Abstract
Purpose
To determine if microRNAs are differentially expressed in the follicular fluid of women with PCOS compared to fertile oocyte donors and identify associated altered gene expression.
Methods
Women undergoing IVF who met Rotterdam criteria for PCOS or who were fertile oocyte donors were recruited from a private IVF center. Individual follicle fluid was collected at the time of oocyte retrieval. MicroRNA analysis was performed using microarray and validated using real-time PCR on additional samples. Potential gene targets were identified and their expression analyzed by real time PCR.
Results
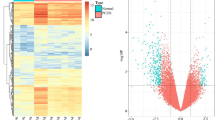
Microarray profiling of human follicular fluid revealed expression of 235 miRNAs, 29 were differentially expressed between the groups. Using PCR validation, 5 miRNAs (32, 34c, 135a, 18b, and 9) showed significantly increased expression in the PCOS group. Pathway analysis revealed genes involved in insulin regulation and inflammation. Three potential target genes were found to have significantly decreased expression in the PCOS group (interleukin 8, synaptogamin 1, and insulin receptor substrate 2).
Conclusions
MicroRNAs are differentially expressed in the follicular fluid of women with PCOS when compared to fertile oocyte donors. There is also altered expression of potential target genes associated with the PCOS phenotype.


Similar content being viewed by others
References
Fauser BC, Tarlatzis BC, Rebar RW, Legro RS, Balen AH, Lobo R, et al. Fauser BC, Tarlatzis BC, Rebar RW, Legro RS, Balen AH, Lobo R. Fertil Steril. 2012;97(1):28–38. doi:10.1016/j.fertnstert.2011.09.024. e25.
Health and fertility in World Health Organization group 2 anovulatory women. Hum Reprod Update. 2012;18(5):586-99. doi:10.1093/humupd/dms019.
Yildiz BO, Bozdag G, Yapici Z, Esinler I, Yarali H. Prevalence, phenotype and cardiometabolic risk of polycystic ovary syndrome under different diagnostic criteria. Hum Reprod. 2012. doi:10.1093/humrep/des232.
Koivunen R, Pouta A, Franks S, Martikainen H, Sovio U, Hartikainen AL, et al. Fecundability and spontaneous abortions in women with self-reported oligo-amenorrhea and/or hirsutism: Northern Finland Birth Cohort 1966 Study. Hum Reprod. 2008;23(9):2134–9. doi:10.1093/humrep/den136.
Sang Q, Yao Z, Wang H, Feng R, Zhao X, Xing Q, et al. Identification of microRNAs in human follicular fluid: characterization of microRNAs that govern steroidogenesis in vitro and are associated with polycystic ovary syndrome in vivo. J Clin Endocrinol Metab. 2013. doi:10.1210/jc.2013-1715.
Ambros V. microRNAs: tiny regulators with great potential. Cell. 2001;107(7):823–6.
McCallie B, Schoolcraft WB, Katz-Jaffe MG. Aberration of blastocyst microRNA expression is associated with human infertility. Fertil Steril. 2010;93(7):2374–82. doi:10.1016/j.fertnstert.2009.01.069.
Barbarotto E, Schmittgen TD, Calin GA. MicroRNAs and cancer: profile, profile, profile. Int J Cancer. 2008;122(5):969–77. doi:10.1002/ijc.23343.
Tatsuguchi M, Seok HY, Callis TE, Thomson JM, Chen JF, Newman M, et al. Expression of microRNAs is dynamically regulated during cardiomyocyte hypertrophy. J Mol Cell Cardiol. 2007;42(6):1137–41. doi:10.1016/j.yjmcc.2007.04.004.
Hawkins SM, Creighton CJ, Han DY, Zariff A, Anderson ML, Gunaratne PH, et al. Functional microRNA involved in endometriosis. Mol Endocrinol. 2011;25(5):821–32. doi:10.1210/me.2010-0371.
Wang C, Yang C, Chen X, Yao B, Zhu C, Li L, et al. Altered profile of seminal plasma microRNAs in the molecular diagnosis of male infertility. Clin Chem. 2011;57(12):1722–31. doi:10.1373/clinchem.2011.169714.
Osman A. MicroRNAs in health and disease–basic science and clinical applications. Clin Lab. 2012;58(5–6):393–402.
da Silveira JC, Veeramachaneni DN, Winger QA, Carnevale EM, Bouma GJ. cell-secreted vesicles in equine ovarian follicular fluid contain miRNAs and proteins: a possible new form of cell communication within the ovarian follicle. Biol Reprod. 2012;86(3):71. doi:10.1095/biolreprod.111.093252.
Weber JA, Baxter DH, Zhang S, Huang DY, Huang KH, Lee MJ, et al. The microRNA spectrum in 12 body fluids. Clin Chem. 2010;56(11):1733–41. doi:10.1373/clinchem.2010.147405.
Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA. 2008;105(30):10513–8. doi:10.1073/pnas.0804549105.
Gallo A, Tandon M, Alevizos I, Illei GG. The majority of microRNAs detectable in serum and saliva is concentrated in exosomes. PLoS One. 2012;7(3):e30679. doi:10.1371/journal.pone.0030679.
Ajit SK. Circulating microRNAs as biomarkers, therapeutic targets, and signaling molecules. Sensors (Basel). 2012;12(3):3359–69. doi:10.3390/s120303359.
Kuwabara Y, Ono K, Horie T, Nishi H, Nagao K, Kinoshita M, et al. Increased microRNA-1 and microRNA-133a levels in serum of patients with cardiovascular disease indicate myocardial damage. Circ Cardiovasc Genet. 2011;4(4):446–54. doi:10.1161/circgenetics.110.958975.
Zhang Y, Jia Y, Zheng R, Guo Y, Wang Y, Guo H, et al. Plasma microRNA-122 as a biomarker for viral-, alcohol-, and chemical-related hepatic diseases. Clin Chem. 2010;56(12):1830–8. doi:10.1373/clinchem.2010.147850.
Pfaffl MW, Horgan GW, Dempfle L. Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 2002;30(9):e36.
Burks DJ. Font de Mora J, Schubert M, Withers DJ, Myers MG, Towery HH et al. IRS-2 pathways integrate female reproduction and energy homeostasis. Nature. 2000;407(6802):377–82.
Runesson E, Ivarsson K, Janson PO, Brannstrom M. Gonadotropin- and cytokine-regulated expression of the chemokine interleukin 8 in the human preovulatory follicle of the menstrual cycle. J Clin Endocrinol Metab. 2000;85(11):4387–95.
Zhou Y, Zhu Y, Zhang S, Wang H, Wang S, Yang X. MicroRNA expression profiles in premature ovarian failure patients and its potential regulate functions. Chinese journal of birth health and heredity. 2011;19:20–2.
Neganova I, Al-Qassab H, Heffron H, Selman C, Choudhury AI, Lingard SJ, et al. Role of central nervous system and ovarian insulin receptor substrate 2 signaling in female reproductive function in the mouse. Biol Reprod. 2007;76(6):1045–53. doi:10.1095/biolreprod.106.059360.
Tucker WC, Chapman ER. Role of synaptotagmin in Ca2 + -triggered exocytosis. Biochem J. 2002;366(Pt 1):1–13. doi:10.1042/bj20020776.
Lang J, Fukuda M, Zhang H, Mikoshiba K, Wollheim CB. The first C2 domain of synaptotagmin is required for exocytosis of insulin from pancreatic beta-cells: action of synaptotagmin at low micromolar calcium. EMBO J. 1997;16(19):5837–46. doi:10.1093/emboj/16.19.5837.
Nakajima-Nagata N, Sugai M, Sakurai T, Miyazaki J, Tabata Y, Shimizu A. Pdx-1 enables insulin secretion by regulating synaptotagmin 1 gene expression. Biochem Biophys Res Commun. 2004;318(3):631–5. doi:10.1016/j.bbrc.2004.04.071.
Nyhlen K, Gautam C, Andersson R, Srinivas U. Modulation of cytokine-induced production of IL-8 in vitro by interferons and glucocorticosteroids. Inflammation. 2004;28(2):77–88.
Shimizu T, Kaji A, Murayama C, Magata F, Shirasuna K, Wakamiya K, et al. Effects of interleukin-8 on estradiol and progesterone production by bovine granulosa cells from large follicles and progesterone production by luteinizing granulosa cells in culture. Cytokine. 2012;57(1):175–81. doi:10.1016/j.cyto.2011.11.007.
Donadeu FX, Schauer SN, Sontakke SD. Involvement of miRNAs in ovarian follicular and luteal development. J Endocrinol. 2012;215(3):323–34. doi:10.1530/joe-12-0252.
Berezikov E. Evolution of microRNA diversity and regulation in animals. Nat Rev Genet. 2011;12(12):846–60. doi:10.1038/nrg3079.
DA Zawadski JK. Diagnostic for polycystic ovary syndrome: Towards a rational approach. Polycystic Ovary Syndrome (Current Issues in Endocrinology and Metabolism). Boston: Blackwell Scientific Inc.; 1992.
Azziz R, Carmina E, Dewailly D, Diamanti-Kandarakis E, Escobar-Morreale HF, Futterweit W, et al. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009;91(2):456–88. doi:10.1016/j.fertnstert.2008.06.035.
Abbott DH, Bacha F. Ontogeny of polycystic ovary syndrome and insulin resistance in utero and early childhood. Fertil Steril. 2013;100(1):2–11. doi:10.1016/j.fertnstert.2013.05.023.
Srivastava A, Goldberger H, Dimtchev A, Ramalinga M, Chijioke J, Marian C, et al. MicroRNA Profiling in Prostate Cancer - The Diagnostic Potential of Urinary miR-205 and miR-214. PLoS One. 2013;8(10):e76994. doi:10.1371/journal.pone.0076994.
Roncarati R, Anselmi CV, Losi MA, Papa L, Cavarretta E, Costa Martins PD, et al. Circulating miR-29a, Among Other Upregulated microRNAs, is the Only Biomarker for Both Hypertrophy and Fibrosis in Patients with Hypertrophic Cardiomyopathy. J Am Coll Cardiol. 2013. doi:10.1016/j.jacc.2013.09.041.
Aguado-Fraile E, Ramos E, Conde E, Rodriguez M, Liano F, Garcia-Bermejo ML. microRNAs in the kidney: Novel biomarkers of Acute Kidney Injury. Nefrologia. 2013. doi:10.3265/Nefrologia.pre2013.Aug.12198.
Kiko T, Nakagawa K, Tsuduki T, Furukawa K, Arai H, Miyazawa T. MicroRNAs in Plasma and Cerebrospinal Fluid as Potential Markers for Alzheimer’s Disease. J Alzheimers Dis. 2013. doi:10.3233/jad-130932.
Chen YH, Heneidi S, Lee JM, Layman LC, Stepp DW, Gamboa GM, et al. miRNA-93 inhibits GLUT4 and is overexpressed in adipose tissue of polycystic ovary syndrome patients and women with insulin resistance. Diabetes. 2013;62(7):2278–86. doi:10.2337/db12-0963.
Acknowledgments
We would like to acknowledge Andrew P. Bradford for his assistance with manuscript editing.
Conflict of interest
LWR received Clinical Research Fellowship and Mentor Award Supported by Pfizer, Inc. for research presented at ENDO 2012 and an ASRM Corporate Member Council In-training Travel Award for the IFFS/ASRM 2013. BM, RA, WBS, DM, and MGKJ have nothing to disclose.
Funding
This study was self-funded by the Colorado Center for Reproductive Medicine and the National Foundation for Fertility Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule Follicular fluid from PCOS patients and oocyte donors was compared for microRNA and target gene expression. Five microRNAs were overexpressed and 3 target genes were decreased in the PCOS group.
Rights and permissions
About this article
Cite this article
Roth, L.W., McCallie, B., Alvero, R. et al. Altered microRNA and gene expression in the follicular fluid of women with polycystic ovary syndrome. J Assist Reprod Genet 31, 355–362 (2014). https://doi.org/10.1007/s10815-013-0161-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-013-0161-4