.prs.0000225430.42531.c2.
Huang CC, Orvis GD, Wang Y, Behringer RR. Stromal-to-epithelial transition during postpartum endometrial regeneration. PLoS One. 2012;7(8):e44285. https://doi.org/10.1371/journal.pone.0044285.
Caley MP, Martins VL, O'Toole EA. Metalloproteinases and wound healing. Adv Wound Care (New Rochelle). 2015;4(4):225–34. https://doi.org/10.1089/wound.2014.0581.
Yan C, Boyd DD. Regulation of matrix metalloproteinase gene expression. J Cell Physiol. 2007;211(1):19–26. https://doi.org/10.1002/jcp.20948.
de Mos M, van der Windt AE, Jahr H, van Schie HT, Weinans H, Verhaar JA, et al. Can platelet-rich plasma enhance tendon repair? A cell culture study. Am J Sports Med. 2008;36(6):1171–8. https://doi.org/10.1177/0363546508314430.
Shin MK, Lee JW, Kim YI, Kim YO, Seok H, Kim NI. The effects of platelet-rich clot releasate on the expression of MMP-1 and type I collagen in human adult dermal fibroblasts: PRP is a stronger MMP-1 stimulator. Mol Biol Rep. 2014;41(1):3–8. https://doi.org/10.1007/s11033-013-2718-9.
Lange-Consiglio A, Cazzaniga N, Garlappi R, Spelta C, Pollera C, Perrini C, et al. Platelet concentrate in bovine reproduction: effects on in vitro embryo production and after intrauterine administration in repeat breeder cows. Reprod Biol Endocrinol. 2015;13:65. https://doi.org/10.1186/s12958-015-0064-6.
Zhang Q, Ding D, Liu X, Guo SW. Activated platelets induce estrogen receptor beta expression in endometriotic stromal cells. Gynecol Obstet Investig. 2015;80(3):187–92. https://doi.org/10.1159/000377629.
Acknowledgments
We thank Dr. Joshua Robinson, PhD (UCSF), for his help with time-lapse microscopy.
Financial support
Funding was provided by IntegraMed Fertility 2016 Research Grant (LA), NIH NCTRI P50HD055764 (LCG).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Supplemental Figure 1
Wound-healing assays for endometrial stromal fibroblasts (eSF, A), endometrial mesenchymal stem cells (eMSC, B), bone marrow-derived mesenchymal stem cells (BM-MSC, C) and Ishikawa cells (D). Representative images were obtained at 100× magnification. Images show the relative migration distance after incubation at different time points. aPRP = activated platelet rich plasma, naPRP = non-activated platelet rich plasma, aPPP = activated platelet poor plasma, naPPP = non-activated platelet poor plasma. (PDF 3982 kb)
Supplemental Figure 2
Transwell migration assays for endometrial stromal fibroblasts (eSF, A), endometrial mesenchymal stem cells (eMSC, B), bone marrow-derived mesenchymal stem cells (BM-MSC, C) and Ishikawa cells (D) at different time points. Representative images were obtained at 200× magnification. aPRP = activated platelet rich plasma, naPRP = non-activated platelet rich plasma, aPPP = activated platelet poor plasma, naPPP = non-activated platelet poor plasma. (PDF 2600 kb)
Rights and permissions
About this article
Cite this article
Aghajanova, L., Houshdaran, S., Balayan, S. et al. In vitro evidence that platelet-rich plasma stimulates cellular processes involved in endometrial regeneration. J Assist Reprod Genet 35, 757–770 (2018). https://doi.org/10.1007/s10815-018-1130-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-018-1130-8