Abstract
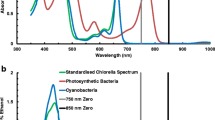
DMSO (dimethyl sulphoxide, (CH3)2SO) is an alternative solvent for spectroscopic assay of chlorophylls (Chls) but has mainly been used on Chl a & b organisms. Here, we develop algorithms for the estimation of Chl a, b & c1c2, and d in DMSO solvent. The common unicellular green alga Chlorella sp. (Chl a & b) is used as an example of an oxygenic photo-organism with Chl a as the primary photosynthetic pigment and Chl b as the accessory Chl. The cyanobacterium Synechococcus is used as an example of organisms containing only Chl a. The diatom Chaetoceros sp. is used as the representative Chl a & c1c2 organism. The unusual chlorobacterium Acaryochloris marina has Chl d & a. Algorithms for use on mixed phytoplankton and algal mats (Chl a, b & c) were also developed. The algorithms for DMSO solvent are compared to those developed for 90% acetone as the benchmark solvent. DMSO solvent offers the advantages of low volatility, low toxicity, low flammability, biodegradability, and ease of transport and is an effective extractant of chlorophylls. However, there appear to be significant storage problems with DMSO extracts of chlorophylls because of its high freezing point (+18.4°C) and probable breakdown in chlorophylls in DMSO over several days.




Similar content being viewed by others
References
Allen MM (1973) Methods for cyanobacteria. In: Stein JR (ed) Handbook of Phycological Methods: Culture Methods and Growth Measurements. Cambridge University Press, Cambridge, pp 127–138
Arnon PI (1949) Copper enzymes in isolated chloroplast. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Barnes JD, Balaguer L, Manrique E, Elvira S, Davison AW (1992) A reappraisal of the use of DMSO for the extraction and determination of chlorophylls a and b in lichens and higher plants. Environ Exp Bot 32:85–100
Cochran WG, Snedecor GW (1989) Statistical methods, 8th edn. Iowa State University Press, Ames 503p
Frigaard N-U, Larsen KL, Cox RP (1996) Spectrochromatography of photosynthetic pigments as a fingerprinting technique for microbial phototrophs. FEMS Microbiol Ecol 20:69–77
García AL, Nicholás N (1997) Establishing errors in the spectrophotometric determination of chlorophylls using different solvents. J Plant Physiol 152:392–398
Humphrey GF, Jeffrey SW (1997) Test of accuracy of spectrophotometric equations for the simultaneous determination of chlorophylls a, b, c1 and c2. In: Jeffrey SW, Mantoura RFC, Wright SW (eds) Phytoplankton Pigments in Oceanography: Guidelines to Modern Methods. UNESCO, Paris, pp 616–621
Jeffrey SW, Humphrey GF (1975) New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and phytoplankton. Biochem Physiol Pflanz (BPP) 167:191–194
Jeffrey SW, Welschmeyer NA (1997) Spectrophotometric and fluorometric equations in common use in oceanography. In: Jeffrey SW, Mantoura RFC, Wright SW (eds) Phytoplankton Pigments in Oceanography: Guidelines to Modern Methods. UNESCO, Paris, pp 597–615
Lee PA, de Mora SJ, Levasseur M (1999) A review of dimethylsulfoxide in aquatic environments. Atmosphere-Ocean 37:439–456
Li Y, Scales N, Blankenship RE, Willows RD, Chen M (2012) Extinction coefficient for red-shifted chlorophylls: chlorophyll d and chlorophyll f. Biochim Biophys Acta 1817:1292–1298
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Lichtenthaler HK, Wellburn AR (1983) Determination of total carotenoids and chlorophylls a and b in leaf extracts in different solvents. Biochem Soc Trans 11:591–592
McLachlan J (1973) Growth media – marine. In: Stein JR (ed) Handbook of Phycological Methods: Culture Methods and Growth Measurements. Cambridge University Press, Cambridge, pp 25–51
Porra RJ (1990) A simple method for extracting chlorophylls from the recalcitrant alga, Nannochloris atomus, without formation of spectroscopically-different magnesium-rhodochlorin derivatives. Biochim Biophys Acta 1019:137–141
Porra RJ (1991) Recent advances and reassessments in chlorophyll extraction and assay procedures for terrestrial, aquatic and marine organisms, including recalcitrant algae. In: Scheer H (ed) Chlorophylls. CRC Press, Boca Raton, pp 31–57
Porra RJ (2002) The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. Photosynth Res 73:633–640
Porra RJ (2006) Spectrophotometric assays for plant, algal and bacterial chlorophylls. In: Grimm B, Porra RJ, Ruediger W, Sheer H (eds) Chlorophylls and Bacteriochlorophylls: Biochemistry, Biophysics, Functions and Applications. Springer, Dordrecht, pp 95–106
Porra RJ (2011) A proven simultaneous equation assay for chlorophyll a and b using aqueous acetone and similar assays for recalcitrant algae. In: Roy S, Llewellyn CA, Egeland ES, Johnsen G (eds) Phytoplankton Pigments: Characterization, Chemotaxonomy and Applications in Oceanography. SCOR-UNESCO 2011, Appendix 8A. Cambridge University Press, Cambridge, pp 366–371
Porra RJ, Thompson WA, Kreidemann PE (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta 975:384–394
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stanier RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Ritchie RJ (2006) Consistent sets of spectrophotometric equations for acetone, methanol and ethanol solvents. Photosynth Res 89:27–41
Ritchie RJ (2008) Universal chlorophyll equations for estimating chlorophylls a, b, c & d and total chlorophylls in natural assemblages of photosynthetic organisms using acetone, methanol or ethanol solvents. Photosynthetica 46:115–126
Ritchie RJ (2018) Measurement of chlorophylls a and b and bacteriochlorophyll a in organisms from hypereutrophic auxinic waters. J Appl Phycol 30:3075–3087
Ritchie RJ, Sma-Air S (2020) Using integrating sphere spectrophotometry in unicellular algal research. J Appl Phycol 32:2947–2958
Ronen R, Galun M (1984) Pigment extraction from lichens with dimethylsulfoxide (DMSO) and estimation of chlorophyll degradation. Environ Exp Bot 24:239–245
Scheer H (1991) Structure and occurrence of chlorophylls. In: Scheer H (ed) Chlorophylls and Bacteriochlorophylls. CRC Press, Boca Raton, pp 3–30
Scheer H (2006) An overview of chlorophylls and bacteriochlorophylls: biochemistry, biophysics, junctions and applications. In: Scheer H (ed) Chlorophylls and Bacteriochlorophylls. Springer, Dordrecht, pp 1–26
Shinano T, Lei TT, Kawamukai T, Inoue MT, Koike T, Tadano T (1996) Dimethylsulfoxide method for the extraction of chlorophylls a and b from the leaves of wheat, field bean, dwarf bamboo and oak. Photosynthetica 32:409–414
Siebeneichler SC, Barbosa JS, Cruz AMM, Ramos MAD, Fernandes HE, Nacimento VL (2019) Comparison between extraction methods of photosynthetic pigments in Acacia mangium. Commun Plant Sci 9:1–5
Spiese CE, Kieber DJ, Nomura CT, Kiene RP (2009) Reduction of dimethylsulfoxide to dimethylsulfide by marine phytoplankton. Limnol Oceanogr 54:560–570
Sumata N, Haque CI, Nishika J, Suprakash R (2014) Spectrophotometric analysis of chlorophylls and carotenoids from commonly grown fern species by using various extraction solvents. Res J Chem Sci 4:63–69
Tait MA, Hik DS (2003) Is dimethylsulfoxide a reliable solvent for extracting chlorophyll under field conditions? Photosynth Res 78:87–91
ThermoFisher Scientific (2018) Safety data sheet: dimethyl sulf(ph)oxide: DMSO (D-139-1: D139-RS19: NC1115865), CAS-no- 67-68-5. Revision date 17-Jan-2018. Fisher Scientific, NJ, USA
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313
Acknowledgements
The Faculty of Environment and Technology of Prince of Songkla University-Phuket provided access to the facilities for the project as part of its policy of promoting the aquaculture industry in Southern Thailand. Andaman Environment and Natural Disaster Research Centre (ANED) also provided facilities and support. Phuket Marine Biological Centre (PMBC), Phuket, Thailand, kindly provided the cultures of Chlorella vulgaris and Chaetoceros sp.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ritchie, R.J., Sma-Air, S. & Phongphattarawat, S. Using DMSO for chlorophyll spectroscopy. J Appl Phycol 33, 2047–2055 (2021). https://doi.org/10.1007/s10811-021-02438-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-021-02438-8