Abstract
Problem
Maternal stress perception is frequently alleged as a cause of infertility, miscarriages, late pregnancy complications or impaired fetal development. The purpose of the present review is to critically assess the biological and epidemiological evidence that considers the plausibility of a stress link to human reproductive failure.
Methods
All epidemiological studies published between 1980 and 2007 that tested the link between stress exposure and impaired reproductive success in humans were identified. Study outcomes were evaluated on the basis of how associations were predicted, tested and integrated with theories of etiology arising from recent scientific developments in the basic sciences. Further, published evidence arising from basic science research has been assessed in order to provide a mechanistic concept and biological evidence for the link between stress perception and reproductive success.
Results
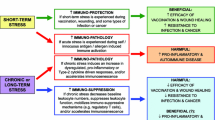
Biological evidence points to an immune–endocrine disequilibrium in response to stress and describes a hierarchy of biological mediators involved in a stress trigger to reproductive failure. Epidemiological evidence presents positive correlations between various pregnancy failure outcomes with pre-conception negative life events and elevated daily urinary cortisol. Strikingly, a relatively new conceptual approach integrating the two strands of evidence suggests the programming of stress susceptibility in mother and fetus via a so-called pregnancy stress syndrome.
Conclusions
An increasing specificity of knowledge is available about the types and impact of biological and social pathways involved in maternal stress responses. The present evidence is sufficient to warrant a reconsideration of conventional views on the etiology of reproductive failure. Physicians and patients will benefit from the adaptation of this integrated evidence to daily clinical practice.
Similar content being viewed by others
References
Frisch RE, McArthur JW. Menstrual cycles: fatness as a determinant as a minimum weight for height necessary for their maintenance or onset. Science 1974;185:949–51.
Genazzani AD, Ricchieri F, Lanzoni C, Strucchi C, Jasonni VM. Diagnostic and therapeutic approach to hypothalamic amenorrhea. Ann NY Acad Sci 2006;1092:103–13.
Woods NF, Lentz MJ, Mitchell ES, Heitkemper M, Shaver J, Henker R. Perceived stress, physiologic stress arousal, and premenstrual symptoms: group differences and intra-individual patterns. Res Nurs Health 1998;21:511–23.
Kaplan JR, Manuck SB. Ovarian function, stress and disease: a primate continuum. ILAR J 2004;45:89–97.
Mancuso RA, Dunkel Schetter C, Rini C, Roesch SC, Hobel CJ. Maternal prenatal anxiety and corticotropin releasing hormone associated with timing of delivery. Psychosom Med 2004;66:762–9.
Orr ST, James SA, Prince CB. Maternal prenatal depressive symptoms and spontaneous preterm births among African-American women in Baltimore, Maryland. Am J Epidemiol 2002;156:797–802.
Rice F, Jones I, Thapar A. The impact of gestational stress and prenatal growth on emotional problems in offspring: a review. Acta Psychiatr Scand 2007;116:154–5.
Chung TKH, Lau TK, Yip ASK, Chiu HFK, Lee DTS. Antepartum depressive symptomatology is associated with adverse obstetric and neonatal outcomes. Psychosom Med 2001;63:830–4.
Kossakowska-Petrycka K, Walecka-Matyja K. Psychological causative factors in postpartum depression amongst women with normal and high-risk pregnancies. Ginekol Pol 2007;78:544–8.
Riecher-Rossler A, Hofecker Fallahpour M. Postpartum depression: do we still need this diagnostic term? Acta Psychiatr Scand Suppl 2003;418:51–6.
Barsom SH, Mansfield PK, Koch PB, Gierach G, West SG. Association between psychological stress and menstrual cycle characteristics in perimenopausal women. Women’s Health Issues 2004;14:235–41.
Allsworth JE, Zierler S, Lapane KL, Krieger N, Hogan JW, Harlow BL. Longitudinal study of the inception of perimenopause in relation to lifetime history of sexual or physical violence. J Epidemiol Community Health 2004;58:938–43.
Barnea ER, Tal J. Stress-related reproductive failure. J In Vitro Fertil Embryo Transf 2001;8:15–23.
Neponmaschy PA, Sheiner E, Mastorakos G, Arck PC. Stress, immune function and reproduction. Ann NY Acad Sci 2007;1113:350–64.
Arck PC, Rose M, Hertwig K, Hagen E, Hildebrandt M, Klapp BF. Stress and immune mediators in miscarriage. Hum Reprod 2001;16:1505–11.
Arck PC, Knackstedt M, Blois S. NeuroEndocrineImmune circuitry challenging pregnancy maintenance and fetal health. J Reprod Med Endocrin 2004;2:98–102.
Szekeres-Bartho J. Immunological relationship between the mother and the fetus. Int Rev Immunol 2002;21:471–95.
Blois SM, Joachim R, Kandil J, Margni R, Tometten M, Klapp BF, et al. Depletion of CD8+ cells abolishes the pregnancy protective effect of progesterone substitution with dydrogesterone in mice by altering the Th1/Th2 cytokine profile. J Immunol 2004;172:5893–9.
Kalinka J, Szekeres-Bartho J. The impact of dydrogesterone supplementation on hormonal profile and progesterone-induced blocking factor concentrations in women with threatened abortion. Am J Reprod Immunol 2005;53:166–71.
Arck P, Hansen PJ, Jericevic BM, Piccinni MP, Szekeres-Bartho J. Progesterone during pregnancy: endocrine-immune cross talk in mammalian species and the role of stress. Am J Reprod Immunol 2007;58:268–79.
Chrousos GP. An integrated view of the stress response and stress-related behavioral and/or somatic disorders. Hans Selye Centennial Lecture, Proceedings of the Second World Conference on Stress, 2007, Budapest.
Chrousos GP. Neuroendocrinology of stress and female reproductive function. Proceedings of the 5th European Congress of Reproductive Immunology, 2007, Berlin.
Cacioppo JT, Berntson GG, Malarkey WB, Kiecolt-Glaser JK, Sheridan JF, Poehlmann KM, et al. Autonomic, neuroendocrine, and immune responses to psychological stress: the reactivity hypothesis. Ann N Y Acad Sci 1998;840:664–73.
Cannon WB. The emergency function of the adrenal medulla in pain and the major emotions. Am J Physiol 1914;33:356–72.
Qiu BS, Vallance BA, Blennerhassett PA, Collins SM. The role of CD4+ lymphocytes in the susceptibility of mice to stress-induced reactivation of experimental colitis. Nat Med 1999;5:1178–82.
Chandler N, Jacobson S, Esposito P, Conolly R, Theoharides TC. Acute stress shortens the time of onset of experimental allergic encephalomyelitis (EAE) in SJL/J mice. Brain Behav Immun 2002;16:757–63.
Cutolo M, Sulli A, Pizzorni C, Craviotto C, Straub RH. Hypothalamic–pituitary–adrenocortical and gonadal functions in rheumatoid arthritis. Ann NY Acad Sci 2003;992:107–17.
Blois SM, Tometten M, Kandil J, Hagen E, Klapp BF, Margni RA, Arck PC. ICAM-1/LFA-1 cross talk is a proximate mediator capable of disrupting immune integration and tolerance mechanism at the feto-maternal interface in murine pregnancies. J Immunol 2005;174:1820–9.
Blalock JE. The syntax of immune–neuroendocrine communication. Immunol Today 1994;15:504–11.
Glaser R, Kiecolt-Glaser JK. Stress-induced immune dysfunction: implications for health. Nat Rev Immunol 2005;5:243–51.
Aloe L, Alleva E, Bohm A, Levi-Montalcini R. Aggressive behavior induces release of nerve growth factor from mouse salivary gland into the bloodstream. Proc Natl Acad Sci U S A 1986;83:6184–7.
Peters EMJ, Handjiski B, Kuhlmei A, Hagen E, Bielas H, Braun A, et al. Neurogenic inflammation in stress-induced termination of murine hair growth is promoted by nerve growth factor. Am J Pathol 2004;165:259–71.
Tometten M, Blois S, Kuhlmei A, Stretz A, Klapp BF, Arck PC. Nerve growth factor translates stress response and subsequent murine abortion via adhesion molecule-dependent pathways. Biol Reprod 2006;74:674–83.
Paus R, Theoharides TC, Arck PC. Neuroimmunoendocrine circuitry of the ‘brain–skin connection’. Trends Immunol 2006;27:32–9.
Dahlström A, Mya-Tu M, Fuxe K, Zetterström BE. Observations on adrenergic innervation of dog heart. Am J Physiol 1965;209:689–92.
Sanders VM, Baker RA, Ramer-Quinn DS, Kasprowicz DJ, Fuchs BA, Street NE. Differential expression of the beta2- adrenergic receptor by Th1 and Th2 clones: implications for cytokine production and B cell help. J Immunol 1997;158:4200–10.
Dhabhar FS. Acute stress enhances while chronic stress suppresses skin immunity. The role of stress hormones and leukocyte trafficking. Ann NY Acad Sci 2000;917:876–93.
von Euler US, Gaddum JH. An unidendified depressor substance in certain tissue extracts. J Physiol (Lond) 1931;72:74–87.
Arck PC, Merali FS, Stanisz AM, Stead RH, Chaouat G, Manuel J, et al. Stress-induced murine abortion associated with substance P-dependent alteration in cytokines in maternal uterine decidua. Biol Reprod 1995;53:814–9.
Hunt SP, Mantyh PW. The molecular dynamics of pain control. Nat Rev Neurosci 2001;2:83–91.
Cao YQ, Mantyh PW, Carlson EJ, Gillespie AM, Epstein CJ, Basbaum AI. Primary afferent tachykinins are required to experience moderate to intense pain. Nature 1998;392:390–4.
Webster EL, Barrientos RM, Contoreggi C, Isaac MG, Ligier S, Garby KE, et al. Corticotropin releasing hormone (CRH) antagonist attenuates adjuvant induced arthritis: role of CRH in peripheral inflammation. J Rheumatol 2002;29:1252–61.
Chrousos GP. The hypothalamic–pituitary–adrenal axis and immune-mediated inflammation. N Engl J Med 1995;332:1351–62.
Theoharides TC, Singh LK, Boucher W, Pang X, Letourneau R, Webster E, et al. Corticotropin-releasing hormone induces skin mast cell degranulation and increased vascular permeability, a possible explanation for its pro-inflammatory effects. Endocrinology 1998;139:403–13.
Arck PC, Handjiski B, Peters EMJ, Peter AS, Hagen E, Fischer A, et al. Stress inhibits hair growth in mice by induction of premature catagen development and deleterious perifollicular inflammatory events via neuropeptide Substance P-dependent pathways. Am J Pathol 2003;162:803–14.
Raychaudhuri SP, Raychaudhuri SK. Role of NGF and neurogenic inflammation in the pathogenesis of psoriasis. Prog Brain Res 2004;146:433–7.
Dantzer R. Cytokine-induced sickness behaviour: a neuroimmune response to activation of innate immunity. Eur J Pharmacol 2004;500:399–411.
Watkins LR, Maier SF. The pain of being sick: implications of immune-to-brain communication for understanding pain. Annu Rev Psychol 2000;51:29–57.
Lindsay JR, Nieman LK. The Hypothalamic–Pituitary–Adrenal Axis, in Pregnancy: challenges in disease detection and treatment. Endocr Rev 2005;26:775–99.
De Herder WWW, Lamberts SWJ. Overview of hyper- and hypocortisolism. In: Margioris A, Chrousos G, editors. Adrenal disorders. Totowa: Humana Press; 2001.
Prior J. Estrogen’s storm season. Centre for menstrual cycle and ovulation research. Vancouver: University of British Columbia and VCHRI; 2005.
Burton GJ, Jauniaux E, Charnock-Jones DS. Human early placental development: potential roles of the endometrial glands. Placenta 2007;28:S64–69.
Mastorakos G, Ilias I. Maternal and fetal hypothalamic–pituitary–adrenal axes during pregnancy and postpartum. Ann NY Acad Sci 2003;997:136–49.
Kalantaridou SN, Zoumakis E, Makrigiannakis A, Godoy H, Chrousos GP. The role of corticotropin-releasing hormone in blastocyst implantation and early fetal immunotolerance. Horm Metab Res 2007;39:474–7.
Kalantaridou SN, Makrigiannakis A, Mastorakos G, Chrousos GP. Roles of reproductive corticotropin-releasing hormone. Ann NY Acad Sci 2003;997:129–35.
Makrigiannakis A, Zoumakis E, Kalantaridou S, Chrousos G, Gravanis A. Uterine and embryonic trophoblast CRH promotes implantation and maintenance of early pregnancy. Ann NY Acad Sci 2003;997:85–92.
Vitoratos N, Papatheodorou DC, Kalantaridou SN, Mastorakos G. Reproductive corticotropin-releasing hormone. Ann NY Acad Sci 2006;1092:310–8.
Glynn LM, Schetter CD, Chicz-DeMet A, Hobel CJ, Sandman CA. Ethnic differences in adrenocorticotropic hormone, cortisol and corticotropin-releasing hormone during pregnancy. Peptides 2007;28:1155–61.
Magiakou MA, Mastorakos G, Webster E, Chrousos GP. The hypothalamic–pituitary–adrenal axis and the female reproductive system. Ann NY Acad Sci 1997;816:42–56.
Wiebold JL, Stanfield PH, Becker WC, Hillers JK. The effect of restraint stress in early pregnancy in mice. J Reprod Fertil 1986;78:185–92.
Joachim R, Zenclussen AC, Polgar B, Douglas AJ, Fest S, Knackstedt M, et al. The progesterone derivative dydrogesterone abrogates murine stress-triggered abortion by inducing a Th2 biased local immune response. Steroids 2003;68:931–40.
Blois SM, Ilarregui JM, Tometten M, Garcia M, Orsal AS, Cordo-Russo R, et al. A pivotal role for galectin-1 in fetomaternal tolerance. Nat Med 2007;13:1450–7.
Szekeres-Bartho J, Polgar B, Kozma N, Miko E, Par G, Szereday L, et al. Progesterone-dependent immunomodulation. Chem Immunol Allergy 2005;89:118–25.
Raghupathy R, Al Mutawa E, Makhseed M, Azizieh F, Szekeres-Bartho J. Modulation of cytokine production by dydrogesterone in lymphocytes from women with recurrent miscarriage. BJOG 2005;1128:1096–101.
Omar MH, Mashita MK, Lim PS, Jamil MA. Dydrogesterone in threatened abortion: pregnancy outcome. J Steroid Biochem Mol Biol 2005;97:421–5.
O’Hare T, Creed F. Life events and miscarriage. Br J Psychiatry 1995;167:799–805.
Neugebauer R, Kline J, Stein Z, Shrout P, Warburton D, Susser M. Association of Stressful Life Events with Chromosomally Normal Spontaneous Abortion. Am J Epidemiol 1996;143:588–96.
Hjollund NH. Spontaneous abortion and physical strain around implantation: a follow-up study of first-pregnancy planners. Epidemiology 2000;11:18–23.
Hamilton Boyles S, Ness RB, Grisson AJ, Marcovic N, Bromberger J, CiFellie D. Life events stress and the association with spontaneous abortion in gravid women at an urban emergency department. Health Psychol 2000;19:510–4.
Sugiura-Ogasawara M, Furukawa TA, Nakano Y, Hori S, Aoki K, Kitamura T. Depression as a potential causal factor in subsequent miscarriage in recurrent spontaneous aborters. Hum Reprod 2002;17:2580–4.
Nepomnaschy PA, Welch K, McConnell DS, Strassman BI, England BG. Stress and female reproductive function: a study of daily variations in cortisol, gonadotrophins, and gonadal steroids in a rural Mayan population. Am J Human Biol 2004;16:523–32.
Bashour H, Abdul Salam A. Pregnancy outcomes and psychosocial stress. Int J Gynaecol Obstet 2001;73:1179–81.
Klonoff-Cohen H, Chu E, Natarajan L, Sieber W. A prospective study of stress among women undergoing in vitro fertilization or gamete intrafallopian transfer. Fertil Steril 2001;76:675–87.
Cooper BC, Gerber JR, McGettrick AL, Johnson JV. Perceived infertility-related stress correlates with in vitro fertilization outcome. Fertil Steril 2007;88:714–7.
Nelson DB, Grisso JA, Joffe MM, Brensinger C, Shaw L, Datner E. Does stress influence pregnancy loss. Ann Epidemiol 2003;13:223–9.
Bergant AM, Reinstadler K, Moncayo HE, Solder E, Heim K, Ulmer H. Spontaneous abortion and psychosomatics: a prospective study on the impact of psychological factors as a cause for recurrent spontaneous abortion. Hum Reprod 1997;12:1106–10.
Fenster L, Schaefer C, Mathur A, Hiatt RA, Pieper C, Hubbard AE, et al. Psychologic stress in the workplace and spontaneous abortion. Am J Epidemiol 1995;142:1176–83.
Kline J, Stein ZA, Susser M, Warburton D. Smoking: a risk factor for spontaneous abortion. New Engl J Med 1977;297:793–6.
Kline J, Stein Z, Shrout P, Susser M, Warburton D. Drinking during pregnancy and spontaneous abortion. Lancet 1980;2:176–80.
Stein Z, Susser M, Saenger G, Marolla F. The Dutch hunger winter of 1944–1945. New York: Oxford University Press; 1975.
Arck PC, Rücke M, Rose M, Szekeres-Bartho J, Douglas AJ, Pritsch M, et al. Early risk factors for spontaneous abortion: a prospective cohort study in pregnant women. Reprod Biol 2008;in press.
Elsenbruch S, Benson S, Rucke M, Rose M, Dudenhausen J, Pincus-Knackstedt MK, et al. Arck PC Social support during pregnancy: effects on maternal depressive symptoms, smoking and pregnancy outcomes. Hum Reprod 2007;22:869–77.
Nepomnaschy PA, Welch KB, McConnell DS, Low BS, Strassman BI, England BG. Cortisol levels and very early pregnancy loss in humans. PNAS 2006;103:3938–42.
Hjollund NH, Bonde JPE, Henriksen TB, Giwercman A, Olsen J. Reproductive effects of male psychologic stress. Epidemiology 2004;15:21–7.
Warburton D, Kline J, Stein Z, Hutzler M, Chin A, Hassold T. Does the karyotype of a spontaneous abortion predict the karyotype of a subsequent abortion? Evidence from 273 women with two karyotyped spontaneous abortions. Am J Hum Genet 1987;41:465–83.
Ogasawara M, Aoki K, Okada S, Suzumori K. Embryonic karyotype of abortuses in relation to the number of previous miscarriages. Fertil Steril 2000;73:300–4.
Morikawa M, Yamada H, Kato EH, Shimada S, Yamada T, Minakami H. Embryo loss pattern is predominant in miscarriages with normal chromosome karyotype among women with repeated miscarriage. Hum Reprod 2004;19:2644–7.
Stern JJ, Dorfmann AD, Gutierrez-Najar AJ, Cerrillo M, Coulam CB. Frequency of abnormal karyotypes among abortuses from women with and without a history of recurrent spontaneous abortion. Fertil Steril 1996;65:250–3.
Boivin J. A review of psychosocial interventions in infertility. Soc Sci Med 2003;57:2325–41.
Royal College of Obstetricians and Gynecologists. The Investigation and treatment of couples with recurrent miscarriage. 2003, Guideline No. 17, RCOG-UK.
Clifford K, Rai R, Regan L. Future pregnancy outcome in unexplained recurrent first trimester miscarriage. Hum Reprod 1997;12:387–9.
Stray Pedersen B, Stray Pedersen S. Etiologic factors and subsequent reproductive performance in 195 couples with a prior history of habitual abortion. Am J Obstet Gynecol 1984;148:140–6.
Aleman A, Althabe F, Belizán J, Bergel E. Bed rest during pregnancy for preventing miscarriage. Cochrane Database Syst Rev 2005;2:CD003576.
Lede R, Duley L. Uterine muscle relaxant drugs for threatened miscarriage. Cochrane Database Syst Rev 2005;3:CD002857.
Rumbold A, Middleton P, Crowther CA. Vitamin supplementation for preventing miscarriage. Cochrane Database Syst Rev 2005;2:CD004073.
Kramer MS, MacDonald SW. Aerobic exercise for women during pregnancy. Cochrane Database of Syst Rev 2006;3:CD000180.
Ancel P, Saurel-Cubizolles M, Di Renzo GC, Papiernik E, Breart G. Risk factors for 14–21 week abortions: a case control study in Europe. Hum Reprod 2000;15:2426–32.
Wadhwa PD. Psychoneuroendocrine processes in human pregnancy influence fetal development and health. Psychoneuroendocrinology 2005;8:724–43.
Federenko IS, Wadhwa PD. Women’s mental health during pregnancy influences fetal and infant developmental and health outcomes. CNS Spectr 2004;9:198–206.
Federenko IS, Nagamine M, Hellhammer DH, Wadhwa PD, Wust S. The heritability of hypothalamus pituitary adrenal axis responses to psychosocial stress is context dependent. J Clin Endocrinol Metab 2007;89:6244–50.
Nierop A, Bratsikas A, Klinkenberg A, Nater UM, Zimmerman R, Ehlert U. Prolonged salivary cortisol recovery in second-trimester pregnant women and attenuated salivary amylase responses to psychosocial stress in human pregnancy. J Clin Endocrinol Metab 2006;91:1329–35.
Heinrichs M, Baumgartner T, Kirschbaum C, Ehlert U. Social support and oxytocin interact to suppress cortisol and subjective responses to psychosocial stress. Biol Psychiatry 2003;54:1389–98.
Sata F, Yamada H, Kondo T, Gong Y, Tozaki S, Kobashi G, et al. Glutathione S-transferase M1 and T1 polymorphisms and the risk of recurrent pregnancy loss. Mol Hum Reprod 2003;9:165–9.
Sata F, Yamada H, Suzuki K, Saijo Y, Kato EH, Morikawa M, et al. Caffeine intake, CYP1A2 polymorphism and the risk of recurrent pregnancy loss. Mol Hum Reprod 2005;11:357–60.
Selman H, Mariani M, Barnocchi N, Mencacci A, Bistoni F, Arena S, et al. Examination of bacterial contamination at the time of embryo transfer, and its impact on the IVF/pregnancy outcome. J Assist Reprod Genet 2007;24:395–9.
Culhane JF, Rauh V, McCollum KF, Hogan VK, Agnew K, Wadhwa PD. Maternal stress is associated with bacterial vaginosis in human pregnancy. Matern Child Health J 2001;2:127–34.
Meaney MJ, Szyf M, Seckl JR. Epigenetic mechanisms of perinatal programming of hypothalamic–pituitary–adrenal function and health. Trends Mol Med 2007;13:269–77.
Budge H, Stephenson T, Symonds ME. Maternal nutrient restriction is not equivalent to maternal biological stress. Current Drug Targets 2007;8:888–93.
MacLaughlin SM, McMillen IC. Impact of periconceptional undernutrition on the development of the hypothalamo–pituitary–adrenal axis: does the timing of parturition start at conception. Current Drug Targets 2007;8:880–7.
Hougaard KS, Hansen AM. Enhancement of developmental toxicity effects of chemicals by gestational stress: a review. Neurotoxicol Teratol 2007;29:425–45.
Casey Jacob M. Psychological distress by type of fertility barrier. Hum Reprod 2006;22:885–94.
Ramezanzadeh F, Aghssa MM, Abedinia N, Zayeri F, Khanafshar N, Shariat Jafarabadi M. A survey of relationship between anxiety, depression and duration of infertility. BMC Womens Health 2004;4:9–18.
Born L, Soares CN, Phillips S, Jung M, Steiner M. Women and reproductive-related trauma. Ann NY Acad Sci 2006;1071:491–4.
Cai Y, Feng W. Famine, social disruption, and involuntary fetal loss: evidence from chinese survey data. Demography 2005;42:301–5.
Berczi I, Stephano AQ, Kovacs K. Stress, host defense and healing. Proceedings of the Second World Conference of Stress, 2007, Budapest.
Mujica-Parodi LR, Strey HH, Robert Savoy F, Cox D, Ravindranath B, Botanov Y. Second-hand stress: neurobiological evidence for a human alarm pheromone. Proceedings of the Second World Conference on Stress, 2007, Budapest.
Goncharova ND, Bogatyrenko TN. Diurnal and age changes in stress responsiveness of the hypothalamic–pituitary–adrenal (HPA) axis and erythrocyte antioxidant enzymes. Proceedings of the Second World Congress on Stress, 2007, Budapest.
Takai N, Yamaguchi M, Aragaki T, Eto K, Uchihashi K, Nishikawa Y. Gender-specific differences in salivary biomarker responses to acute psychological stress. Ann NY Acad Sci 2007;1098:510–5.
Obel C, Hedegaard M, Henriksen TB, Secher NJ, Olsen V, Levine S. Stress and salivary cortisol during pregnancy. Psychoneuroendocrinology 2005;30:647–6.
Symonds ME, Stephenson M, Gardner DS, Budge H. Long-term effects of nutritional programming of the embryo and fetus: mechanisms and critical windows. Reprod Fertil Dev 2007;19:53–3.
Nybo Andersen AM, Wohlfart J, Christens P, Olsen J, Melbye M. Maternal age and fetal loss: population based register linkage study. BMJ 2000;320:1708–12.
Edge D, Rogers A. Dealing with it: Black Caribbean women’s response to adversity and psychological distress associated with pregnancy, childbirth, and early motherhood. Soc Sci Med 2005;61:15–25.
Creel S, Christiansen D, Lilley S, Winnie JA Jr. Predation risk reduces reproductive physiology and demography of elk. Science 2007;315:960.
Wasser SK, Barash DP. Reproductive suppression among female mammals. Q Rev Biol 1983;58:513–38.
Packer C, Collins DA, Sindimwo A, Goodall J. Reproductive constraints on aggressive competition in female baboons. Nature 1995;373:60–3.
Barker DJP, Eriksson JG, Forsen T, Osmond C. Fetal origins of adult disease: strength of effects and biological basis. Int J Epidemiol 2002;31:1235–9.
Acknowledgements
Valuable assistance to KLK was provided by Dr. James Tansey, Assistant Professor and Chair of Business Ethics at the Sauder School of Business, UBC. The authors are grateful for the support provided by Mirjam Ruecke and Ronny Schwierzinski in performing a prospective cohort trial.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule
Scientific evidence in synthesis supports the plausibility of a stress trigger to human reproductive failure; higher risk may be explained by pregnancy stress syndrome.
Appendix
Appendix
Box 1
Stress and human reproductive failure: recommendations for future research | |
|---|---|
Prospective study design with multiple evaluation time points during pregnancy and a large number of patients (500 or more) | |
Development of measurable indices for pregnancy stress syndrome | |
Psychometric tools | |
Biological markers such as neurohormones (urinary cortisol, CRH), neuropeptides (substance P), sex/pregnancy-related hormones (progesterone, estradiol, βHCG) | |
Environmental risk assessment (noise, chemical exposure) | |
Indicators of increased susceptibility or resilience to a stress risk to pregnancy failure via hereditary, cultural/ethnic and gender factors | |
Genetic profile | |
Cultural/ethnic context of the study population | |
Consideration of risk subgroups combining variables suspected to confer high risk, for example | |
High stress perception with low social support, age over 30 years, low BMI, low progesterone and smoking |
Rights and permissions
About this article
Cite this article
Nakamura, K., Sheps, S. & Clara Arck, P. Stress and reproductive failure: past notions, present insights and future directions. J Assist Reprod Genet 25, 47–62 (2008). https://doi.org/10.1007/s10815-008-9206-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-008-9206-5