Abstract
Purpose
Identification of the unique genes playing critical roles in human embryo cleavage.
Methods
Isolation of human ePAB cDNA using human ovary cDNA libraries and mouse ePAB amino acid sequences, followed by analysis of its expression pattern in various adult tissues and stages during early oocyte development excluding ePABP2.
Results
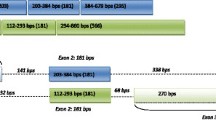
Human ePAB encodes a 330-aa protein and is located on chromosome 20q12-q13.1. The amino acid sequence is 72% homologous with that of mouse ePab. Human ePAB has only three RRMs and lacks a PABP domain; the expression pattern is nonspecific in adult tissues and detected in all stages, from oocyte to blastocyst. Human ePABP2 encodes a 282-aa protein and is located on chromosome 16q24.3. The amino acid sequence is 68% homologous with mouse ePabp2.
Conclusions
We identified human ePAB and ePABP2 cDNA. Human ePAB cDNA is not expressed specific to the ovary. Biological discrepancies exist between the human and the mouse.






Similar content being viewed by others
References
Van Voorthis BJ. In vitro fertilization. N Engl J Med. 2007;356:379–86.
Steptoe PC, Edwards RG. Birth after the reimplantation of a human embryo. Lancet 1978;2:366.
Papanikolaou EG, Camus M, Kolibianakis EM, Van Landuyt L, Van Steirteghem A, Devroey P. In vitro fertilization with single blastocyst-stage versus single cleavage-stage embryos. N Engl J Med. 2006;354:1139–46.
Gardner DK, Surrey E, Minjarez D, Leitz A, Stevens J, Schoolcraft WB. Single blastocyst transfer: a prospective randomized trial. Fertil Steril. 2004;81:551–5.
Cui SXS, Kim NH. Maternally derived transcripts: identification and characterisation during oocyte maturation and early cleravage. Reprod Fertil Dev. 2007;19:25–34.
Stebbins-Boaz B, Hake LE, Richter JD. CPEB controls the cytoplasmic polyadenylation of cyclin, Cdk2 and c-mos mRNAs and is necessary for oocyte maturation in Xenopus. EMBO J. 1996;15:2582–92.
Mendez R, Hake LE, Andresson T, Littlepage LE, Ruderman JV, Richter JD. Phosphorylation of CPE binding factor by Reg2 regulates translation of c-mos mRNA. Nature 2000;404:302–7.
Gebauer F, Xu W, Cooper GM, Richter JD. Translational control by cytoplasmic polyadenylation of c-mos mRNA is necessary for oocyte maturation in the mouse. EMBO J. 1994;13:5712–20.
Oh B, Hwang S, McLaughlin J, Solter D, Knowles BB. Timely translation during the mouse oocyte-to-embryo transition. Development 2000;127:3795–803.
Croisman I, Huang YS, Mendez R, Cao Q, Theurkauf W, Richter JD. CPEB, maskin, and cyclin B1 mRNA at the mitotic apparatus: implications for local translational control of cell division. Cell 2000;103:435–47.
Uto K, Sagata N. Nek2B, a novel maternal form of Nek2 kinase, is essential for the assembly or maintenance of centrosomes in early Xenopus embryos. EMBO J. 2000;19:1816–26.
Stutz A, Conne B, Huarte J, Gubler P, Volkel V, Flandin P, et al. Masking, unmasking, and regulated polyadenylation cooperate in the translational control of a dormant mRNA in mouse oocytes. Genes Dev. 1998;12:2535–48.
Richter JD, Lorenz LJ. Selective translation of mRNAs at synapses. Curr Opin Neurobiol. 2002;12:300–4.
Cao Q, Richter JD. Dissolution of the maskin-elF4E complex by cytoplasmic polyadenylation and poly(A)-binding protein controls cyclin B1 mRNA translation and oocyte maturation. EMBO J. 2002;21:3852–62.
Blobel G. A protein molecular weight 78,000 bound to the polyadenylate region of eukaryotic messenger RNAs. Proc Natl Acad Sci USA. 1973;70:924–28.
Mangus DA, Evans MC Jacobson A. Poly(A)-binding proteins: multifunctional scaffolds for the posttranscriptional control of gene expression. Genome Biol. 2003;4:223.
Kuhn U, Eahle E. Structure and function of poly(A) binding proteins. Biochim Biophys Acta. 2004;1678:67–84.
Wahle E. A novel poly(A)-binding protein acts as a specificity factor in the second phase of messenger RNA polyadenylation. Cell 1991;66:759–68.
Wahle E, Lustig A, Jeno P, Maurer P. Mammalian poly(A)-binding protein II. Physical properties and binding to polynucleotides. J Biol Chem. 1993;268:2937–45.
Good PJ, Abler L, Herring D, Sheets MD. Xenopus embryonic poly(A) binding protein 2 (ePABP2) defines a new family of cytoplasmic poly(A) binding proteins expressed during the early stages of vertebrate development. Genesis 2004;38:166–75.
Voeltz GK, Ongkasuwan J, Standart N, Steitz JA. A novel embryonic poly(A) binding protein, ePAB, regulates mRNA deadenylation in Xenopus egg extracts. Genes Dev. 2001;15:774–88.
Seli E, Lalioti MD, Flaherty SM, Sakkas D, Terzi N, Steitz JA. An embryonic poly(A)-binding protein (ePAB) is expressed in mouse oocytes and early preomplantation embryos. Proc Natl Acad Sci USA. 2005;102:367–72.
Melo EO, Dhalia R, de Sa CM, Standart N, de Melo Neto OP. Identification of a C-terminal poly(A)-binding protein (PABP)–PABP interaction domain. J Biol Chem. 2003;278:46357–68.
Hamatani T, Ko MSh, Yamada M, Kuji N, Mizusawa Y, Shoji M, et al. Global gene expression profiling of preimplantation embryos. Hum Cell. 2006;19:98–117.
Cosson B, Braun F, Paillard L, Blackshear P, Osborne HB. Identification of a novel Xenopus laevis poly(A) binding protein. Biol Cell. 2004;96:519–27.
Acknowledgments
This study was supported by a Grant-in-Aid for Scientific Research (No. 19591887) from the Ministries of Education, Culture, Sports, Science and Technology and Health, Labour and Welfare of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule
We successfully isolated the human ePAB and ePABP2 cDNAs and analyzed the expression patterns in humans.
Rights and permissions
About this article
Cite this article
Sakugawa, N., Miyamoto, T., Sato, H. et al. Isolation of the human ePAB and ePABP2 cDNAs and analysis of the expression patterns. J Assist Reprod Genet 25, 215–221 (2008). https://doi.org/10.1007/s10815-008-9220-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-008-9220-7